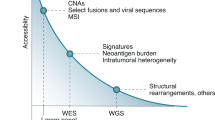
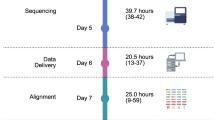
Abstract
Precision oncology applies genomic and other molecular analyses of tumor biopsies to improve the diagnosis and treatment of cancers. In addition to identifying therapeutic options, precision oncology tracks the response of a tumor to an intervention at the molecular level and detects drug resistance and the mechanisms by which it occurs. Integrative genomics can include sequencing specific panels of genes, exomes, or the entire triad of the patient's germline, tumor exome, and tumor transcriptome. Although the capabilities of sequencing technologies continue to improve, widespread adoption of genomics-driven precision oncology in the clinic has been held back by logistical, regulatory, financial, and ethical considerations. Nevertheless, integrative clinical sequencing programs applied at the point of care have the potential to improve the clinical management of cancer patients.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Beatson, G. On the treatment of inoperable cases of carcinoma of the mamma: suggestions for a new method of treatment, with illustrative cases. Lancet 148, 162–165 (1896).
Huggins, C., Stevens, R.E. Jr. & Hodges, C.V. Studies on prostatic cancer: Ii. the effects of castration on advanced carcinoma of the prostate gland. Arch. Surg. 43, 209–223 (1941).
Hanahan, D. & Weinberg, R.A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Mukherjee, S. The Emperor of All Maladies: A Biography of Cancer (Simon & Schuster, 2010).
Ledermann, J. et al. Olaparib maintenance therapy in platinum-sensitive relapsed ovarian cancer. N. Engl. J. Med. 366, 1382–1392 (2012).
Sharma, P. & Allison, J.P. The future of immune checkpoint therapy. Science 348, 56–61 (2015).
Collins, F.S. & Varmus, H. A new initiative on precision medicine. N. Engl. J. Med. 372, 793–795 (2015).
Varmus, H. The transformation of oncology. Science 352, 123 (2016).
McCarthy, M. US president endorses “moonshot” effort to cure cancer. Br. Med. J. 352, i213 (2016).
de Bono, J.S. & Ashworth, A. Translating cancer research into targeted therapeutics. Nature 467, 543–549 (2010).
Le Tourneau, C. et al. Molecularly targeted therapy based on tumour molecular profiling versus conventional therapy for advanced cancer (SHIVA): a multicentre, open-label, proof-of-concept, randomised, controlled phase 2 trial. Lancet Oncol. 16, 1324–1334 (2015).
Mullard, A. Use of personalized cancer drugs runs ahead of the science. Nature https://doi.org/10.1038/nature.2015.18389 (2015).
Kris, M.G. et al. Using multiplexed assays of oncogenic drivers in lung cancers to select targeted drugs. J. Am. Med. Assoc. 311, 1998–2006 (2014).
Pasche, B. & Grant, S.C. Non-small cell lung cancer and precision medicine: a model for the incorporation of genomic features into clinical trial design. J. Am. Med. Assoc. 311, 1975–1976 (2014).
Colwell, J. NCI-MATCH Trial Draws Strong Interest. Cancer Discov. 6, 334 (2016).
Abrams, J. et al. National Cancer Institute's Precision Medicine Initiatives for the new National Clinical Trials Network. Am. Soc. Clin. Oncol. Educ. Book 2014, 71–76 (2014).
Herbst, R.S. et al. Lung Master Protocol (Lung-MAP)-a biomarker-driven protocol for accelerating development of therapies for squamous cell lung cancer: SWOG S1400. Clin. Cancer Res. 21, 1514–1524 (2015).
Alden, R.S., Mandrekar, S.J. & Oxnard, G.R. Designing a definitive trial for adjuvant targeted therapy in genotype defined lung cancer: the ALCHEMIST trials. Chin. Clin. Oncol. 4, 37 (2015).
Govindan, R. et al. ALCHEMIST Trials: a golden opportunity to transform outcomes in early-stage non-small cell lung cancer. Clin. Cancer Res. 21, 5439–5444 (2015).
Brower, V. NCI-MATCH pairs tumor mutations with matching drugs. Nat. Biotechnol. 33, 790–791 (2015).
McNeil, C. NCI-MATCH launch highlights new trial design in precision-medicine era. J. Natl. Cancer Inst. 107, djv193 (2015).
Mullard, A. NCI-MATCH trial pushes cancer umbrella trial paradigm. Nat. Rev. Drug Discov. 14, 513–515 (2015).
Anonymous. NCI prepares to launch MATCH trial. Cancer Discov. 5, 685 (2015).
Chantrill, L.A. et al. Precision Medicine for Advanced Pancreas Cancer: The Individualized Molecular Pancreatic Cancer Therapy (IMPaCT) trial. Clin. Cancer Res. 21, 2029–2037 (2015).
Schwaederle, M. et al. Precision Oncology: The UC San Diego Moores Cancer Center PREDICT experience. Mol. Cancer Ther. 15, 743–752 (2016).
Meric-Bernstam, F. et al. A decision support framework for genomically informed investigational cancer therapy. J. Natl. Cancer Inst. 107, djv098 (2015).
Cheng, D.T. et al. Memorial Sloan Kettering-Integrated Mutation Profiling of Actionable Cancer Targets (MSK-IMPACT): a hybridization capture-based next-generation sequencing clinical assay for solid tumor molecular oncology. J. Mol. Diagn. 17, 251–264 (2015).
Hyman, D.M. et al. Precision medicine at Memorial Sloan Kettering Cancer Center: clinical next-generation sequencing enabling next-generation targeted therapy trials. Drug Discov. Today 20, 1422–1428 (2015).
Frampton, G.M. et al. Development and validation of a clinical cancer genomic profiling test based on massively parallel DNA sequencing. Nat. Biotechnol. 31, 1023–1031 (2013).
Robinson, D.R. et al. Integrative clinical genomics of metastatic cancer. Nature 548, 297–303 (2017).
Iyer, G. et al. Genome sequencing identifies a basis for everolimus sensitivity. Science 338, 221 (2012).
Wagle, N. et al. Activating mTOR mutations in a patient with an extraordinary response on a phase I trial of everolimus and pazopanib. Cancer Discov. 4, 546–553 (2014).
Al-Ahmadie, H. et al. Synthetic lethality in ATM-deficient RAD50-mutant tumors underlies outlier response to cancer therapy. Cancer Discov. 4, 1014–1021 (2014).
Lovly, C.M. et al. Rationale for co-targeting IGF-1R and ALK in ALK fusion-positive lung cancer. Nat. Med. 20, 1027–1034 (2014).
Van Allen, E.M. et al. Genomic correlate of exceptional erlotinib response in head and neck squamous cell carcinoma. JAMA Oncol. 1, 238–244 (2015).
Ahronian, L.G. et al. Clinical acquired resistance to RAF inhibitor combinations in BRAF-mutant colorectal cancer through MAPK pathway alterations. Cancer Discov. 5, 358–367 (2015).
Zaretsky, J.M. et al. Mutations associated with acquired resistance to PD-1 blockade in melanoma. N. Engl. J. Med. 375, 819–829 (2016).
Takebe, N., McShane, L. & Conley, B. Biomarkers: exceptional responders-discovering predictive biomarkers. Nat. Rev. Clin. Oncol. 12, 132–134 (2015).
Chang, D.K., Grimmond, S.M., Evans, T.R.J. & Biankin, A.V. Mining the genomes of exceptional responders. Nat. Rev. Cancer 14, 291–292 (2014).
Mehra, N., Lorente, D. & de Bono, J.S. What have we learned from exceptional tumour responses?: Review and perspectives. Curr. Opin. Oncol. 27, 267–275 (2015).
Arango, N.P. et al. A feasibility study of returning clinically actionable somatic genomic alterations identified in a research laboratory. Oncotarget 8, 41806–41814 (2017).
Beltran, H. et al. Whole-exome sequencing of metastatic cancer and biomarkers of treatment response. JAMA Oncol. 1, 466–474 (2015).
Perry, J.A. et al. Complementary genomic approaches highlight the PI3K/mTOR pathway as a common vulnerability in osteosarcoma. Proc. Natl. Acad. Sci. USA 111, E5564–E5573 (2014).
Bellmunt, J. et al. Somatic copy number abnormalities and mutations in PI3K/AKT/mTOR pathway have prognostic significance for overall survival in platinum treated locally advanced or metastatic urothelial tumors. PLoS One 10, e0124711 (2015).
Kim, Y. et al. Integrative and comparative genomic analysis of lung squamous cell carcinomas in East Asian patients. J. Clin. Oncol. 32, 121–128 (2014).
Liu, Y. et al. Clinical significance of CTNNB1 mutation and Wnt pathway activation in endometrioid endometrial carcinoma. J. Natl. Cancer Inst. 106, dju245 (2014).
Cancer Genome Atlas Network. Genomic classification of cutaneous melanoma. Cell 161, 1681–1696 (2015).
LoRusso, P.M. et al. Pilot trial of selecting molecularly guided therapy for patients with non-V600 BRAF-mutant metastatic melanoma: experience of the SU2C/MRA Melanoma Dream Team. Mol. Cancer Ther. 14, 1962–1971 (2015).
Sekulic, A. et al. Personalized treatment of Sézary syndrome by targeting a novel CTLA4:CD28 fusion. Mol. Genet. Genomic Med. 3, 130–136 (2015).
Roychowdhury, S. et al. Personalized oncology through integrative high-throughput sequencing: a pilot study. Sci. Transl. Med. 3, 111ra121 (2011).
ACS Commission on Cancer. Cancer Program Standards 2012: Ensuring Patient-Centered Care V1.0. (Chicago: American College of Surgeons; 2012).
Keating, N.L. et al. Tumor boards and the quality of cancer care. J. Natl. Cancer Inst. 105, 113–121 (2013).
Robinson, D. et al. Integrative clinical genomics of advanced prostate cancer. Cell 161, 1215–1228 (2015).
Brahmer, J.R. et al. Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N. Engl. J. Med. 366, 2455–2465 (2012).
Castle, J.C. et al. Exploiting the mutanome for tumor vaccination. Cancer Res. 72, 1081–1091 (2012).
Homet Moreno, B. & Ribas, A. Anti-programmed cell death protein-1/ligand-1 therapy in different cancers. Br. J. Cancer 112, 1421–1427 (2015).
Rizvi, N.A. et al. Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science 348, 124–128 (2015).
Schumacher, T.N. & Schreiber, R.D. Neoantigens in cancer immunotherapy. Science 348, 69–74 (2015).
Mody, R.J. et al. Integrative clinical sequencing in the management of refractory or relapsed cancer in youth. J. Am. Med. Assoc. 314, 913–925 (2015).
Schilsky, R.L. Implementing personalized cancer care. Nat. Rev. Clin. Oncol. 11, 432–438 (2014).
Farmer, H. et al. Targeting the DNA repair defect in BRCA mutant cells as a therapeutic strategy. Nature 434, 917–921 (2005).
Gelmon, K.A. et al. Olaparib in patients with recurrent high-grade serous or poorly differentiated ovarian carcinoma or triple-negative breast cancer: a phase 2, multicentre, open-label, non-randomised study. Lancet Oncol. 12, 852–861 (2011).
Ledermann, J. et al. Olaparib maintenance therapy in patients with platinum-sensitive relapsed serous ovarian cancer: a preplanned retrospective analysis of outcomes by BRCA status in a randomised phase 2 trial. Lancet Oncol. 15, 852–861 (2014).
Mateo, J. et al. DNA-repair defects and olaparib in metastatic prostate cancer. N. Engl. J. Med. 373, 1697–1708 (2015).
van der Noll, R. et al. Long-term safety and anti-tumour activity of olaparib monotherapy after combination with carboplatin and paclitaxel in patients with advanced breast, ovarian or fallopian tube cancer. Br. J. Cancer 113, 396–402 (2015).
Le, D.T. et al. PD-1 blockade in tumors with mismatch-repair deficiency. N. Engl. J. Med. 372, 2509–2520 (2015).
Lu, Y. et al. Most common 'sporadic' cancers have a significant germline genetic component. Hum. Mol. Genet. 23, 6112–6118 (2014).
Lu, C. et al. Patterns and functional implications of rare germline variants across 12 cancer types. Nat. Commun. 6, 10086 (2015).
Zhang, J. et al. Germline mutations in predisposition genes in pediatric cancer. N. Engl. J. Med. 373, 2336–2346 (2015).
Seifert, B.A. et al. Germline analysis from tumor-germline sequencing dyads to identify clinically actionable secondary findings. Clin. Cancer Res. 22, 4087–4094 (2016).
Pritchard, C.C. et al. Inherited DNA-repair gene mutations in men with metastatic prostate cancer. N. Engl. J. Med. 375, 443–453 (2016).
Newman, A.M. et al. Robust enumeration of cell subsets from tissue expression profiles. Nat. Methods 12, 453–457 (2015).
Lonigro, R.J. et al. Detection of somatic copy number alterations in cancer using targeted exome capture sequencing. Neoplasia 13, 1019–1025 (2011).
Davoli, T., Uno, H., Wooten, E.C. & Elledge, S.J. Tumor aneuploidy correlates with markers of immune evasion and with reduced response to immunotherapy. Science 355, aaf8399 (2017).
Zanetti, M. Chromosomal chaos silences immune surveillance. Science 355, 249–250 (2017).
Alexandrov, L.B. et al. Mutational signatures associated with tobacco smoking in human cancer. Science 354, 618–622 (2016).
Alexandrov, L.B. et al. Signatures of mutational processes in human cancer. Nature 500, 415–421 (2013).
Alexandrov, L.B. & Stratton, M.R. Mutational signatures: the patterns of somatic mutations hidden in cancer genomes. Curr. Opin. Genet. Dev. 24, 52–60 (2014).
Helleday, T., Eshtad, S. & Nik-Zainal, S. Mechanisms underlying mutational signatures in human cancers. Nat. Rev. Genet. 15, 585–598 (2014).
Behjati, S. et al. Mutational signatures of ionizing radiation in second malignancies. Nat. Commun. 7, 12605 (2016).
Roberts, S.A. & Gordenin, D.A. Hypermutation in human cancer genomes: footprints and mechanisms. Nat. Rev. Cancer 14, 786–800 (2014).
Topalian, S.L. et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N. Engl. J. Med. 366, 2443–2454 (2012).
Li, G.M. Mechanisms and functions of DNA mismatch repair. Cell Res. 18, 85–98 (2008).
Dudley, J.C., Lin, M.T., Le, D.T. & Eshleman, J.R. Microsatellite instability as a biomarker for PD-1 blockade. Clin. Cancer Res. 22, 813–820 (2016).
D'Andrea, A.D. & Grompe, M. The Fanconi anaemia/BRCA pathway. Nat. Rev. Cancer 3, 23–34 (2003).
Lord, C.J. & Ashworth, A. BRCAness revisited. Nat. Rev. Cancer 16, 110–120 (2016).
Turner, N., Tutt, A. & Ashworth, A. Hallmarks of 'BRCAness' in sporadic cancers. Nat. Rev. Cancer 4, 814–819 (2004).
Alexandrov, L.B., Nik-Zainal, S., Siu, H.C., Leung, S.Y. & Stratton, M.R. A mutational signature in gastric cancer suggests therapeutic strategies. Nat. Commun. 6, 8683 (2015).
Akashi-Tanaka, S. et al. BRCAness predicts resistance to taxane-containing regimens in triple negative breast cancer during neoadjuvant chemotherapy. Clin. Breast Cancer 15, 80–85 (2015).
Engert, F., Kovac, M., Baumhoer, D., Nathrath, M. & Fulda, S. Osteosarcoma cells with genetic signatures of BRCAness are susceptible to the PARP inhibitor talazoparib alone or in combination with chemotherapeutics. Oncotarget 8, 48794–48806 (2017).
Hong, S. et al. Complete durable response from carboplatin and olaparib in a heavily pretreated triple-negative metastatic breast cancer with germline BRCA2 and “BRCAness” mutations. J. Oncol. Pract. 12, 270–272 (2016).
Lorusso, D. et al. Prospective phase II trial of trabectedin in BRCA-mutated and/or BRCAness phenotype recurrent ovarian cancer patients: the MITO 15 trial. Ann. Oncol. 27, 487–493 (2016).
Waddell, N. et al. Whole genomes redefine the mutational landscape of pancreatic cancer. Nature 518, 495–501 (2015).
Henderson, S., Chakravarthy, A., Su, X., Boshoff, C. & Fenton, T.R. APOBEC-mediated cytosine deamination links PIK3CA helical domain mutations to human papillomavirus-driven tumor development. Cell Reports 7, 1833–1841 (2014).
Kuong, K.J. & Loeb, L.A. APOBEC3B mutagenesis in cancer. Nat. Genet. 45, 964–965 (2013).
Li, B. et al. Comprehensive analyses of tumor immunity: implications for cancer immunotherapy. Genome Biol. 17, 174 (2016).
Kreiter, S. et al. Mutant MHC class II epitopes drive therapeutic immune responses to cancer. Nature 520, 692–696 (2015).
Cancer Genome Atlas Research Network. Comprehensive molecular characterization of urothelial bladder carcinoma. Nature 507, 315–322 (2014).
Khoury, J.D. et al. Landscape of DNA virus associations across human malignant cancers: analysis of 3,775 cases using RNA-Seq. . J Virol. 87, 8916–8926 (2013).
Gubin, M.M. et al. Checkpoint blockade cancer immunotherapy targets tumour-specific mutant antigens. Nature 515, 577–581 (2014).
Tran, E. et al. T-cell transfer therapy targeting mutant KRAS in cancer. N. Engl. J. Med. 375, 2255–2262 (2016).
Rosenberg, S.A. et al. Durable complete responses in heavily pretreated patients with metastatic melanoma using T-cell transfer immunotherapy. Clin. Cancer Res. 17, 4550–4557 (2011).
Sharma, P., Hu-Lieskovan, S., Wargo, J.A. & Ribas, A. Primary, adaptive, and acquired resistance to cancer immunotherapy. Cell 168, 707–723 (2017).
Weinhold, N., Jacobsen, A., Schultz, N., Sander, C. & Lee, W. Genome-wide analysis of noncoding regulatory mutations in cancer. Nat. Genet. 46, 1160–1165 (2014).
Fredriksson, N.J., Ny, L., Nilsson, J.A. & Larsson, E. Systematic analysis of noncoding somatic mutations and gene expression alterations across 14 tumor types. Nat. Genet. 46, 1258–1263 (2014).
Versteege, I. et al. Truncating mutations of hSNF5/INI1 in aggressive paediatric cancer. Nature 394, 203–206 (1998).
Liu, X.S. & Mardis, E.R. Applications of immunogenomics to cancer. Cell 168, 600–612 (2017).
Rowley, J.D., Le Beau, M.M. & Rabbitts, T.H. Chromosomal Translocations and Genome Rearrangements in Cancer (Springer, 2015).
Kumar-Sinha, C., Kalyana-Sundaram, S. & Chinnaiyan, A.M. Landscape of gene fusions in epithelial cancers: seq and ye shall find. Genome Med. 7, 129 (2015).
Robinson, D.R. et al. Identification of recurrent NAB2-STAT6 gene fusions in solitary fibrous tumor by integrative sequencing. Nat. Genet. 45, 180–185 (2013).
Wu, Y.M. et al. Identification of targetable FGFR gene fusions in diverse cancers. Cancer Discov. 3, 636–647 (2013).
Antonarakis, E.S. et al. AR-V7 and resistance to enzalutamide and abiraterone in prostate cancer. N. Engl. J. Med. 371, 1028–1038 (2014).
Scher, H.I. et al. Association of AR-V7 on circulating tumor cells as a treatment-specific biomarker with outcomes and survival in castration-resistant prostate cancer. JAMA Oncol. 2, 1441–1449 (2016).
Wiesner, T. et al. Alternative transcription initiation leads to expression of a novel ALK isoform in cancer. Nature 526, 453–457 (2015).
Kong-Beltran, M. et al. Somatic mutations lead to an oncogenic deletion of met in lung cancer. Cancer Res. 66, 283–289 (2006).
Dhanasekaran, S.M. et al. Transcriptome meta-analysis of lung cancer reveals recurrent aberrations in NRG1 and Hippo pathway genes. Nat. Commun. 5, 5893 (2014).
Handorf, C.R. Gene expression analysis and immunohistochemistry in evaluation of cancer of unknown primary: time for a patient-centered approach. J. Natl. Compr. Canc. Netw. 9, 1415–1420 (2011).
Wei, I.H., Shi, Y., Jiang, H., Kumar-Sinha, C. & Chinnaiyan, A.M. RNA-Seq accurately identifies cancer biomarker signatures to distinguish tissue of origin. Neoplasia 16, 918–927 (2014).
Massard, C., Loriot, Y. & Fizazi, K. Carcinomas of an unknown primary origin--diagnosis and treatment. Nat. Rev. Clin. Oncol. 8, 701–710 (2011).
Matthew, E.M. et al. A multiplexed marker-based algorithm for diagnosis of carcinoma of unknown primary using circulating tumor cells. Oncotarget 7, 3662–3676 (2016).
Oien, K.A. & Dennis, J.L. Diagnostic work-up of carcinoma of unknown primary: from immunohistochemistry to molecular profiling. Ann. Oncol. 23(Suppl. 10), x271–x277 (2012).
Vincent, M., Perell, K., Nielsen, F.C., Daugaard, G. & Hansen, N.R. Modeling tissue contamination to improve molecular identification of the primary tumor site of metastases. Bioinformatics 30, 1417–1423 (2014).
Horn, S. et al. TERT promoter mutations in familial and sporadic melanoma. Science 339, 959–961 (2013).
Huang, F.W. et al. Highly recurrent TERT promoter mutations in human melanoma. Science 339, 957–959 (2013).
Borah, S. et al. Cancer. TERT promoter mutations and telomerase reactivation in urothelial cancer. Science 347, 1006–1010 (2015).
Reitman, Z.J., Pirozzi, C.J. & Yan, H. Promoting a new brain tumor mutation: TERT promoter mutations in CNS tumors. Acta Neuropathol. 126, 789–792 (2013).
Killela, P.J. et al. TERT promoter mutations occur frequently in gliomas and a subset of tumors derived from cells with low rates of self-renewal. Proc. Natl. Acad. Sci. USA 110, 6021–6026 (2013).
Vinagre, J. et al. Frequency of TERT promoter mutations in human cancers. Nat. Commun. 4, 2185 (2013).
Brat, D.J. et al. Comprehensive, integrative genomic analysis of diffuse lower-grade gliomas. N. Engl. J. Med. 372, 2481–2498 (2015).
Piscuoglio, S. et al. Massively parallel sequencing of phyllodes tumours of the breast reveals actionable mutations, and TERT promoter hotspot mutations and TERT gene amplification as likely drivers of progression. J. Pathol. 238, 508–518 (2016).
Abedalthagafi, M.S. et al. ARID1A and TERT promoter mutations in dedifferentiated meningioma. Cancer Genet. 208, 345–350 (2015).
Assié, G. et al. Integrated genomic characterization of adrenocortical carcinoma. Nat. Genet. 46, 607–612 (2014).
Wilson, B.G. & Roberts, C.W. SWI/SNF nucleosome remodellers and cancer. Nat. Rev. Cancer 11, 481–492 (2011).
Masliah-Planchon, J., Bièche, I., Guinebretière, J.M., Bourdeaut, F. & Delattre, O. SWI/SNF chromatin remodeling and human malignancies. Annu. Rev. Pathol. 10, 145–171 (2015).
Ronan, J.L., Wu, W. & Crabtree, G.R. From neural development to cognition: unexpected roles for chromatin. Nat. Rev. Genet. 14, 347–359 (2013).
Kandoth, C. et al. Integrated genomic characterization of endometrial carcinoma. Nature 497, 67–73 (2013).
Varela, I. et al. Exome sequencing identifies frequent mutation of the SWI/SNF complex gene PBRM1 in renal carcinoma. Nature 469, 539–542 (2011).
Cancer Genome Atlas Research Network. Comprehensive molecular characterization of clear cell renal cell carcinoma. Nature 499, 43–49 (2013).
Biegel, J.A. et al. Germ-line and acquired mutations of INI1 in atypical teratoid and rhabdoid tumors. Cancer Res. 59, 74–79 (1999).
Jackson, E.M. et al. Genomic analysis using high-density single nucleotide polymorphism-based oligonucleotide arrays and multiplex ligation-dependent probe amplification provides a comprehensive analysis of INI1/SMARCB1 in malignant rhabdoid tumors. Clin. Cancer Res. 15, 1923–1930 (2009).
Sévenet, N. et al. Constitutional mutations of the hSNF5/INI1 gene predispose to a variety of cancers. Am. J. Hum. Genet. 65, 1342–1348 (1999).
Gui, Y. et al. Frequent mutations of chromatin remodeling genes in transitional cell carcinoma of the bladder. Nat. Genet. 43, 875–878 (2011).
Guo, G. et al. Whole-genome and whole-exome sequencing of bladder cancer identifies frequent alterations in genes involved in sister chromatid cohesion and segregation. Nat. Genet. 45, 1459–1463 (2013).
Khuong-Quang, D.A. et al. K27M mutation in histone H3.3 defines clinically and biologically distinct subgroups of pediatric diffuse intrinsic pontine gliomas. Acta Neuropathol. 124, 439–447 (2012).
Lewis, P.W. et al. Inhibition of PRC2 activity by a gain-of-function H3 H3 mutation found in pediatric glioblastoma. Science 340, 857–861 (2013).
Schwartzentruber, J. et al. Driver mutations in histone H3.3 and chromatin remodelling genes in paediatric glioblastoma. Nature 482, 226–231 (2012).
Wu, G. et al. The genomic landscape of diffuse intrinsic pontine glioma and pediatric non-brainstem high-grade glioma. Nat. Genet. 46, 444–450 (2014).
Ahuja, N., Sharma, A.R. & Baylin, S.B. Epigenetic therapeutics: a new weapon in the war against cancer. Annu. Rev. Med. 67, 73–89 (2016).
Baylin, S.B. & Jones, P.A. A decade of exploring the cancer epigenome - biological and translational implications. Nat. Rev. Cancer 11, 726–734 (2011).
Krishnamurthy, N., Spencer, E., Torkamani, A. & Nicholson, L. Liquid biopsies for cancer: coming to a patient near you. J. Clin. Med. 6 doi:10.3390/jcm6010003 (2017).
Karachaliou, N. Mayo-de-Las-Casas, C., Molina-Vila, M.A. & Rosell, R. Real-time liquid biopsies become a reality in cancer treatment. Ann. Transl. Med. 3, 36 (2015).
Aravanis, A.M., Lee, M. & Klausner, R.D. Next-generation sequencing of circulating tumor DNA for early cancer detection. Cell 168, 571–574 (2017).
Uchida, J. et al. Diagnostic accuracy of noninvasive genotyping of EGFR in lung cancer patients by deep sequencing of plasma cell-free DNA. Clin. Chem. 61, 1191–1196 (2015).
Guo, N. et al. Circulating tumor DNA detection in lung cancer patients before and after surgery. Sci. Rep. 6, 33519 (2016).
Heitzer, E., Ulz, P., Geigl, J.B. & Speicher, M.R. Non-invasive detection of genome-wide somatic copy number alterations by liquid biopsies. Mol. Oncol. 10, 494–502 (2016).
Lamb, J. et al. The Connectivity Map: using gene-expression signatures to connect small molecules, genes, and disease. Science 313, 1929–1935 (2006).
Pavlova, N.N. & Thompson, C.B. The emerging hallmarks of cancer metabolism. Cell Metab. 23, 27–47 (2016).
Rolland, T. et al. A proteome-scale map of the human interactome network. Cell 159, 1212–1226 (2014).
Sohal, D.P. et al. Prospective clinical study of precision oncology in solid tumors. J. Natl. Cancer Inst. 108, djv332 (2015).
Wang, A.Z. Precision cancer medicine: hype or hope? Sci. Transl. Med. 7, 306ec164 (2015).
Simonds, N.I. et al. Comparative effectiveness research in cancer genomics and precision medicine: current landscape and future prospects. J. Natl. Cancer Inst. 105, 929–936 (2013).
Prasad, V., Fojo, T. & Brada, M. Precision oncology: origins, optimism, and potential. Lancet Oncol. 17, e81–e86 (2016).
Prasad, V. Perspective: the precision-oncology illusion. Nature 537, S63 (2016).
Massard, C. et al. High-throughput genomics and clinical outcome in hard-to-treat advanced cancers: results of the MOSCATO 01 trial. Cancer Discov. 7, 586–595 (2017).
Foster, M.W., Mulvihill, J.J. & Sharp, R.R. Evaluating the utility of personal genomic information. Genet. Med. 11, 570–574 (2009).
Green, R.C. et al. Clinical sequencing exploratory research consortium: accelerating evidence-based practice of genomic medicine. Am. J. Hum. Genet. 99, 246 (2016).
Beck, T.F., Mullikin, J.C. & Biesecker, L.G. Systematic evaluation of Sanger validation of next-generation sequencing variants. Clin. Chem. 62, 647–654 (2016).
Raymond, V.M. et al. Germline findings in tumor-only sequencing: points to consider for clinicians and laboratories. J. Natl. Cancer Inst. 108, djv351 (2015).
Amendola, L.M. et al. Performance of ACMG-AMP variant-interpretation guidelines among nine laboratories in the clinical sequencing exploratory research consortium. Am. J. Hum. Genet. 99, 247 (2016).
Jarvik, G.P. & Browning, B.L. Consideration of cosegregation in the pathogenicity classification of genomic variants. Am. J. Hum. Genet. 98, 1077–1081 (2016).
Parsons, D.W. et al. Diagnostic yield of clinical tumor and germline whole-exome sequencing for children with solid tumors. JAMA Oncol. 2, 616–624 (2016).
Amendola, L.M. et al. Actionable exomic incidental findings in 6503 participants: challenges of variant classification. Genome Res. 25, 305–315 (2015).
Shirts, B.H. et al. CSER and eMERGE: current and potential state of the display of genetic information in the electronic health record. J. Am. Med. Inform. Assoc. 22, 1231–1242 (2015).
Everett, J.N., Mody, R.J., Stoffel, E.M. & Chinnaiyan, A.M. Incorporating genetic counseling into clinical care for children and adolescents with cancer. Future Oncol. 12, 883–886 (2016).
Gray, S.W. et al. Social and behavioral research in genomic sequencing: approaches from the Clinical Sequencing Exploratory Research Consortium Outcomes and Measures Working Group. Genet. Med. 16, 727–735 (2014).
Lawler, M. et al. Sharing clinical and genomic data on cancer - the need for global solutions. N. Engl. J. Med. 376, 2006–2009 (2017).
Auffray, C. et al. Making sense of big data in health research: towards an EU action plan. Genome Med. 8, 71 (2016).
Wagle, N. et al. High-throughput detection of actionable genomic alterations in clinical tumor samples by targeted, massively parallel sequencing. Cancer Discov. 2, 82–93 (2012).
Beltran, H. et al. Targeted next-generation sequencing of advanced prostate cancer identifies potential therapeutic targets and disease heterogeneity. Eur. Urol. 63, 920–926 (2013).
Van Allen, E.M. et al. Whole-exome sequencing and clinical interpretation of formalin-fixed, paraffin-embedded tumor samples to guide precision cancer medicine. Nat. Med. 20, 682–688 (2014).
Cieslik, M. et al. The use of exome capture RNA-seq for highly degraded RNA with application to clinical cancer sequencing. Genome Res. 25, 1372–1381 (2015).
Gargis, A.S. et al. Assuring the quality of next-generation sequencing in clinical laboratory practice. Nat. Biotechnol. 30, 1033–1036 (2012).
Abecasis, G.R. et al. A map of human genome variation from population-scale sequencing. Nature 467, 1061–1073 (2010).
Auton, A. et al. A global reference for human genetic variation. Nature 526, 68–74 (2015).
Amberger, J.S., Bocchini, C.A., Schiettecatte, F., Scott, A.F. & Hamosh, A. OMIM.org: Online Mendelian Inheritance in Man (OMIM®), an online catalog of human genes and genetic disorders. Nucleic Acids Res. 43, D789–D798 (2015).
Fokkema, I.F., den Dunnen, J.T. & Taschner, P.E. LOVD: easy creation of a locus-specific sequence variation database using an “LSDB-in-a-box” approach. Hum. Mutat. 26, 63–68 (2005).
Fokkema, I.F. et al. LOVD v.2.0: the next generation in gene variant databases. Hum. Mutat. 32, 557–563 (2011).
Landrum, M.J. et al. ClinVar: public archive of interpretations of clinically relevant variants. Nucleic Acids Res. 44 D1, D862–D868 (2016).
Green, R.C. et al. ACMG recommendations for reporting of incidental findings in clinical exome and genome sequencing. Genet. Med. 15, 565–574 (2013).
Acknowledgements
This work was supported in part by the NIH Early Detection Research Network Award U01 CA214170, the NIH Clinical Sequencing Exploratory Research (CSER) Award NIH 1UM1HG006508, a Prostate SPORE Award P50 CA186786, and awards from the Prostate Cancer Foundation. A.M.C. is an American Cancer Society Research Professor, a Howard Hughes Medical Institute Investigator, and a Taubman Scholar of the University of Michigan. We thank S. Ellison, scientific writer, for editorial help with the manuscript and R. Kunkel for figure artwork. Helpful discussions with the members of the MI_Oncoseq team including D. Robinson, R. Lonigro, M. Cieslik, Y.-M. Wu, S.M. Dhanasekaran, P. Vats, and X. Cao are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
A.M.C. currently serves on the scientific advisory board of Tempus.
Supplementary information
Supplementary References
References for Figure 1 (PDF 289 kb)
Supplementary Table 1
Summary of actionable germline aberrations in cancer predisposition genes (XLSX 20 kb)
Supplementary Table 2a and 2b
A. Germline aberrations currently in clinical trials. Summary of actionable somatic aberrations in cancer genes. (XLSX 34 kb)
Rights and permissions
About this article
Cite this article
Kumar-Sinha, C., Chinnaiyan, A. Precision oncology in the age of integrative genomics. Nat Biotechnol 36, 46–60 (2018). https://doi.org/10.1038/nbt.4017
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.4017
This article is cited by
-
Deep learning in cancer genomics and histopathology
Genome Medicine (2024)
-
New clinical trial design in precision medicine: discovery, development and direction
Signal Transduction and Targeted Therapy (2024)
-
Understanding inequities in precision oncology diagnostics
Nature Cancer (2023)
-
Integrated genomic sequencing in myeloid blast crisis chronic myeloid leukemia (MBC-CML), identified potentially important findings in the context of leukemogenesis model
Scientific Reports (2022)
-
Whole-exome sequencing in eccrine porocarcinoma indicates promising therapeutic strategies
Cancer Gene Therapy (2022)