Abstract
Efficient genome editing with Cas9–sgRNA in vivo has required the use of viral delivery systems, which have limitations for clinical applications. Translational efforts to develop other RNA therapeutics have shown that judicious chemical modification of RNAs can improve therapeutic efficacy by reducing susceptibility to nuclease degradation. Guided by the structure of the Cas9–sgRNA complex, we identify regions of sgRNA that can be modified while maintaining or enhancing genome-editing activity, and we develop an optimal set of chemical modifications for in vivo applications. Using lipid nanoparticle formulations of these enhanced sgRNAs (e-sgRNA) and mRNA encoding Cas9, we show that a single intravenous injection into mice induces >80% editing of Pcsk9 in the liver. Serum Pcsk9 is reduced to undetectable levels, and cholesterol levels are significantly lowered about 35% to 40% in animals. This strategy may enable non-viral, Cas9-based genome editing in the liver in clinical settings.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Cox, D.B., Platt, R.J. & Zhang, F. Therapeutic genome editing: prospects and challenges. Nat. Med. 21, 121–131 (2015).
Jinek, M. et al. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 337, 816–821 (2012).
Cong, L. et al. Multiplex genome engineering using CRISPR/Cas systems. Science 339, 819–823 (2013).
Mali, P. et al. RNA-guided human genome engineering via Cas9. Science 339, 823–826 (2013).
Doudna, J.A. & Charpentier, E. Genome editing. The new frontier of genome engineering with CRISPR-Cas9. Science 346, 1258096 (2014).
Swiech, L. et al. In vivo interrogation of gene function in the mammalian brain using CRISPR-Cas9. Nat. Biotechnol. 33, 102–106 (2015).
Long, C. et al. Postnatal genome editing partially restores dystrophin expression in a mouse model of muscular dystrophy. Science 351, 400–403 (2016).
Nelson, C.E. et al. In vivo genome editing improves muscle function in a mouse model of Duchenne muscular dystrophy. Science 351, 403–407 (2016).
Tabebordbar, M. et al. In vivo gene editing in dystrophic mouse muscle and muscle stem cells. Science 351, 407–411 (2016).
Schumann, K. et al. Generation of knock-in primary human T cells using Cas9 ribonucleoproteins. Proc. Natl. Acad. Sci. USA 112, 10437–10442 (2015).
Ran, F.A. et al. In vivo genome editing using Staphylococcus aureus Cas9. Nature 520, 186–191 (2015).
Yin, H. et al. Therapeutic genome editing by combined viral and non-viral delivery of CRISPR system components in vivo. Nat. Biotechnol. 34, 328–333 (2016).
Wang, D. et al. Adenovirus-mediated somatic genome editing of Pten by CRISPR/Cas9 in mouse liver in spite of Cas9-specific immune responses. Hum. Gene Ther. 26, 432–442 (2015).
Kay, M.A. State-of-the-art gene-based therapies: the road ahead. Nat. Rev. Genet. 12, 316–328 (2011).
Chew, W.L. et al. A multifunctional AAV-CRISPR-Cas9 and its host response. Nat. Methods 13, 868–874 (2016).
Zuris, J.A. et al. Cationic lipid-mediated delivery of proteins enables efficient protein-based genome editing in vitro and in vivo. Nat. Biotechnol. 33, 73–80 (2015).
Wang, M., Glass, Z.A. & Xu, Q. Non-viral delivery of genome-editing nucleases for gene therapy. Gene Ther. 24, 144–150 (2017).
Behlke, M.A. Chemical modification of siRNAs for in vivo use. Oligonucleotides 18, 305–320 (2008).
Deleavey, G.F. & Damha, M.J. Designing chemically modified oligonucleotides for targeted gene silencing. Chem. Biol. 19, 937–954 (2012).
Yin, H. et al. Non-viral vectors for gene-based therapy. Nat. Rev. Genet. 15, 541–555 (2014).
Chiu, Y.-L. & Rana, T.M. rere siRNA function in RNAi: a chemical modification analysis. RNA 9, 1034–1048 (2003).
Hendel, A. et al. Chemically modified guide RNAs enhance CRISPR-Cas genome editing in human primary cells. Nat. Biotechnol. 33, 985–989 (2015).
Rahdar, M. et al. Synthetic CRISPR RNA-Cas9-guided genome editing in human cells. Proc. Natl. Acad. Sci. USA 112, E7110–E7117 (2015).
Gilbert, L.A. et al. CRISPR-mediated modular RNA-guided regulation of transcription in eukaryotes. Cell 154, 442–451 (2013).
Burnett, J.C., Rossi, J.J. & Tiemann, K. Current progress of siRNA/shRNA therapeutics in clinical trials. Biotechnol. J. 6, 1130–1146 (2011).
Nishimasu, H. et al. Crystal structure of Cas9 in complex with guide RNA and target DNA. Cell 156, 935–949 (2014).
Jiang, F., Zhou, K., Ma, L., Gressel, S. & Doudna, J.A. STRUCTURAL BIOLOGY. A Cas9-guide RNA complex preorganized for target DNA recognition. Science 348, 1477–1481 (2015).
Patra, A. et al. 2′-Fluoro RNA shows increased Watson-Crick H-bonding strength and stacking relative to RNA: evidence from NMR and thermodynamic data. Angew. Chem. Int. Ed. 51, 11863–11866 (2012).
Howard, J.A.K., Hoy, V.J., O'Hagan, D. & Smith, G.T. How good is fluorine as a hydrogen bond acceptor? Tetrahedron 52, 12613–12622 (1996).
Dong, Y. et al. Lipopeptide nanoparticles for potent and selective siRNA delivery in rodents and nonhuman primates. Proc. Natl. Acad. Sci. USA 111, 3955–3960 (2014).
Platt, R.J. et al. CRISPR-Cas9 knockin mice for genome editing and cancer modeling. Cell 159, 440–455 (2014).
Navarese, E.P. et al. Effects of proprotein convertase subtilisin/kexin type 9 antibodies in adults with hypercholesterolemia: a systematic review and meta-analysis. Ann. Intern. Med. 163, 40–51 (2015).
Yin, H., Cheng, L., Agarwal, C., Agarwal, R. & Ju, C. Lactoferrin protects against concanavalin A-induced liver injury in mice. Liver Int. 30, 623–632 (2010).
Robbins, M. et al. 2′-O-methyl-modified RNAs act as TLR7 antagonists. Mol. Ther. 15, 1663–1669 (2007).
Tsai, S.Q. et al. GUIDE-seq enables genome-wide profiling of off-target cleavage by CRISPR-Cas nucleases. Nat. Biotechnol. 33, 187–197 (2015).
Fu, Y. et al. High-frequency off-target mutagenesis induced by CRISPR-Cas nucleases in human cells. Nat. Biotechnol. 31, 822–826 (2013).
Zetsche, B. et al. Cpf1 is a single RNA-guided endonuclease of a class 2 CRISPR-Cas system. Cell 163, 759–771 (2015).
Si-Tayeb, K., Lemaigre, F.P. & Duncan, S.A. Organogenesis and development of the liver. Dev. Cell 18, 175–189 (2010).
Chen, H.Z. et al. Canonical and atypical E2Fs regulate the mammalian endocycle. Nat. Cell Biol. 14, 1192–1202 (2012).
Suzuki, K. et al. In vivo genome editing via CRISPR/Cas9 mediated homology-independent targeted integration. Nature 540, 144–149 (2016).
Dominguez, A.A., Lim, W.A. & Qi, L.S. Beyond editing: repurposing CRISPR-Cas9 for precision genome regulation and interrogation. Nat. Rev. Mol. Cell Biol. 17, 5–15 (2016).
Chen, D. et al. Rapid discovery of potent siRNA-containing lipid nanoparticles enabled by controlled microfluidic formulation. J. Am. Chem. Soc. 134, 6948–6951 (2012).
Kauffman, K.J. et al. Optimization of lipid nanoparticle formulations for mRNA delivery in vivo with fractional factorial and definitive screening designs. Nano Lett. 15, 7300–7306 (2015).
Bolukbasi, M.F. et al. DNA-binding-domain fusions enhance the targeting range and precision of Cas9. Nat. Methods 12, 1150–1156 (2015).
Brinkman, E.K., Chen, T., Amendola, M. & van Steensel, B. Easy quantitative assessment of genome editing by sequence trace decomposition. Nucleic Acids Res. 42, e168 (2014).
Yin, H., Cheng, L., Langenbach, R. & Ju, C. Prostaglandin I(2) and E(2) mediate the protective effects of cyclooxygenase-2 in a mouse model of immune-mediated liver injury. Hepatology 45, 159–169 (2007).
Yin, H. et al. Lactoferrin protects against acetaminophen-induced liver injury in mice. Hepatology 51, 1007–1016 (2010).
Zhu, L.J. et al. GUIDEseq: a bioconductor package to analyze GUIDE-Seq datasets for CRISPR-Cas nucleases. BMC Genomics 18, 379 (2017).
Acknowledgements
We thank T. Jacks, P. Sharp, T. Tammela, Z. Weng, G. Gao, E. Sontheimer and A. Vegas for discussions and for sharing reagents, Y. Li and A. Park for technical assistance, and K. Cormier for histology. This work is supported by grants from the National Institutes of Health (NIH), 5R00CA169512, DP2HL137167 and P01HL131471 (to W.X.). V.K. acknowledges support from the Russian Scientific Fund, Grant number 14-34-00017. H.Y. is supported by Skoltech Center and 5-U54-CA151884-04 (NIH). This work is supported in part by Cancer Center Support (core) grant P30-CA14051 from the NIH. We thank the Swanson Biotechnology Center for technical support.
Author information
Authors and Affiliations
Contributions
H.Y. conceived of and designed the study, and directed the project. H.Y., S.S., C.-Q.S., S.W., L.H.R., K.K., H.M., Q.W., E.M., M.F.B., L.J.Z., S.-Y.K., T.Z., S.A.W., A.O., J.D., R.L.B. and W.X. performed experiments and analyzed data. C.-Q.S. made the figures with H.Y. W.X., V.K. and R.L. provided conceptual advice. H.Y. wrote the manuscript with comments from all authors. D.G.A. supervised the project.
Corresponding author
Ethics declarations
Competing interests
H.Y., D.G.A. and R.L. have applied for patents related to this study. D.G.A. is a scientific co-founder of CRISPR Therapeutics.
Integrated supplementary information
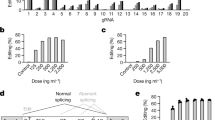
Supplementary Figure 1 Determine the activities of chemically modified sgRNAs in cells.
HEK293 cells stably expressing both EF1α promoter-GFP and EFs promoter-spCas9 were incubated with a GFP targeting sgRNA with various chemical modifications or without modification (unmodified, as Native Strand). a) FACS analysis was performed after 6-7 days. Indels at GFP locus in total DNA from HEK293 cells were determined by b) T7EI assay and c) TIDE analysis. n = 4.
Supplementary Figure 2 Determine the activities of unmodified, 5'&3' modified and e-sgRNA in cells.
a) HEK293 cells expressing both GFP and spCas9 were incubated with various doses of unmodified, 5'&3' modified and e-sgRNA targeting GFP. TIDE analysis was performed to determine indels at GFP locus. b) HEK293 cells expressing GFP were incubated with Cas9 mRNA and e-sgRNA, or Cas9 protein/e-sgRNA Ribonucleoproteins (RNP). TIDE analysis was performed to determine indels at GFP locus. n = 4.
Supplementary Figure 3 In vivo delivery of chemical modified sgRNAs and Cas9 mRNA induced knockout of targeted gene.
C57BL/6 mice were i.v. injected with two e-sgRNAs targeting Pcsk9 (PCSK9-1 & 2) and Cas9 mRNA encapsulated in lipid nanoparticles (Fig. 4). PCR was performed using two pairs of primers. The size of uncut and deletion bands (red pairs) is indicated. Both primer pairs show similar level of deletion band. Gel images were cropped. See full length gels with molecular weight standard in Supplementary information
Supplementary Figure 4 Determine the genome editing efficiency in hepatocytes and hepatic nonparenchymal cells.
C57BL/6 female mice were injected with lipid nanoparticles encapsulated with two e-sgRNAs targeting Pcsk9 and Cas9 mRNA. Hepatocytes enriched population and hepatic nonparenchymal cells (NPC) were isolated from the liver. Indels at Pcsk9 locus in total DNA from the liver, hepatocytes enriched population and NPC were measured by T7EI assay. Red arrow indicates PCR fragment with deletion, and blue arrow indicates cleaved fragment. n = 3. Gel images were cropped. See full length gels with molecular weight standard in Supplementary information
Supplementary Figure 5 Determine the activities of unmodified, 5'&3' modified and e-sgRNA in vivo.
C57BL/6 female mice were injected with lipid nanoparticles encapsulated with two unmodified, 5'&3'sgRNAs or e-sgRNAs targeting Pcsk9 and Cas9 mRNA. Indels at Pcsk9 locus in total DNA from the liver were measured by T7EI assay. Red arrow indicates PCR fragment with deletion, and blue arrow indicates cleaved fragment. n = 4 for native sgRNA and 5'&3' sgRNA groups, n=3 for e-sgRNA group. Gel images were cropped. See full length gels with molecular weight standard in Supplementary information
Supplementary Figure 6 Determine the activities of unmodified sgRNA targeting Fah in FAHmut/mut mice.
a) The unmodified sgRNA targeting Fumarylacetoacetate hydrolase (Fah) was formulated into LNP (nano.sgRNA) and co-injected with Cas9 mRNA encapsulated into LNP (nano.Cas9). FAHmut/mut mice were kept on NTBC water and euthanized 7 days after treatment to estimate indels rate. b) Indels of total DNA from liver by Illumina sequencing. n = 3.
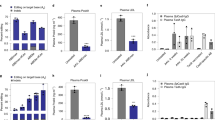
Supplementary Figure 7 Determine the toxicity in mice after CRISPR treatment.
Mice were injected with lipid nanoparticles encapsulated with two e-sgRNAs targeting Pcsk9 and Cas9 mRNA. a) H&E staining of liver 5 days after injection. b) Body weight before and 10 days after injections. c-e) Serum biomarkers c) 5 days, d) 1 day and e) 18 days after injection. f) H&E staining of liver 1 days and 18 days after injection. g) Serum cytokines and chemokines 1 day after injection. n = 4. Scale bars are 200μm.
Supplementary Figure 8 Determine the activity of genome editing in lung and spleen of mice after CRISPR treatment.
C57BL/6 or FAHmut/mut mice were injected with lipid nanoparticles encapsulated with e-sgRNAs targeting Pcsk9, or Fah (for FAHmut/mut mice) or ROSA26 and Cas9 mRNA. Indels at a) Pcsk9, b) Fah and c) ROSA26 loci in total DNA from the lung and spleen were measured by T7EI assay. n = 3. Gel images were cropped. See full length gels with molecular weight standard in Supplementary information
Supplementary Figure 9 Determine the indels at the Fah sgRNA's off-target site obtained from GUIDE-seq.
FAHmut/mut mice were injected with lipid nanoparticles encapsulated with e-sgRNA targeting Fah and Cas9 mRNA. Indels at off-target site 1 (OT1) concluded from GUIDE-seq were measured by T7EI assay. n = 4. Gel images were cropped. See full length gels with molecular weight standard in Supplementary information
Supplementary Figure 10 Full-length gels and blots with molecular weight standards for Supplementary Figures.
a) Full-length gels for Supplementary Figure 3; b-c) Full-length gels for Supplementary Figures 4 and 5; d-f) Full-length gels for Supplementary Figure 8; g) Full-length gels for Supplementary Figure 9
Supplementary information
Rights and permissions
About this article
Cite this article
Yin, H., Song, CQ., Suresh, S. et al. Structure-guided chemical modification of guide RNA enables potent non-viral in vivo genome editing. Nat Biotechnol 35, 1179–1187 (2017). https://doi.org/10.1038/nbt.4005
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.4005
This article is cited by
-
CRISPR technologies for genome, epigenome and transcriptome editing
Nature Reviews Molecular Cell Biology (2024)
-
Improving prime editing with an endogenous small RNA-binding protein
Nature (2024)
-
Durable and efficient gene silencing in vivo by hit-and-run epigenome editing
Nature (2024)
-
In vivo LNP-CRISPR Approaches for the Treatment of Hemophilia
Molecular Diagnosis & Therapy (2024)
-
Enrichment strategies to enhance genome editing
Journal of Biomedical Science (2023)