Abstract
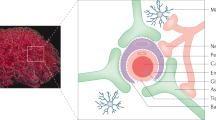
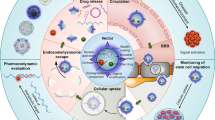
Oligonucleotide therapies are currently experiencing a resurgence driven by advances in backbone chemistry and discoveries of novel therapeutic pathways that can be uniquely and efficiently modulated by the oligonucleotide drugs. A quarter of a century has passed since oligonucleotides were first applied in living mammalian brain to modulate gene expression. Despite challenges in delivery to the brain, multiple oligonucleotide-based compounds are now being developed for treatment of human brain disorders by direct delivery inside the blood brain barrier (BBB). Notably, the first new central nervous system (CNS)-targeted oligonucleotide-based drug (nusinersen/Spinraza) was approved by US Food and Drug Administration (FDA) in late 2016 and several other compounds are in advanced clinical trials. Human testing of brain-targeted oligonucleotides has highlighted unusual pharmacokinetic and pharmacodynamic properties of these compounds, including complex active uptake mechanisms, low systemic exposure, extremely long half-lives, accumulation and gradual release from subcellular depots. Further work on oligonucleotide uptake, development of formulations for delivery across the BBB and relevant disease biology studies are required for further optimization of the oligonucleotide drug development process for brain applications.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Banks, W.A. et al. Delivery across the blood-brain barrier of antisense directed against amyloid beta: reversal of learning and memory deficits in mice overexpressing amyloid precursor protein. J. Pharmacol. Exp. Ther. 297, 1113–1121 (2001).
Farr, S.A., Erickson, M.A., Niehoff, M.L., Banks, W.A. & Morley, J.E. Central and peripheral administration of antisense oligonucleotide targeting amyloid-β protein precursor improves learning and memory and reduces neuroinflammatory cytokines in Tg2576 (AβPPswe) mice. J. Alzheimer's Dis. 40, 1005–1016 (2014).
Juliano, R.L. The delivery of therapeutic oligonucleotides. Nucleic Acids Res. 44, 6518–6548 (2016).
Eckstein, F. Nucleoside phosphorothioates. J. Am. Chem. Soc. 92, 4718–4723 (1970).
Stirchak, E.P., Summerton, J.E. & Weller, D.D. Uncharged stereoregular nucleic acid analogs: 2. Morpholino nucleoside oligomers with carbamate internucleoside linkages. Nucleic Acids Res. 17, 6129–6141 (1989).
Kupryushkin, M.S., Pyshnyi, D.V. & Stetsenko, D.A. Phosphoryl guanidines: a new type of nucleic acid analogues. Acta Naturae 6, 116–118 (2014).
Nielsen, P.E., Egholm, M., Berg, R.H. & Buchardt, O. Sequence-selective recognition of DNA by strand displacement with a thymine-substituted polyamide. Science 254, 1497–1500 (1991).
Bobst, A.M., Rottman, F. & Cerutti, P.A. Effect of the methylation of the 2′-hydroxyl groups in polyadenylic acid on its structure in weakly acidic and neutral solutions and on its capability to form ordered complexes with polyuridylic acid. J. Mol. Biol. 46, 221–234 (1969).
Evers, M.M. & Toonen, L.J. van Roon-Mom, W.M. Antisense oligonucleotides in therapy for neurodegenerative disorders. Adv. Drug Deliv. Rev. 87, 90–103 (2015).
Cook, P.D. & Guinosso, C.J. 2′-O-modified nucleosides and phosphoramidites. US patent no. 5,914,396 (1995).
Wahlestedt, C. et al. Potent and nontoxic antisense oligonucleotides containing locked nucleic acids. Proc. Natl. Acad. Sci. USA 97, 5633–5638 (2000).
Goyenvalle, A. et al. Functional correction in mouse models of muscular dystrophy using exon-skipping tricyclo-DNA oligomers. Nat. Med. 21, 270–275 (2015).
Hua, Y. et al. Peripheral SMN restoration is essential for long-term rescue of a severe spinal muscular atrophy mouse model. Nature 478, 123–126 (2011).
Passini, M.A. et al. Antisense oligonucleotides delivered to the mouse CNS ameliorate symptoms of severe spinal muscular atrophy. Sci. Transl. Med. 3, 72ra18 (2011).
Williams, J.H. et al. Oligonucleotide-mediated survival of motor neuron protein expression in CNS improves phenotype in a mouse model of spinal muscular atrophy. J. Neurosci. 29, 7633–7638 (2009).
Porensky, P.N. et al. A single administration of morpholino antisense oligomer rescues spinal muscular atrophy in mouse. Hum. Mol. Genet. 21, 1625–1638 (2012).
Zhou, H. et al. A novel morpholino oligomer targeting ISS-N1 improves rescue of severe spinal muscular atrophy transgenic mice. Hum. Gene Ther. 24, 331–342 (2013).
Ho, W., Zhang, X.Q. & Xu, X. Biomaterials in siRNA Delivery: A Comprehensive Review. Adv. Healthc. Mater. 5, 2715–2731 (2016).
Ozcan, G., Ozpolat, B., Coleman, R.L., Sood, A.K. & Lopez-Berestein, G. Preclinical and clinical development of siRNA-based therapeutics. Adv. Drug Deliv. Rev. 87, 108–119 (2015).
Qiu, Y., Lam, J.K., Leung, S.W. & Liang, W. Delivery of RNAi therapeutics to the airways-from bench to bedside. Molecules 21, E1249 (2016).
Kim, H.J., Kim, A., Miyata, K. & Kataoka, K. Recent progress in development of siRNA delivery vehicles for cancer therapy. Adv. Drug Deliv. Rev. 104, 61–77 (2016).
Lima, W.F., De Hoyos, C.L., Liang, X.H. & Crooke, S.T. RNA cleavage products generated by antisense oligonucleotides and siRNAs are processed by the RNA surveillance machinery. Nucleic Acids Res. 44, 3351–3363 (2016).
Miller, V.M. et al. Allele-specific silencing of dominant disease genes. Proc. Natl. Acad. Sci. USA 100, 7195–7200 (2003).
Ward, A.J., Norrbom, M., Chun, S., Bennett, C.F. & Rigo, F. Nonsense-mediated decay as a terminating mechanism for antisense oligonucleotides. Nucleic Acids Res. 42, 5871–5879 (2014).
Haas, M. et al. European Medicines Agency review of ataluren for the treatment of ambulant patients aged 5 years and older with Duchenne muscular dystrophy resulting from a nonsense mutation in the dystrophin gene. Neuromuscul. Disord. 25, 5–13 (2015).
Kerem, E. et al. Ataluren for the treatment of nonsense-mutation cystic fibrosis: a randomised, double-blind, placebo-controlled phase 3 trial. Lancet Respir. Med. 2, 539–547 (2014).
Shimizu-Motohashi, Y., Miyatake, S., Komaki, H., Takeda, S. & Aoki, Y. Recent advances in innovative therapeutic approaches for Duchenne muscular dystrophy: from discovery to clinical trials. Am. J. Transl. Res. 8, 2471–2489 (2016).
Arechavala-Gomeza, V., Khoo, B. & Aartsma-Rus, A. Splicing modulation therapy in the treatment of genetic diseases. Appl. Clin. Genet. 7, 245–252 (2014).
Pereira, P., Queiroz, J.A., Figueiras, A. & Sousa, F. Current progress on microRNAs-based therapeutics in neurodegenerative diseases. Wiley Interdiscip. Rev. RNA http://dx.doi.org/10.1002/wrna.1409 (2016).
Catalanotto, C., Cogoni, C. & Zardo, G. MicroRNA in control of gene expression: an overview of nuclear functions. Int. J. Mol. Sci. 17, E1712 (2016).
Cheng, C.J. et al. MicroRNA silencing for cancer therapy targeted to the tumour microenvironment. Nature 518, 107–110 (2015).
Wahlestedt, C. Targeting long non-coding RNA to therapeutically upregulate gene expression. Nat. Rev. Drug Discov. 12, 433–446 (2013).
Nakagawa, S. & Kageyama, Y. Nuclear lncRNAs as epigenetic regulators-beyond skepticism. Biochim. Biophys. Acta 1839, 215–222 (2014).
Magistri, M., Faghihi, M.A., St Laurent, G., III & Wahlestedt, C. Regulation of chromatin structure by long noncoding RNAs: focus on natural antisense transcripts. Trends Genet. 28, 389–396 (2012).
Katayama, S. et al. Antisense transcription in the mammalian transcriptome. Science 309, 1564–1566 (2005).
Modarresi, F. et al. Inhibition of natural antisense transcripts in vivo results in gene-specific transcriptional upregulation. Nat. Biotechnol. 30, 453–459 (2012).
Halley, P. et al. Regulation of the apolipoprotein gene cluster by a long noncoding RNA. Cell Rep. 6, 222–230 (2014).
Hsiao, J. et al. Upregulation of haploinsufficient gene expression in the brain by targeting a long non-coding RNA improves seizure phenotype in a model of Dravet syndrome. EBioMedicine 9, 257–277 (2016).
Zhao, J. et al. Genome-wide identification of polycomb-associated RNAs by RIP-seq. Mol. Cell 40, 939–953 (2010).
Janowski, B.A. et al. Activating gene expression in mammalian cells with promoter-targeted duplex RNAs. Nat. Chem. Biol. 3, 166–173 (2007).
Chu, Y., Yue, X., Younger, S.T., Janowski, B.A. & Corey, D.R. Involvement of argonaute proteins in gene silencing and activation by RNAs complementary to a non-coding transcript at the progesterone receptor promoter. Nucleic Acids Res. 38, 7736–7748 (2010).
Portnoy, V. et al. saRNA-guided Ago2 targets the RITA complex to promoters to stimulate transcription. Cell Res. 26, 320–335 (2016).
Wang, B. et al. Small-activating RNA can change nucleosome positioning in human fibroblasts. J. Biomol. Screen. 21, 634–642 (2016).
Hu, J. et al. Promoter-associated small double-stranded RNA interacts with heterogeneous nuclear ribonucleoprotein A2/B1 to induce transcriptional activation. Biochem. J. 447, 407–416 (2012).
Kleinman, M.E. et al. Sequence- and target-independent angiogenesis suppression by siRNA via TLR3. Nature 452, 591–597 (2008).
Koller, E. et al. Mechanisms of single-stranded phosphorothioate modified antisense oligonucleotide accumulation in hepatocytes. Nucleic Acids Res. 39, 4795–4807 (2011).
Juliano, R.L., Ming, X. & Nakagawa, O. Cellular uptake and intracellular trafficking of antisense and siRNA oligonucleotides. Bioconjug. Chem. 23, 147–157 (2012).
Wagenaar, T.R. et al. Identification of the endosomal sorting complex required for transport-I (ESCRT-I) as an important modulator of anti-miR uptake by cancer cells. Nucleic Acids Res. 43, 1204–1215 (2015).
Shen, W., Liang, X.H. & Crooke, S.T. Phosphorothioate oligonucleotides can displace NEAT1 RNA and form nuclear paraspeckle-like structures. Nucleic Acids Res. 42, 8648–8662 (2014).
Naganuma, T. & Hirose, T. Paraspeckle formation during the biogenesis of long non-coding RNAs. RNA Biol. 10, 456–461 (2013).
Liang, X.H. et al. Hsp90 protein interacts with phosphorothioate oligonucleotides containing hydrophobic 2′-modifications and enhances antisense activity. Nucleic Acids Res. 44, 3892–3907 (2016).
Shemesh, C.S. et al. Pharmacokinetic and pharmacodynamic investigations of ION-353382, a model antisense oligonucleotide: using alpha-2-macroglobulin and murinoglobulin double-knockout mice. Nucleic Acid Ther. 26, 223–235 (2016).
Zhu, J.Y. et al. Efficient nuclear drug translocation and improved drug efficacy mediated by acidity-responsive boronate-linked dextran/cholesterol nanoassembly. Biomaterials 52, 281–290 (2015).
Liang, X.H., Shen, W., Sun, H., Prakash, T.P. & Crooke, S.T. TCP1 complex proteins interact with phosphorothioate oligonucleotides and can co-localize in oligonucleotide-induced nuclear bodies in mammalian cells. Nucleic Acids Res. 42, 7819–7832 (2014).
Miller, C.M. et al. Stabilin-1 and Stabilin-2 are specific receptors for the cellular internalization of phosphorothioate-modified antisense oligonucleotides (ASOs) in the liver. Nucleic Acids Res. 44, 2782–2794 (2016).
Ezzat, K. et al. Self-assembly into nanoparticles is essential for receptor mediated uptake of therapeutic antisense oligonucleotides. Nano Lett. 15, 4364–4373 (2015).
Ezzat, K. et al. Scavenger receptor-mediated uptake of cell-penetrating peptide nanocomplexes with oligonucleotides. FASEB J. 26, 1172–1180 (2012).
Lorenz, P., Baker, B.F., Bennett, C.F. & Spector, D.L. Phosphorothioate antisense oligonucleotides induce the formation of nuclear bodies. Mol. Biol. Cell 9, 1007–1023 (1998).
Marcusson, E.G., Bhat, B., Manoharan, M., Bennett, C.F. & Dean, N.M. Phosphorothioate oligodeoxyribonucleotides dissociate from cationic lipids before entering the nucleus. Nucleic Acids Res. 26, 2016–2023 (1998).
Heemskerk, H. et al. Preclinical PK and PD studies on 2′-O-methyl-phosphorothioate RNA antisense oligonucleotides in the mdx mouse model. Mol. Ther. 18, 1210–1217 (2010).
Hammond, S.M. et al. Systemic peptide-mediated oligonucleotide therapy improves long-term survival in spinal muscular atrophy. Proc. Natl. Acad. Sci. USA 113, 10962–10967 (2016).
Craft, S. et al. Intranasal insulin therapy for Alzheimer disease and amnestic mild cognitive impairment: a pilot clinical trial. Arch. Neurol. 69, 29–38 (2012).
Hashizume, R. et al. New therapeutic approach for brain tumors: Intranasal delivery of telomerase inhibitor GRN163. Neuro-oncol. 10, 112–120 (2008).
Ma, Q. et al. Inhibition of microRNA-210 provides neuroprotection in hypoxic-ischemic brain injury in neonatal rats. Neurobiol. Dis. 89, 202–212 (2016).
Ferrés-Coy, A. et al. Therapeutic antidepressant potential of a conjugated siRNA silencing the serotonin transporter after intranasal administration. Mol. Psychiatry 21, 328–338 (2016).
Bleier, B.S., Kohman, R.E., Feldman, R.E., Ramanlal, S. & Han, X. Permeabilization of the blood-brain barrier via mucosal engrafting: implications for drug delivery to the brain. PLoS One 8, e61694 (2013).
Wahlestedt, C., Yee, F., Yoo, H., Koob, G.F. & Heilig, M. in Molecular Neurobiology, Proceedings of the second NIMH Conference (eds. Zalcman, S., Scheller, R. & Tsien, R.) (1992).
Wahlestedt, C. et al. Antisense oligodeoxynucleotides to NMDA-R1 receptor channel protect cortical neurons from excitotoxicity and reduce focal ischaemic infarctions. Nature 363, 260–263 (1993).
Wahlestedt, C., Pich, E.M., Koob, G.F., Yee, F. & Heilig, M. Modulation of anxiety and neuropeptide Y-Y1 receptors by antisense oligodeoxynucleotides. Science 259, 528–531 (1993).
Wahlestedt, C. Antisense oligonucleotide strategies in neuropharmacology. Trends Pharmacol. Sci. 15, 42–46 (1994).
Standifer, K.M., Chien, C.C., Wahlestedt, C., Brown, G.P. & Pasternak, G.W. Selective loss of delta opioid analgesia and binding by antisense oligodeoxynucleotides to a delta opioid receptor. Neuron 12, 805–810 (1994).
Geary, R.S., Norris, D., Yu, R. & Bennett, C.F. Pharmacokinetics, biodistribution and cell uptake of antisense oligonucleotides. Adv. Drug Deliv. Rev. 87, 46–51 (2015).
Rigo, F. et al. Pharmacology of a central nervous system delivered 2′-O-methoxyethyl-modified survival of motor neuron splicing oligonucleotide in mice and nonhuman primates. J. Pharmacol. Exp. Ther. 350, 46–55 (2014).
Verhaart, I.E. et al. Dose-dependent pharmacokinetic profiles of 2′-O-methyl phosphorothioate antisense oligonucleotidesin mdx mice. Nucleic Acid Ther. 23, 228–237 (2013).
Kordasiewicz, H.B. et al. Sustained therapeutic reversal of Huntington's disease by transient repression of huntingtin synthesis. Neuron 74, 1031–1044 (2012).
Miller, T.M. et al. An antisense oligonucleotide against SOD1 delivered intrathecally for patients with SOD1 familial amyotrophic lateral sclerosis: a phase 1, randomised, first-in-man study. Lancet Neurol. 12, 435–442 (2013).
Shen, L. et al. Effects of repeated complement activation associated with chronic treatment of cynomolgus monkeys with 2′-o-methoxyethyl modified antisense oligonucleotide. Nucleic Acid Ther. 26, 236–249 (2016).
Shen, L. et al. Mechanistic understanding for the greater sensitivity of monkeys to antisense oligonucleotide-mediated complement activation compared with humans. J. Pharmacol. Exp. Ther. 351, 709–717 (2014).
Henry, S.P. et al. Considerations for the characterization and interpretation of results related to alternative complement activation in monkeys associated with oligonucleotide-based therapeutics. Nucleic Acid Ther. 26, 210–215 (2016).
Voit, T. et al. Safety and efficacy of drisapersen for the treatment of Duchenne muscular dystrophy (DEMAND II): an exploratory, randomised, placebo-controlled phase 2 study. Lancet Neurol. 13, 987–996 (2014).
Engelhardt, J.A. Comparative renal toxicopathology of antisense oligonucleotides. Nucleic Acid Ther. 26, 199–209 (2016).
Merlini, L. & Sabatelli, P. Improving clinical trial design for Duchenne muscular dystrophy. BMC Neurol. 15, 153 (2015).
Rigo, F., Hua, Y., Krainer, A.R. & Bennett, C.F. Antisense-based therapy for the treatment of spinal muscular atrophy. J. Cell Biol. 199, 21–25 (2012).
Chiriboga, C.A. et al. Results from a phase 1 study of nusinersen (ISIS-SMN(Rx)) in children with spinal muscular atrophy. Neurology 86, 890–897 (2016).
Haché, M. et al. Intrathecal injections in children with spinal muscular atrophy: nusinersen clinical trial experience. J. Child Neurol. 31, 899–906 (2016).
Hua, Y. et al. Motor neuron cell-nonautonomous rescue of spinal muscular atrophy phenotypes in mild and severe transgenic mouse models. Genes Dev. 29, 288–297 (2015).
Sintusek, P. et al. Histopathological defects in intestine in severe spinal muscular atrophy mice are improved by systemic antisense oligonucleotide treatment. PLoS One 11, e0155032 (2016).
Bogdanik, L.P. et al. Systemic, postsymptomatic antisense oligonucleotide rescues motor unit maturation delay in a new mouse model for type II/III spinal muscular atrophy. Proc. Natl. Acad. Sci. USA 112, E5863–E5872 (2015).
Osman, E.Y. et al. Morpholino antisense oligonucleotides targeting intronic repressor Element1 improve phenotype in SMA mouse models. Hum. Mol. Genet. 23, 4832–4845 (2014).
Evers, M.M. et al. Ataxin-3 protein modification as a treatment strategy for spinocerebellar ataxia type 3: removal of the CAG containing exon. Neurobiol. Dis. 58, 49–56 (2013).
Cavalieri, S., Pozzi, E., Gatti, R.A. & Brusco, A. Deep-intronic ATM mutation detected by genomic resequencing and corrected in vitro by antisense morpholino oligonucleotide (AMO). Eur. J. Hum. Genet. 21, 774–778 (2013).
Donnelly, C.J. et al. RNA toxicity from the ALS/FTD C9ORF72 expansion is mitigated by antisense intervention. Neuron 80, 415–428 (2013).
Lagier-Tourenne, C. et al. Targeted degradation of sense and antisense C9orf72 RNA foci as therapy for ALS and frontotemporal degeneration. Proc. Natl. Acad. Sci. USA 110, E4530–E4539 (2013).
Sareen, D. et al. Targeting RNA foci in iPSC-derived motor neurons from ALS patients with a C9ORF72 repeat expansion. Sci. Transl. Med. 5, 208ra149 (2013).
Jiang, J. et al. Gain of toxicity from ALS/FTD-linked repeat expansions in C9ORF72 is alleviated by antisense oligonucleotides targeting GGGGCC-containing RNAs. Neuron 90, 535–550 (2016).
Gotkine, M. et al. Presymptomatic treatment with acetylcholinesterase antisense oligonucleotides prolongs survival in ALS (G93A-SOD1) mice. BioMed Res. Int. 2013, 845345 (2013).
Koval, E.D. et al. Method for widespread microRNA-155 inhibition prolongs survival in ALS-model mice. Hum. Mol. Genet. 22, 4127–4135 (2013).
Southwell, A.L. et al. In vivo evaluation of candidate allele-specific mutant huntingtin gene silencing antisense oligonucleotides. Mol. Ther. 22, 2093–2106 (2014).
Benson, M.D. et al. Suppression of choroid plexus transthyretin levels by antisense oligonucleotide treatment. Amyloid 17, 43–49 (2010).
Sztainberg, Y. et al. Reversal of phenotypes in MECP2 duplication mice using genetic rescue or antisense oligonucleotides. Nature 528, 123–126 (2015).
Meng, L. et al. Towards a therapy for Angelman syndrome by targeting a long non-coding RNA. Nature 518, 409–412 (2015).
Hinrich, A.J. et al. Therapeutic correction of ApoER2 splicing in Alzheimer's disease mice using antisense oligonucleotides. EMBO Mol. Med. 8, 328–345 (2016).
Kim, J. et al. microRNA-33 regulates ApoE lipidation and amyloid-β metabolism in the brain. J. Neurosci. 35, 14717–14726 (2015).
Jan, A. et al. Direct intracerebral delivery of a miR-33 antisense oligonucleotide into mouse brain increases brain ABCA1 expression. [Corrected]. Neurosci. Lett. 598, 66–72 (2015).
Farr, S.A. et al. Antisense oligonucleotide against GSK-3β in brain of SAMP8 mice improves learning and memory and decreases oxidative stress: Involvement of transcription factor Nrf2 and implications for Alzheimer disease. Free Radic. Biol. Med. 67, 387–395 (2014).
DeVos, S.L. et al. Tau reduction prevents neuronal loss and reverses pathological tau deposition and seeding in mice with tauopathy. Sci. Transl. Med. 9, eaag0481 (2017).
Obika, S. et al. Synthesis of 2′-O,4′-C-methyleneuridine and -cytidine. Novel bicyclic nucleosides having a fixed C3′-endo sugar puckering. Tetrahedr. Lett. 38, 8735–8738 (1997).
Steffens, R. & Leumann, C.J. Synthesis and thermodynamic and biophysical properties of tricyclo-DNA. J. Am. Chem. Soc. 121, 3249–3255 (1999).
Zamecnik, P.C. & Stephenson, M.L. Inhibition of Rous sarcoma virus replication and cell transformation by a specific oligodeoxynucleotide. Proc. Natl. Acad. Sci. USA 75, 280–284 (1978).
Stephenson, M.L. & Zamecnik, P.C. Inhibition of Rous sarcoma viral RNA translation by a specific oligodeoxyribonucleotide. Proc. Natl. Acad. Sci. USA 75, 285–288 (1978).
Donis-Keller, H. Site specific enzymatic cleavage of RNA. Nucleic Acids Res. 7, 179–192 (1979).
Guerrier-Takada, C., Gardiner, K., Marsh, T., Pace, N. & Altman, S. The RNA moiety of ribonuclease P is the catalytic subunit of the enzyme. Cell 35, 849–857 (1983).
Bass, B.L. & Cech, T.R. Specific interaction between the self-splicing RNA of Tetrahymena and its guanosine substrate: implications for biological catalysis by RNA. Nature 308, 820–826 (1984).
Elbashir, S.M. et al. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature 411, 494–498 (2001).
Wlotzka, B. et al. In vivo properties of an anti-GnRH Spiegelmer: an example of an oligonucleotide-based therapeutic substance class. Proc. Natl. Acad. Sci. USA 99, 8898–8902 (2002).
Azad, R.F., Driver, V.B., Tanaka, K., Crooke, R.M. & Anderson, K.P. Antiviral activity of a phosphorothioate oligonucleotide complementary to RNA of the human cytomegalovirus major immediate-early region. Antimicrob. Agents Chemother. 37, 1945–1954 (1993).
Kumar, R. et al. The first analogues of LNA (locked nucleic acids): phosphorothioate-LNA and 2′-thio-LNA. Bioorg. Med. Chem. Lett. 8, 2219–2222 (1998).
DeVos, S.L. et al. Tau reduction prevents neuronal loss and reverses pathological tau deposition and seeding in mice with tauopathy. Sci. Transl. Med. 9, 374 (2017).
Acknowledgements
RNA-related work in C.W.'s laboratory is currently funded by NIH grants DA035592, NS071674 and AA023781.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
O. Khorkova is employed by OPKO Health.
Rights and permissions
About this article
Cite this article
Khorkova, O., Wahlestedt, C. Oligonucleotide therapies for disorders of the nervous system. Nat Biotechnol 35, 249–263 (2017). https://doi.org/10.1038/nbt.3784
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.3784
This article is cited by
-
Mapping PTBP2 binding in human brain identifies SYNGAP1 as a target for therapeutic splice switching
Nature Communications (2023)
-
Amplifying gene expression with RNA-targeted therapeutics
Nature Reviews Drug Discovery (2023)
-
Rescue of neuropsychiatric phenotypes in a mouse model of 16p11.2 duplication syndrome by genetic correction of an epilepsy network hub
Nature Communications (2023)
-
The dysregulation of miRNAs in epilepsy and their regulatory role in inflammation and apoptosis
Functional & Integrative Genomics (2023)
-
Nanodelivery of nucleic acids
Nature Reviews Methods Primers (2022)