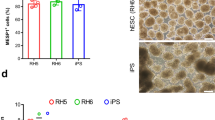
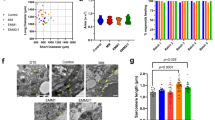
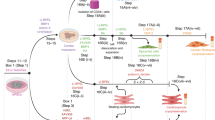
Abstract
The inability of multipotent cardiovascular progenitor cells (CPCs) to undergo multiple divisions in culture has precluded stable expansion of precursors of cardiomyocytes and vascular cells. This contrasts with neural progenitors, which can be expanded robustly and are a renewable source of their derivatives. Here we use human pluripotent stem cells bearing a cardiac lineage reporter to show that regulated MYC expression enables robust expansion of CPCs with insulin-like growth factor-1 (IGF-1) and a hedgehog pathway agonist. The CPCs can be patterned with morphogens, recreating features of heart field assignment, and controllably differentiated to relatively pure populations of pacemaker-like or ventricular-like cardiomyocytes. The cells are clonogenic and can be expanded for >40 population doublings while retaining the ability to differentiate into cardiomyocytes and vascular cells. Access to CPCs will allow precise recreation of elements of heart development in vitro and facilitate investigation of the molecular basis of cardiac fate determination. This technology is applicable for cardiac disease modeling, toxicology studies and tissue engineering.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Drawnel, F.M. et al. Disease modeling and phenotypic drug screening for diabetic cardiomyopathy using human induced pluripotent stem cells. Cell Reports 9, 810–820 (2014).
Matsa, E., Burridge, P.W. & Wu, J.C. Human stem cells for modeling heart disease and for drug discovery. Sci. Transl. Med. 6, 239ps6 (2014).
Lundy, S.D., Gantz, J.A., Pagan, C.M., Filice, D. & Laflamme, M.A. Pluripotent stem cell derived cardiomyocytes for cardiac repair. Curr. Treat. Options Cardiovasc. Med. 16, 319 (2014).
Bhave, M., Akhter, N. & Rosen, S.T. Cardiovascular toxicity of biologic agents for cancer therapy. Oncology 28, 482–490 (2014).
Birket, M.J. & Mummery, C.L. Pluripotent stem cell derived cardiovascular progenitors—a developmental perspective. Dev. Biol. 400, 169–179 (2015).
Vincent, S.D. & Buckingham, M.E. How to make a heart: the origin and regulation of cardiac progenitor cells. Curr. Top. Dev. Biol. 90, 1–41 (2010).
Cai, C.-L. et al. Isl1 identifies a cardiac progenitor population that proliferates prior to differentiation and contributes a majority of cells to the heart. Dev. Cell 5, 877–889 (2003).
Lints, T.J., Parsons, L.M., Hartley, L., Lyons, I. & Harvey, R.P. Nkx-2.5: a novel murine homeobox gene expressed in early heart progenitor cells and their myogenic descendants. Development 119, 419–431 (1993).
Ma, Q., Zhou, B. & Pu, W.T. Reassessment of Isl1 and Nkx2–5 cardiac fate maps using a Gata4-based reporter of Cre activity. Dev. Biol. 323, 98–104 (2008).
Laugwitz, K.-L. et al. Postnatal isl1+ cardioblasts enter fully differentiated cardiomyocyte lineages. Nature 433, 647–653 (2005).
Moretti, A. et al. Multipotent embryonic isl1+ progenitor cells lead to cardiac, smooth muscle, and endothelial cell diversification. Cell 127, 1151–1165 (2006).
Wu, S.M. et al. Developmental origin of a bipotential myocardial and smooth muscle cell precursor in the mammalian heart. Cell 127, 1137–1150 (2006).
Mommersteeg, M.T.M. et al. Molecular pathway for the localized formation of the sinoatrial node. Circ. Res. 100, 354–362 (2007).
Wiese, C. et al. Circ. Res. 104, 388–397 (2009).
Liang, X. et al. HCN4 dynamically marks the first heart field and conduction system precursors. Circ. Res. 113, 399–407 (2013).
Später, D. et al. A HCN4+ cardiomyogenic progenitor derived from the first heart field and human pluripotent stem cells. Nat. Cell Biol. 15, 1098–1106 (2013).
Kattman, S.J. et al. Stage-specific optimization of activin/nodal and BMP signaling promotes cardiac differentiation of mouse and human pluripotent stem cell lines. Cell Stem Cell 8, 228–240 (2011).
Wierstra, I. & Alves, J. The c-myc promoter: still mysterY and challenge. Adv. Cancer Res. 99, 113–333 (2008).
Gittenberger-De Groot, A.C. et al. Nkx2.5 negative myocardium of the posterior heart field and its correlation with podoplanin expression in cells from the developing cardiac pacemaking and conduction system. Anat. Rec. 290, 115–122 (2007).
Dyer, L.A. et al. BMP signaling modulates hedgehog-induced secondary heart field proliferation. Dev. Biol. 348, 167–176 (2010).
Gude, N. et al. Akt promotes increased cardiomyocyte cycling and expansion of the cardiac progenitor cell population. Circ. Res. 99, 381–388 (2006).
Keren-Politansky, A., Keren, A. & Bengal, E. Neural ectoderm-secreted FGF initiates the expression of Nkx2.5 in cardiac progenitors via a p38 MAPK/CREB pathway. Dev. Biol. 335, 374–384 (2009).
Liberatore, C.M., Searcy-Schrick, R.D., Vincent, E.B. & Yutzey, K.E. Nkx-2.5 gene induction in mice is mediated by a Smad consensus regulatory region. Dev. Biol. 244, 243–256 (2002).
Reifers, F., Walsh, E.C., Leger, S., Stainier, D.Y. & Brand, M. Induction and differentiation of the zebrafish heart requires fibroblast growth factor 8 (fgf8/acerebellar). Development 127, 225–235 (2000).
Schultheiss, T.M., Burch, J.B. & Lassar, A.B. A role for bone morphogenetic proteins in the induction of cardiac myogenesis. Genes Dev. 11, 451–462 (1997).
Barron, M., Gao, M. & Lough, J. Requirement for BMP and FGF signaling during cardiogenic induction in non-precardiac mesoderm is specific, transient, and cooperative. Dev. Dyn. 218, 383–393 (2000).
Inman, G.J. et al. SB-431542 is a potent and specific inhibitor of transforming growth factor-β superfamily type I activin receptor-like kinase (ALK) receptors ALK4, ALK5, and ALK7. Mol. Pharmacol. 62, 65–74 (2002).
Den Hartogh, S.C. et al. Dual reporter MESP1mCherry/w-NKX2–5eGFP/w hESCs enable studying early human cardiac differentiation. Stem Cells 33, 56–67 (2015).
Yang, L. et al. Human cardiovascular progenitor cells develop from a KDR+ embryonic-stem-cell-derived population. Nature 453, 524–528 (2008).
Ardehali, R. et al. Prospective isolation of human embryonic stem cell-derived cardiovascular progenitors that integrate into human fetal heart tissue. Proc. Natl. Acad. Sci. USA 110, 3405–3410 (2013).
van Berlo, J.H. et al. c-kit+ cells minimally contribute cardiomyocytes to the heart. Nature 509, 337–341 (2014).
James, D. et al. Expansion and maintenance of human embryonic stem cell-derived endothelial cells by TGFβ inhibition is Id1 dependent. Nat. Biotechnol. 28, 161–166 (2010).
Marques, S.R., Lee, Y., Poss, K.D. & Yelon, D. Reiterative roles for FGF signaling in the establishment of size and proportion of the zebrafish heart. Dev. Biol. 321, 397–406 (2008).
de Pater, E. et al. Bmp signaling exerts opposite effects on cardiac differentiation. Circ. Res. 110, 578–587 (2012).
Ueno, S. et al. Biphasic role for Wnt/β-catenin signaling in cardiac specification in zebrafish and embryonic stem cells. Proc. Natl. Acad. Sci. USA 104, 9685–9690 (2007).
Goumans, M.-J. et al. TGF-β1 induces efficient differentiation of human cardiomyocyte progenitor cells into functional cardiomyocytes in vitro. Stem Cell Res. 1, 138–149 (2007).
Verkerk, A.O., van Ginneken, A.C.G. & Wilders, R. Pacemaker activity of the human sinoatrial node: role of the hyperpolarization-activated current, I(f). Int. J. Cardiol. 132, 318–336 (2009).
Xi, J. et al. Comparison of contractile behavior of native murine ventricular tissue and cardiomyocytes derived from embryonic or induced pluripotent stem cells. FASEB J. 24, 2739–2751 (2010).
Jackson, T. et al. The c-myc proto-oncogene regulates cardiac development in transgenic mice. Mol. Cell. Biol. 10, 3709–3716 (1990).
Noseda, M., Peterkin, T., Simões, F.C., Patient, R. & Schneider, M.D. Cardiopoietic factors extracellular signals for cardiac lineage commitment. Circ. Res. 108, 129–152 (2011).
Cai, W. et al. Coordinate Nodal and BMP inhibition directs Baf60c-dependent cardiomyocyte commitment. Genes Dev. 27, 2332–2344 (2013).
Waldo, K.L. et al. Conotruncal myocardium arises from a secondary heart field. Development 128, 3179–3188 (2001).
Milgrom-Hoffman, M. et al. The heart endocardium is derived from vascular endothelial progenitors. Development 138, 4777–4787 (2011).
Zhang, Q. et al. Direct differentiation of atrial and ventricular myocytes from human embryonic stem cells by alternating retinoid signals. Cell Res. 21, 579–587 (2011).
Devalla, H.D. et al. Atrial-like cardiomyocytes from human pluripotent stem cells are a robust preclinical model for assessing atrial-selective pharmacology. EMBO Mol. Med. 7, 394–410 (2015).
Kattman, S.J., Huber, T.L. & Keller, G.M. Multipotent Flk-1+ cardiovascular progenitor cells give rise to the cardiomyocyte, endothelial, and vascular smooth muscle lineages. Dev. Cell 11, 723–732 (2006).
Qyang, Y. et al. The renewal and differentiation of Isl1+ cardiovascular progenitors are controlled by a WNT/β-catenin pathway. Cell Stem Cell 1, 165–179 (2007).
Bu, L. et al. Human ISL1 heart progenitors generate diverse multipotent cardiovascular cell lineages. Nature 460, 113–117 (2009).
Domian, I.J. et al. Generation of functional ventricular heart muscle from mouse ventricular progenitor cells. Science 326, 426–429 (2009).
Moretti, A. et al. Mouse and human induced pluripotent stem cells as a source for multipotent Isl1+ cardiovascular progenitors. FASEB J. 24, 700–711 (2010).
Elliott, D.A. et al. NKX2–5eGFP/w hESCs for isolation of human cardiac progenitors and cardiomyocytes. Nat. Methods 8, 1037–1040 (2011).
Miano, J.M., Cserjesi, P., Ligon, K.L., Periasamy, M. & Olson, E.N. Smooth muscle myosin heavy chain exclusively marks the smooth muscle lineage during mouse embryogenesis. Circ. Res. 75, 803–812 (1994).
Bellin, M. et al. Isogenic human pluripotent stem cell pairs reveal the role of a KCNH2 mutation in long-QT syndrome. EMBO J. 32, 3161–3175 (2013).
Maherali, N. et al. A high-efficiency system for the generation and study of human induced pluripotent stem cells. Cell Stem Cell 3, 340–345 (2008).
Birket, M.J. et al. PGC-1-α and reactive oxygen species regulate human embryonic stem cell-derived cardiomyocyte function. Stem Cell Rep. 1, 560–574 (2013).
Hao, J. et al. In vivo structure activity relationship study of dorsomorphin analogs identifies selective VEGF and BMP inhibitors. ACS Chem. Biol. 5, 245–253 (2010).
Rape, A.D., Guo, W.-H. & Wang, Y.-L. The regulation of traction force in relation to cell shape and focal adhesions. Biomaterials 32, 2043–2051 (2011).
Acknowledgements
We acknowledge the contributions of I. Marcuccio and S. Casini. Work in C.L.M.'s lab is supported by Cardiovascular Research Netherlands (CVON HUSTCARE), The Netherlands Institute of Regenerative Medicine (NIRM), the European Research Council (ERCAdG 323182 STEMCARDIOVASC) and The Netherlands Organization for Scientific Research (NWO-FOM FOM 09MMC02). Work in R.P.'s lab is supported by the Netherlands Organization for Scientific Research (ZonMW-TOP 40-00812-98-12086 and ZonMW-MKMD 40-42600-98-036) and the Rembrandt Institute of Cardiovascular Science. V.V.O. was supported by the European Community's Seventh Framework Programme (FP7/2007-2013) grant agreement 602423.
Author information
Authors and Affiliations
Contributions
M.J.B. and C.L.M. designed the study. M.J.B., M.C.R., A.O.V., A.R.L. and V.S. performed experiments and analyzed data. H.D.D. analyzed data. M.J.B., C.L.M. and A.O.V. wrote the manuscript. D.W. maintained and differentiated the PSCs. S.C.d.H. generated and provided the MESP1+ RNA. M.B. generated and provided the original hPSC lines. V.V.O. helped with experimental design. R.P. and C.L.M. supervised the study. All authors edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–14 and Supplementary Tables 1 and 2 (PDF 14182 kb)
Data Set 1
Microarray Gene Lists (XLSX 52 kb)
Source Data 1
Source Data Figure 1 (XLSX 14 kb)
Source Data 2
Source Data Figure 2 (XLSX 10 kb)
Source Data 3
Source Data Figure 3 (XLSX 14 kb)
Source Data 4
Source Data Figure 4 (XLSX 19 kb)
Source Data 5
Source Data Figure 5 (XLSX 13 kb)
Rights and permissions
About this article
Cite this article
Birket, M., Ribeiro, M., Verkerk, A. et al. Expansion and patterning of cardiovascular progenitors derived from human pluripotent stem cells. Nat Biotechnol 33, 970–979 (2015). https://doi.org/10.1038/nbt.3271
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.3271
This article is cited by
-
Recent advances in regulating the proliferation or maturation of human-induced pluripotent stem cell-derived cardiomyocytes
Stem Cell Research & Therapy (2023)
-
Long-term in vitro recording of cardiac action potentials on microelectrode arrays for chronic cardiotoxicity assessment
Archives of Toxicology (2023)
-
The method of sinus node-like pacemaker cells from human induced pluripotent stem cells by BMP and Wnt signaling
Cell Biology and Toxicology (2023)
-
Reprogramming of fibroblasts into expandable cardiovascular progenitor cells via small molecules in xeno-free conditions
Nature Biomedical Engineering (2022)
-
Surfing the clinical trials of mesenchymal stem cell therapy in ischemic cardiomyopathy
Stem Cell Research & Therapy (2021)