Abstract
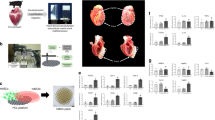
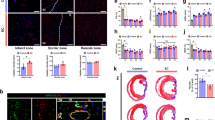
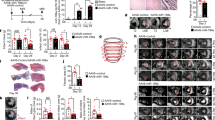
In a cell-free approach to regenerative therapeutics, transient application of paracrine factors in vivo could be used to alter the behavior and fate of progenitor cells to achieve sustained clinical benefits. Here we show that intramyocardial injection of synthetic modified RNA (modRNA) encoding human vascular endothelial growth factor-A (VEGF-A) results in the expansion and directed differentiation of endogenous heart progenitors in a mouse myocardial infarction model. VEGF-A modRNA markedly improved heart function and enhanced long-term survival of recipients. This improvement was in part due to mobilization of epicardial progenitor cells and redirection of their differentiation toward cardiovascular cell types. Direct in vivo comparison with DNA vectors and temporal control with VEGF inhibitors revealed the greatly increased efficacy of pulse-like delivery of VEGF-A. Our results suggest that modRNA is a versatile approach for expressing paracrine factors as cell fate switches to control progenitor cell fate and thereby enhance long-term organ repair.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
19 September 2013
In the version of this article initially published online, W.T.P. was not included in the correspondence line. Three sentences have also been added to Author Contributions. The omissions have been corrected for the print, PDF and HTML versions of this article.
References
Kariko, K., Buckstein, M., Ni, H. & Weissman, D. Suppression of RNA recognition by Toll-like receptors: the impact of nucleoside modification and the evolutionary origin of RNA. Immunity 23, 165–175 (2005).
Kariko, K., Muramatsu, H., Keller, J.M. & Weissman, D. Increased erythropoiesis in mice injected with submicrogram quantities of pseudouridine-containing mRNA encoding erythropoietin. Mol. Ther. 20, 948–953 (2012).
Kariko, K. et al. Incorporation of pseudouridine into mRNA yields superior nonimmunogenic vector with increased translational capacity and biological stability. Mol. Ther. 16, 1833–1840 (2008).
Kormann, M.S. et al. Expression of therapeutic proteins after delivery of chemically modified mRNA in mice. Nat. Biotechnol. 29, 154–157 (2011).
Mays, L.E. et al. Modified Foxp3 mRNA protects against asthma through an IL-10-dependent mechanism. J. Clin. Invest. 123, 1216–1228 (2013).
Wessels, A. et al. Epicardially derived fibroblasts preferentially contribute to the parietal leaflets of the atrioventricular valves in the murine heart. Dev. Biol. 366, 111–124 (2012).
Moretti, A. et al. Multipotent embryonic isl1+ progenitor cells lead to cardiac, smooth muscle, and endothelial cell diversification. Cell 127, 1151–1165 (2006).
Qyang, Y. et al. The renewal and differentiation of Isl1+ cardiovascular progenitors are controlled by a Wnt/beta-catenin pathway. Cell Stem Cell 1, 165–179 (2007).
Bu, L. et al. Human ISL1 heart progenitors generate diverse multipotent cardiovascular cell lineages. Nature 460, 113–117 (2009).
Laugwitz, K.L. et al. Postnatal isl1+ cardioblasts enter fully differentiated cardiomyocyte lineages. Nature 433, 647–653 (2005).
Domian, I.J. et al. Generation of functional ventricular heart muscle from mouse ventricular progenitor cells. Science 326, 426–429 (2009).
Chong, J.J. et al. Adult cardiac-resident MSC-like stem cells with a proepicardial origin. Cell Stem Cell 9, 527–540 (2011).
Cai, C.L. et al. A myocardial lineage derives from Tbx18 epicardial cells. Nature 454, 104–108 (2008).
Loffredo, F.S., Steinhauser, M.L., Gannon, J. & Lee, R.T. Bone marrow-derived cell therapy stimulates endogenous cardiomyocyte progenitors and promotes cardiac repair. Cell Stem Cell 8, 389–398 (2011).
Zhou, B. et al. Epicardial progenitors contribute to the cardiomyocyte lineage in the developing heart. Nature 454, 109–113 (2008).
Zhou, B. et al. Adult mouse epicardium modulates myocardial injury by secreting paracrine factors. J. Clin. Invest. 121, 1894–1904 (2011).
Smart, N. et al. De novo cardiomyocytes from within the activated adult heart after injury. Nature 474, 640–644 (2011).
Garbern, J.C. & Lee, R.T. Cardiac stem cell therapy and the promise of heart regeneration. Cell Stem Cell 12, 689–698 (2013).
Malliaras, K. et al. Cardiomyocyte proliferation and progenitor cell recruitment underlie therapeutic regeneration after myocardial infarction in the adult mouse heart. EMBO Mol. Med. 5, 191–209 (2013).
Ptaszek, L.M., Mansour, M., Ruskin, J.N. & Chien, K.R. Towards regenerative therapy for cardiac disease. Lancet 379, 933–942 (2012).
von Gise, A. et al. WT1 regulates epicardial epithelial to mesenchymal transition through beta-catenin and retinoic acid signaling pathways. Dev. Biol. 356, 421–431 (2011).
Svensson, E.C. et al. Efficient and stable transduction of cardiomyocytes after intramyocardial injection or intracoronary perfusion with recombinant adeno-associated virus vectors. Circulation 99, 201–205 (1999).
Patterson, C. & Runge, M.S. Therapeutic myocardial angiogenesis via vascular endothelial growth factor gene therapy: moving on down the road. Circulation 102, 940–942 (2000).
Hao, X. et al. Myocardial angiogenesis after plasmid or adenoviral VEGF-A(165) gene transfer in rat myocardial infarction model. Cardiovasc. Res. 73, 481–487 (2007).
Eppler, S.M. et al. A target-mediated model to describe the pharmacokinetics and hemodynamic effects of recombinant human vascular endothelial growth factor in humans. Clin. Pharmacol. Ther. 72, 20–32 (2002).
Dor, Y. et al. Conditional switching of VEGF provides new insights into adult neovascularization and pro-angiogenic therapy. EMBO J. 21, 1939–1947 (2002).
Tafuro, S. et al. Inducible adeno-associated virus vectors promote functional angiogenesis in adult organisms via regulated vascular endothelial growth factor expression. Cardiovasc. Res. 83, 663–671 (2009).
Lee, R.J. et al. VEGF gene delivery to myocardium: deleterious effects of unregulated expression. Circulation 102, 898–901 (2000).
Lui, K. et al. Driving vascular endothelial cell fate of human multipotent Isl1+ heart progenitors with VEGF modified mRNA. Cell Res. doi:10.1038/cr.2013.112 (10 September 2013).
Djurovic, S., Iversen, N., Jeansson, S., Hoover, F. & Christensen, G. Comparison of nonviral transfection and adeno-associated viral transduction on cardiomyocytes. Mol. Biotechnol. 28, 21–32 (2004).
Grunewald, M. et al. VEGF-induced adult neovascularization: recruitment, retention, and role of accessory cells. Cell 124, 175–189 (2006).
Warren, L. et al. Highly efficient reprogramming to pluripotency and directed differentiation of human cells with synthetic modified mRNA. Cell Stem Cell 7, 618–630 (2010).
Choi, M.K. et al. A selective contribution of the RIG-I-like receptor pathway to type I interferon responses activated by cytosolic DNA. Proc. Natl. Acad. Sci. USA 106, 17870–17875 (2009).
Stetson, D.B. & Medzhitov, R. Recognition of cytosolic DNA activates an IRF3-dependent innate immune response. Immunity 24, 93–103 (2006).
Su, H. et al. Additive effect of AAV-mediated angiopoietin-1 and VEGF expression on the therapy of infarcted heart. Int. J. Cardiol. 133, 191–197 (2009).
Spilsbury, K., Garrett, K.L., Shen, W.Y., Constable, I.J. & Rakoczy, P.E. Overexpression of vascular endothelial growth factor (VEGF) in the retinal pigment epithelium leads to the development of choroidal neovascularization. Am. J. Pathol. 157, 135–144 (2000).
Nagy, J.A. et al. Permeability properties of tumor surrogate blood vessels induced by VEGF-A. Lab. Invest. 86, 767–780 (2006).
Noyan-Ashraf, M.H. et al. GLP-1R agonist liraglutide activates cytoprotective pathways and improves outcomes after experimental myocardial infarction in mice. Diabetes 58, 975–983 (2009).
Sauve, M. et al. Genetic deletion or pharmacological inhibition of dipeptidyl peptidase-4 improves cardiovascular outcomes after myocardial infarction in mice. Diabetes 59, 1063–1073 (2010).
Yuan, M.J. et al. Myocardial angiogenesis after chronic ghrelin treatment in a rat myocardial infarction model. Regul. Pept. 179, 39–42 (2012).
Shen, B.Q. et al. Homologous up-regulation of KDR/Flk-1 receptor expression by vascular endothelial growth factor in vitro. J. Biol. Chem. 273, 29979–29985 (1998).
Wilm, B., Ipenberg, A., Hastie, N.D., Burch, J.B. & Bader, D.M. The serosal mesothelium is a major source of smooth muscle cells of the gut vasculature. Development 132, 5317–5328 (2005).
Muzumdar, M.D., Tasic, B., Miyamichi, K., Li, L. & Luo, L. A global double-fluorescent Cre reporter mouse. Genesis 45, 593–605 (2007).
Hoshijima, M. et al. Chronic suppression of heart-failure progression by a pseudophosphorylated mutant of phospholamban via in vivo cardiac rAAV gene delivery. Nat. Med. 8, 864–871 (2002).
Kruithof, B.P. et al. BMP and FGF regulate the differentiation of multipotential pericardial mesoderm into the myocardial or epicardial lineage. Dev. Biol. 295, 507–522 (2006).
Katz, T.C. et al. Distinct compartments of the proepicardial organ give rise to coronary vascular endothelial cells. Dev. Cell 22, 639–650 (2012).
Zhou, B. & Pu, W.T. Genetic Cre-loxP assessment of epicardial cell fate using Wt1-driven Cre alleles. Circ. Res. 111, e276–e280 (2012).
Christoffels, V.M. et al. Tbx18 and the fate of epicardial progenitors. Nature 458, E8–E9, E9–10 (2009).
Soriano, P. Generalized lacZ expression with the ROSA26 Cre reporter strain. Nat. Genet. 21, 70–71 (1999).
Liao, R. & Jain, M. Isolation, culture, and functional analysis of adult mouse cardiomyocytes. Methods Mol. Med. 139, 251–262 (2007).
Tarnavski, O. et al. Mouse cardiac surgery: comprehensive techniques for the generation of mouse models of human diseases and their application for genomic studies. Physiol. Genomics 16, 349–360 (2004).
Takagawa, J. et al. Myocardial infarct size measurement in the mouse chronic infarction model: comparison of area- and length-based approaches. J. Appl. Physiol. 102, 2104–2111 (2007).
Lee, W.W. et al. PET/MRI of inflammation in myocardial infarction. J. Am. Coll. Cardiol. 59, 153–163 (2012).
Cerletti, M. et al. Highly efficient, functional engraftment of skeletal muscle stem cells in dystrophic muscles. Cell 134, 37–47 (2008).
Sherwood, R.I. et al. Isolation of adult mouse myogenic progenitors: functional heterogeneity of cells within and engrafting skeletal muscle. Cell 119, 543–554 (2004).
Acknowledgements
This work was funded by US National Institutes of Health U01H100408 (K.R.C.), U01HL098166 (K.R.C.), U01JL100401 (W.T.P.), R01HL094683 (W.T.P.), RC1HL099618 (K.R.C., W.T.P.) and UO1HL100402 (A.J.W.). K.O.L. held a Croucher Foundation Fellowship and A.J.W. is an Early Career Scientist of the Howard Hughes Medical Institute. We thank R. Liao, J. Guan, J. Truelove, L. Bu, M. Stachel, K. Buac, V. Priestly, R. Gazit, K. Ketman, N. Barteneva, A. He, S. Stevens, B. Zhou and L.Warren for all their help in this project. Adult cardiomyocytes were a kind gift from R. Liao (Biological and Biomaterial Science, Department of Medicine, Brigham and Women's Hospital, Harvard Medical School).
Author information
Authors and Affiliations
Contributions
L.Z. (lzangi@enders.tch.harvard.edu) worked in the Rossi, Chien and Pu laboratories, and designed and carried out most of the experiments, analyzed most of the data, and wrote the manuscript. K.O.L. in the Chien lab designed and performed experiments and analyzed the qRT-PCR and immunostaining data, and wrote the manuscript. Her contribution is similar in significance to the contributions of L.Z. A.v.G. performed and analyzed the Wt1-related experiments. Q.M. and R.G. carried out myocardial infarction experiments. W.E. carried out plasmid preparation. L.M.P. performed blinded analysis of imaging data and wrote the manuscript. D.S. performed and analyzed skeletal muscle in vivo transfection. H.X. performed isolation of neonatal mouse cardiomyocytes. M.T. performed and analyzed in vitro transfection of mouse adult myotubes. B.S. carried out and analyzed the MRI experiment. M.N., D.M.B., R.A.L. and A.J.W. designed experiments, analyzed data, and revised the manuscript. D.J.R. (Derrick.Rossi@childrens.harvard.edu) designed in vitro cardiomyocyte experiments and revised the manuscript. K.R.C. (kchien@harvard.edu; kenneth.chien@ki.se) conceived the initial project and experimental studies, and with W.T.P. (wpu@enders.tch.harvard.edu) designed further experiments, analyzed data, and wrote the manuscript. The initial discovery of VEGF-A as a cell fate switch for heart progenitors in general, and its effects in myocardial infarction on the expansion of epicardial heart progenitors along with vascular regeneration was made in the Chien lab (L.Z., K.O.L. and K.R.C.). The Chien lab (L.Z., K.R.C.) and the Pu lab (A.v.G., W.T.P.) worked together to extend and expand these initial results. Address correspondence on ModRNA reagents, delivery and protocols to K.R.C. and on mouse models to W.T.P.
Corresponding authors
Ethics declarations
Competing interests
K.R.C. and D.J.R. are co-founders of Moderna Therapeutics, a Cambridge, Massachusetts company that is developing therapeutics based on modified mRNA. K.R.C. is an advisor to AstraZeneca, which has an interest in cardiovascular therapeutic applications of modRNA.
Supplementary information
Supplementary Text and Figures
Supplemental Appendix, Supplementary Tables 1-6, and Supplementary Figures 1-12 (PDF 24781 kb)
Supplementary video 1
Cine MRI of sham-operated heart, 21 days after operation (MOV 915 kb)
Supplementary video 2
Cine MRI of MI heart, vehicle treated heart, 21 days after operation (MOV 836 kb)
Supplementary video 3
Cine MRI of MI heart, VEGF-A modRNA treated, 21 days after operation (MOV 871 kb)
Rights and permissions
About this article
Cite this article
Zangi, L., Lui, K., von Gise, A. et al. Modified mRNA directs the fate of heart progenitor cells and induces vascular regeneration after myocardial infarction. Nat Biotechnol 31, 898–907 (2013). https://doi.org/10.1038/nbt.2682
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.2682
This article is cited by
-
N1-methylpseudouridylation of mRNA causes +1 ribosomal frameshifting
Nature (2024)
-
Strategies for non-viral vectors targeting organs beyond the liver
Nature Nanotechnology (2024)
-
Regeneration of the heart: from molecular mechanisms to clinical therapeutics
Military Medical Research (2023)
-
Enhancement of bone regeneration by coadministration of angiogenic and osteogenic factors using messenger RNA
Inflammation and Regeneration (2023)
-
Amplifying gene expression with RNA-targeted therapeutics
Nature Reviews Drug Discovery (2023)