Abstract
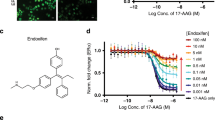
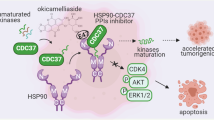
The interaction between the HSP90 chaperone and its client kinases is sensitive to the conformational status of the kinase, and stabilization of the kinase fold by small molecules strongly decreases chaperone interaction. Here we exploit this observation and assay small-molecule binding to kinases in living cells, using chaperones as 'thermodynamic sensors'. The method allows determination of target specificities of both ATP-competitive and allosteric inhibitors in the kinases' native cellular context in high throughput. We profile target specificities of 30 diverse kinase inhibitors against >300 kinases. Demonstrating the value of the assay, we identify ETV6-NTRK3 as a target of the FDA-approved drug crizotinib (Xalkori). Crizotinib inhibits proliferation of ETV6-NTRK3-dependent tumor cells with nanomolar potency and induces the regression of established tumor xenografts in mice. Finally, we show that our approach is applicable to other chaperone and target classes by assaying HSP70/steroid hormone receptor and CDC37/kinase interactions, suggesting that chaperone interactions will have broad application in detecting drug-target interactions in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Taipale, M. et al. Quantitative analysis of HSP90-client interactions reveals principles of substrate recognition. Cell 150, 987–1001 (2012).
Goldstein, D.M., Gray, N.S. & Zarrinkar, P.P. High-throughput kinase profiling as a platform for drug discovery. Nat. Rev. Drug Discov. 7, 391–397 (2008).
Karaman, M.W. et al. A quantitative analysis of kinase inhibitor selectivity. Nat. Biotechnol. 26, 127–132 (2008).
Fabian, M.A. et al. A small molecule-kinase interaction map for clinical kinase inhibitors. Nat. Biotechnol. 23, 329–336 (2005).
Davis, M.I. et al. Comprehensive analysis of kinase inhibitor selectivity. Nat. Biotechnol. 29, 1046–1051 (2011).
Fedorov, O. et al. A systematic interaction map of validated kinase inhibitors with Ser/Thr kinases. Proc. Natl. Acad. Sci. USA 104, 20523–20528 (2007).
Anastassiadis, T., Deacon, S.W., Devarajan, K., Ma, H. & Peterson, J.R. Comprehensive assay of kinase catalytic activity reveals features of kinase inhibitor selectivity. Nat. Biotechnol. 29, 1039–1045 (2011).
Bain, J. et al. The selectivity of protein kinase inhibitors: a further update. Biochem. J. 408, 297–315 (2007).
Eck, M.J. & Manley, P.W. The interplay of structural information and functional studies in kinase drug design: insights from BCR-Abl. Curr. Opin. Cell Biol. 21, 288–295 (2009).
Posy, S.L. et al. Trends in kinase selectivity: insights for target class-focused library screening. J. Med. Chem. 54, 54–66 (2011).
Bantscheff, M. et al. Quantitative chemical proteomics reveals mechanisms of action of clinical ABL kinase inhibitors. Nat. Biotechnol. 25, 1035–1044 (2007).
Patricelli, M.P. et al. In situ kinase profiling reveals functionally relevant properties of native kinases. Chem. Biol. 18, 699–710 (2011).
Barrios-Rodiles, M. et al. High-throughput mapping of a dynamic signaling network in mammalian cells. Science 307, 1621–1625 (2005).
Shah, N.P. et al. Overriding imatinib resistance with a novel ABL kinase inhibitor. Science 305, 399–401 (2004).
O'Hare, T. et al. AP24534, a pan-BCR-ABL inhibitor for chronic myeloid leukemia, potently inhibits the T315I mutant and overcomes mutation-based resistance. Cancer Cell 16, 401–412 (2009).
Waldron, T.T. & Murphy, K.P. Stabilization of proteins by ligand binding: application to drug screening and determination of unfolding energetics. Biochemistry 42, 5058–5064 (2003).
Vaughan, C.K. et al. Structure of an Hsp90-Cdc37-Cdk4 complex. Mol. Cell 23, 697–707 (2006).
Zhang, J. et al. Targeting Bcr-Abl by combining allosteric with ATP-binding-site inhibitors. Nature 463, 501–506 (2010).
Yang, J. et al. Discovery and characterization of a cell-permeable, small-molecule c-Abl kinase activator that binds to the myristoyl binding site. Chem. Biol. 18, 177–186 (2011).
Iacob, R.E., Zhang, J., Gray, N.S. & Engen, J.R. Allosteric interactions between the myristate- and ATP-site of the Abl kinase. PLoS ONE 6, e15929 (2011).
Xu, W. et al. Surface charge and hydrophobicity determine ErbB2 binding to the Hsp90 chaperone complex. Nat. Struct. Mol. Biol. 12, 120–126 (2005).
Picard, D. Chaperoning steroid hormone action. Trends Endocrinol. Metab. 17, 229–235 (2006).
Couture, P., Theriault, C., Simard, J. & Labrie, F. Androgen receptor-mediated stimulation of 17 beta-hydroxysteroid dehydrogenase activity by dihydrotestosterone and medroxyprogesterone acetate in ZR-75–1 human breast cancer cells. Endocrinology 132, 179–185 (1993).
Adcock, I.M., Nasuhara, Y., Stevens, D.A. & Barnes, P.J. Ligand-induced differentiation of glucocorticoid receptor (GR) trans-repression and transactivation: preferential targetting of NF-kappaB and lack of I-kappaB involvement. Br. J. Pharmacol. 127, 1003–1011 (1999).
Gozgit, J.M. et al. Ponatinib (AP24534), a multitargeted pan-FGFR inhibitor with activity in multiple FGFR-amplified or mutated cancer models. Mol. Cancer Ther. 11, 690–699 (2012).
Gozgit, J.M. et al. Potent activity of ponatinib (AP24534) in models of FLT3-driven acute myeloid leukemia and other hematologic malignancies. Mol. Cancer Ther. 10, 1028–1035 (2011).
Dar, A.C. & Shokat, K.M. The evolution of protein kinase inhibitors from antagonists to agonists of cellular signaling. Annu. Rev. Biochem. 80, 769–795 (2011).
Azam, M., Latek, R.R. & Daley, G.Q. Mechanisms of autoinhibition and STI-571/imatinib resistance revealed by mutagenesis of BCR-ABL. Cell 112, 831–843 (2003).
Kwak, E.L. et al. Anaplastic lymphoma kinase inhibition in non-small-cell lung cancer. N. Engl. J. Med. 363, 1693–1703 (2010).
Zou, H.Y. et al. An orally available small-molecule inhibitor of c-Met, PF-2341066, exhibits cytoreductive antitumor efficacy through antiproliferative and antiangiogenic mechanisms. Cancer Res. 67, 4408–4417 (2007).
Lannon, C.L. & Sorensen, P.H. ETV6-NTRK3: a chimeric protein tyrosine kinase with transformation activity in multiple cell lineages. Semin. Cancer Biol. 15, 215–223 (2005).
Orbach, D. et al. Infantile fibrosarcoma: management based on the European experience. J. Clin. Oncol. 28, 318–323 (2010).
Eguchi, M. et al. Fusion of ETV6 to neurotrophin-3 receptor TRKC in acute myeloid leukemia with t(12;15)(p13;q25). Blood 93, 1355–1363 (1999).
Kralik, J.M. et al. Characterization of a newly identified ETV6-NTRK3 fusion transcript in acute myeloid leukemia. Diagn. Pathol. 6, 19 (2011).
Skalova, A. et al. Mammary analogue secretory carcinoma of salivary glands, containing the ETV6-NTRK3 fusion gene: a hitherto undescribed salivary gland tumor entity. Am. J. Surg. Pathol. 34, 599–608 (2010).
Okabe, M. et al. Megakaryocytic differentiation of a leukemic cell line, MC3, by phorbol ester: induction of glycoprotein IIb/IIIa and effects on expression of IL-6, IL-6 receptor, mpl and GATA genes. Leuk. Res. 19, 933–943 (1995).
Tognon, C. et al. The chimeric protein tyrosine kinase ETV6-NTRK3 requires both Ras-Erk1/2 and PI3-kinase-Akt signaling for fibroblast transformation. Cancer Res. 61, 8909–8916 (2001).
Galkin, A.V. et al. Identification of NVP-TAE684, a potent, selective, and efficacious inhibitor of NPM-ALK. Proc. Natl. Acad. Sci. USA 104, 270–275 (2007).
Gadd, S. et al. Mediators of receptor tyrosine kinase activation in infantile fibrosarcoma: a Children's Oncology Group study. J. Pathol. 228, 119–130 (2012).
Chaires, J.B. Calorimetry and thermodynamics in drug design. Annu. Rev. Biophys. 37, 135–151 (2008).
Gong, Y. et al. An atlas of chaperone-protein interactions in Saccharomyces cerevisiae: implications to protein folding pathways in the cell. Mol. Syst. Biol. 5, 275 (2009).
Kerner, M.J. et al. Proteome-wide analysis of chaperonin-dependent protein folding in Escherichia coli. Cell 122, 209–220 (2005).
Mosse, Y.P. et al. Efficacy of crizotinib in children with relapsed/refractory ALK-driven tumors including anaplastic large cell lymphoma and neuroblastoma: a Children's Oncology Group phase I consortium study. J. Clin. Oncol. Supplement Abstract 9500 (2012).
Novo, F.J., de Mendibil, I.O. & Vizmanos, J.L. TICdb: a collection of gene-mapped translocation breakpoints in cancer. BMC Genomics 8, 33 (2007).
Atanassov, I.I., Etchells, J.P. & Turner, S.R. A simple, flexible and efficient PCR-fusion/Gateway cloning procedure for gene fusion, site-directed mutagenesis, short sequence insertion and domain deletions and swaps. Plant Methods 5, 14 (2009).
Koh, E.Y., Chen, T. & Daley, G.Q. Novel retroviral vectors to facilitate expression screens in mammalian cells. Nucleic Acids Res. 30, e142 (2002).
Acknowledgements
We thank the Lindquist laboratory members for valuable discussions and comments on the manuscript. We also thank M. Azam (Cincinnati Children's Hospital Medical Center) for providing mutant BCR-ABL clones and P. Thiru, I. Barrasa and G. Bell (Whitehead Institute) for help with primer design and statistical analysis. M.T. was supported by Human Frontier Science Programme long-term fellowship. S.L. is a Howard Hughes Medical Institute investigator. Support for this study was also provided by the US National Institutes of Health (NIH) Genomics Based Drug Discovery-Driving Medical Projects grant UL1-DE019585, administratively linked to NIH grants RL1-GM084437, RL1-CA133834 and RL1-HG004671.
Author information
Authors and Affiliations
Contributions
M.T. and S.L. planned the project. M.T. designed the experiments, developed the assay, performed the experiments together with I.K., and analyzed the data. Xenograft experiments were done by L.W., and S.S. performed immunohistochemistry on tumor samples. J.Z. tested BCR-ABL variants for GNF-2 sensitivity in BaF/3 cells. J.Z., Q.L. and N.S.G. synthesized and provided kinase inhibitors and helped design experiments. M.T. and S.L. wrote the paper with input from all co-authors.
Corresponding author
Ethics declarations
Competing interests
M.T. and S.L. are named inventors on a patent application on the technology described in this manuscript.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–13, Supplementary Table 1 and Supplementary Discussion (PDF 2174 kb)
Rights and permissions
About this article
Cite this article
Taipale, M., Krykbaeva, I., Whitesell, L. et al. Chaperones as thermodynamic sensors of drug-target interactions reveal kinase inhibitor specificities in living cells. Nat Biotechnol 31, 630–637 (2013). https://doi.org/10.1038/nbt.2620
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.2620
This article is cited by
-
MEK1/2 regulate normal BCR and ABL1 tumor-suppressor functions to dictate ATO response in TKI-resistant Ph+ leukemia
Leukemia (2023)
-
Development of actionable targets of multi-kinase inhibitors (AToMI) screening platform to dissect kinase targets of staurosporines in glioblastoma cells
Scientific Reports (2022)
-
Differential maturation and chaperone dependence of the paralogous protein kinases DYRK1A and DYRK1B
Scientific Reports (2022)
-
Molecular matched targeted therapies for primary brain tumors—a single center retrospective analysis
Journal of Neuro-Oncology (2022)
-
Chaperone mediated detection of small molecule target binding in cells
Nature Communications (2020)