Abstract
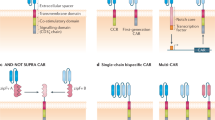
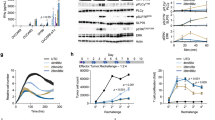
Current T-cell engineering approaches redirect patient T cells to tumors by transducing them with antigen-specific T-cell receptors (TCRs) or chimeric antigen receptors (CARs) that target a single antigen1,2,3. However, few truly tumor-specific antigens have been identified, and healthy tissues that express the targeted antigen may undergo T cell–mediated damage4,5,6,7. Here we present a strategy to render T cells specific for a tumor in the absence of a truly tumor-restricted antigen. T cells are transduced with both a CAR that provides suboptimal activation upon binding of one antigen and a chimeric costimulatory receptor (CCR) that recognizes a second antigen. Using the prostate tumor antigens PSMA and PSCA, we show that co-transduced T cells destroy tumors that express both antigens but do not affect tumors expressing either antigen alone. This 'tumor-sensing' strategy may help broaden the applicability and avoid some of the side effects of targeted T-cell therapies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Sadelain, M., Riviere, I. & Brentjens, R. Targeting tumours with genetically enhanced T lymphocytes. Nat. Rev. Cancer 3, 35–45 (2003).
Ho, W.Y., Blattman, J.N., Dossett, M.L., Yee, C. & Greenberg, P.D. Adoptive immunotherapy: engineering T cell responses as biologic weapons for tumor mass destruction. Cancer Cell 3, 431–437 (2003).
Sadelain, M., Brentjens, R. & Riviere, I. The promise and potential pitfalls of chimeric antigen receptors. Curr. Opin. Immunol. 21, 215–223 (2009).
Robbins, P.F. et al. Tumor regression in patients with metastatic synovial cell sarcoma and melanoma using genetically engineered lymphocytes reactive with NY-ESO-1. J. Clin. Oncol. 29, 917–924 (2011).
Kalos, M. et al. T cells with chimeric antigen receptors have potent antitumor effects and can establish memory in patients with advanced leukemia. Sci. Transl. Med. 3, 95ra73 (2011).
Brentjens, R.J. et al. Safety and persistence of adoptively transferred autologous CD19-targeted T cells in patients with relapsed or chemotherapy refractory B-cell leukemias. Blood 118, 4817–4828 (2011).
Kochenderfer, J.N. et al. B-cell depletion and remissions of malignancy along with cytokine-associated toxicity in a clinical trial of anti-CD19 chimeric-antigen-receptor-transduced T cells. Blood 119, 2709–2720 (2012).
Lamers, C.H. et al. Treatment of metastatic renal cell carcinoma with autologous T-lymphocytes genetically retargeted against carbonic anhydrase IX: first clinical experience. J. Clin. Oncol. 24, e20–e22 (2006).
Morgan, R.A. et al. Case report of a serious adverse event following the administration of T cells transduced with a chimeric antigen receptor recognizing ERBB2. Mol. Ther. 18, 843–851 (2010).
Krause, A. et al. Antigen-dependent CD28 signaling selectively enhances survival and proliferation in genetically modified activated human primary T lymphocytes. J. Exp. Med. 188, 619–626 (1998).
Duong, C.P., Westwood, J.A., Berry, L.J., Darcy, P.K. & Kershaw, M.H. Enhancing the specificity of T-cell cultures for adoptive immunotherapy of cancer. Immunotherapy 3, 33–48 (2011).
Wilkie, S. et al. Dual targeting of ErbB2 and MUC1 in breast cancer using chimeric antigen receptors engineered to provide complementary signaling. J. Clin. Immunol. 1059–1070 (2012).
Brentjens, R.J. et al. Eradication of systemic B-cell tumors by genetically targeted human T lymphocytes co-stimulated by CD80 and interleukin-15. Nat. Med. 9, 279–286 (2003).
Maher, J., Brentjens, R.J., Gunset, G., Riviere, I. & Sadelain, M. Human T-lymphocyte cytotoxicity and proliferation directed by a single chimeric TCRzeta/CD28 receptor. Nat. Biotechnol. 20, 70–75 (2002).
Watts, T.H. TNF/TNFR family members in costimulation of T cell responses. Annu. Rev. Immunol. 23, 23–68 (2005).
Stephan, M.T. et al. T cell-encoded CD80 and 4–1BBL induce auto- and transcostimulation, resulting in potent tumor rejection. Nat. Med. 13, 1440–1449 (2007).
Carpenito, C. et al. Control of large, established tumor xenografts with genetically retargeted human T cells containing CD28 and CD137 domains. Proc. Natl. Acad. Sci. USA 106, 3360–3365 (2009).
Tammana, S. et al. 4–1BB and CD28 signaling plays a synergistic role in redirecting umbilical cord blood T cells against B-cell malignancies. Hum. Gene Ther. 21, 75–86 (2010).
Wang, J. et al. Optimizing adoptive polyclonal T cell immunotherapy of lymphomas, using a chimeric T cell receptor possessing CD28 and CD137 costimulatory domains. Hum. Gene Ther. 18, 712–725 (2007).
Zhong, X.S., Matsushita, M., Plotkin, J., Riviere, I. & Sadelain, M. Chimeric antigen receptors combining 4–1BB and CD28 signaling domains augment PI3kinase/AKT/Bcl-XL activation and CD8+ T cell-mediated tumor eradication. Mol. Ther. 18, 413–420 (2010).
Liao, W., Lin, J.X. & Leonard, W.J. IL-2 family cytokines: new insights into the complex roles of IL-2 as a broad regulator of T helper cell differentiation. Curr. Opin. Immunol. 23, 598–604 (2011).
Shin, S.M. et al. 4–1BB triggers IL-13 production from T cells to limit the polarized, Th1-mediated inflammation. J. Leukoc. Biol. 81, 1455–1465 (2007).
Schwartz, R.H. T cell anergy. Annu. Rev. Immunol. 21, 305–334 (2003).
Saeki, N., Gu, J., Yoshida, T. & Wu, X. Prostate stem cell antigen: a Jekyll and Hyde molecule? Clin. Cancer Res. 16, 3533–3538 (2010).
Lam, J.S. et al. Prostate stem cell antigen is overexpressed in prostate cancer metastases. Clin. Cancer Res. 11, 2591–2596 (2005).
Silver, D.A., Pellicer, I., Fair, W.R., Heston, W.D. & Cordon-Cardo, C. Prostate-specific membrane antigen expression in normal and malignant human tissues. Clin. Cancer Res. 3, 81–85 (1997).
Liu, J.C. et al. Seventeen-gene signature from enriched Her2/Neu mammary tumor-initiating cells predicts clinical outcome for human HER2+:ERalpha- breast cancer. Proc. Natl. Acad. Sci. USA 109, 5832–5837 (2012).
Meyer, M.J. et al. CD44posCD49fhiCD133/2hi defines xenograft-initiating cells in estrogen receptor-negative breast cancer. Cancer Res. 70, 4624–4633 (2010).
Shih, I.-M. & Davidson, B. Pathogenesis of ovarian cancer: clues from selected overexpressed genes. Future Oncol. 5, 1641–1657 (2009).
Strauss, R. et al. Analysis of epithelial and mesenchymal markers in ovarian cancer reveals phenotypic heterogeneity and plasticity. PLoS ONE 6, e16186 (2011).
Gade, T.P. et al. Targeted elimination of prostate cancer by genetically directed human T lymphocytes. Cancer Res. 65, 9080–9088 (2005).
Feldmann, A. et al. Retargeting of T cells to prostate stem cell antigen expressing tumor cells: comparison of different antibody formats. Prostate 71, 998–1011 (2011).
Feldmann, A. et al. Novel humanized and highly efficient bispecific antibodies mediate killing of prostate stem cell antigen-expressing tumor cells by CD8+ and CD4+ T cells. J. Immunol. 189, 3249–3259 (2012).
Orlandi, R., Gussow, D.H., Jones, P.T. & Winter, G. Cloning immunoglobulin variable domains for expression by the polymerase chain reaction. Proc. Natl. Acad. Sci. USA 86, 3833–3837 (1989).
Acknowledgements
We thank I. Riviere for reviewing the manuscript and J. Plotkin and G. Gunset for outstanding technical assistance. Our work is supported by Department of Defense Prostate Cancer Training Award PC101964 (C.C.K.), the Mr. William H. and Mrs. Alice Goodwin and the Commonwealth Foundation for Cancer Research, The Experimental Therapeutics Center of Memorial Sloan-Kettering Cancer Center, the Major Family Fund for Cancer Research at Memorial Sloan-Kettering, the support of Mr. and Mrs. Joel S. Mallah, and Mr. Lewis Sanders.
Author information
Authors and Affiliations
Contributions
C.C.K. designed and performed experiments, analyzed data, and wrote the manuscript. M. Condomines contributed reagents, performed experiments, interpreted results and reviewed the manuscript. M. Cartellieri contributed reagents and performed experiments. M.B. contributed reagents and designed experiments. M.S. designed experiments, analyzed data, interpreted results and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 (PDF 1332 kb)
Rights and permissions
About this article
Cite this article
Kloss, C., Condomines, M., Cartellieri, M. et al. Combinatorial antigen recognition with balanced signaling promotes selective tumor eradication by engineered T cells. Nat Biotechnol 31, 71–75 (2013). https://doi.org/10.1038/nbt.2459
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.2459
This article is cited by
-
CAR T therapies in multiple myeloma: unleashing the future
Cancer Gene Therapy (2024)
-
Challenges and new technologies in adoptive cell therapy
Journal of Hematology & Oncology (2023)
-
Sonogenetic control of multiplexed genome regulation and base editing
Nature Communications (2023)
-
Overcoming on-target, off-tumour toxicity of CAR T cell therapy for solid tumours
Nature Reviews Clinical Oncology (2023)
-
CAR T cell design: approaching the elusive AND-gate
Cell Research (2023)