Abstract
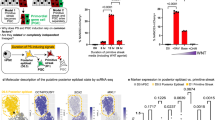
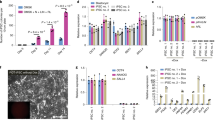
An important risk in the clinical application of human pluripotent stem cells (hPSCs), including human embryonic and induced pluripotent stem cells (hESCs and hiPSCs), is teratoma formation by residual undifferentiated cells. We raised a monoclonal antibody against hESCs, designated anti–stage-specific embryonic antigen (SSEA)-5, which binds a previously unidentified antigen highly and specifically expressed on hPSCs—the H type-1 glycan. Separation based on SSEA-5 expression through fluorescence-activated cell sorting (FACS) greatly reduced teratoma-formation potential of heterogeneously differentiated cultures. To ensure complete removal of teratoma-forming cells, we identified additional pluripotency surface markers (PSMs) exhibiting a large dynamic expression range during differentiation: CD9, CD30, CD50, CD90 and CD200. Immunohistochemistry studies of human fetal tissues and bioinformatics analysis of a microarray database revealed that concurrent expression of these markers is both common and specific to hPSCs. Immunodepletion with antibodies against SSEA-5 and two additional PSMs completely removed teratoma-formation potential from incompletely differentiated hESC cultures.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Blum, B. & Benvenisty, N. The tumorigenicity of human embryonic stem cells. Adv. Cancer Res. 100, 133–158 (2008).
Tang, C. & Drukker, M. Potential barriers to therapeutics utilizing pluripotent cell derivatives: intrinsic immunogenicity of in vitro maintained and matured populations. Semin. Immunopathol. published online, doi: 10.1007/s00281-011-0269-5 (11 April 2011).
Schuldiner, M., Itskovitz-Eldor, J. & Benvenisty, N. Selective ablation of human embryonic stem cells expressing a “suicide” gene. Stem Cells 21, 257–265 (2003).
Cao, F. et al. Molecular imaging of embryonic stem cell misbehavior and suicide gene ablation. Cloning Stem Cells 9, 107–117 (2007).
Choo, A.B. et al. Selection against undifferentiated human embryonic stem cells by a cytotoxic antibody recognizing podocalyxin-like protein-1. Stem Cells 26, 1454–1463 (2008).
Tan, H.L., Fong, W.J., Lee, E.H., Yap, M. & Choo, A. mAb 84, a cytotoxic antibody that kills undifferentiated human embryonic stem cells via oncosis. Stem Cells 27, 1792–1801 (2009).
Drukker, M., Muscat, C. & Weissman, I.L. Generation of a monoclonal antibody library against human embryonic stem cells. Methods Mol. Biol. 407, 63–81 (2007).
Andrews, P.W., Banting, G., Damjanov, I., Arnaud, D. & Avner, P. Three monoclonal antibodies defining distinct differentiation antigens associated with different high molecular weight polypeptides on the surface of human embryonal carcinoma cells. Hybridoma 3, 347–361 (1984).
Shevinsky, L.H., Knowles, B.B., Damjanov, I. & Solter, D. Monoclonal antibody to murine embryos defines a stage-specific embryonic antigen expressed on mouse embryos and human teratocarcinoma cells. Cell 30, 697–705 (1982).
Kannagi, R. et al. Stage-specific embryonic antigens (SSEA-3 and -4) are epitopes of a unique globo-series ganglioside isolated from human teratocarcinoma cells. EMBO J. 2, 2355–2361 (1983).
Thomson, J.A. et al. Embryonic stem cell lines derived from human blastocysts. Science 282, 1145–1147 (1998).
Reubinoff, B.E., Pera, M.F., Fong, C.Y., Trounson, A. & Bongso, A. Embryonic stem cell lines from human blastocysts: somatic differentiation in vitro. Nat. Biotechnol. 18, 399–404 (2000).
Behr, B. et al. Blastocyst-ET and monozygotic twinning. J. Assist. Reprod. Genet. 17, 349–351 (2000).
Stevens, L.C. & Little, C.C. Spontaneous testicular teratomas in an inbred strain of mice. Proc. Natl. Acad. Sci. USA 40, 1080–1087 (1954).
Hentze, H. et al. Teratoma formation by human embryonic stem cells: Evaluation of essential parameters for future safety studies. Stem Cell Res. 2, 198–210 (2009).
Wu, J.C., Sundaresan, G., Iyer, M. & Gambhir, S.S. Noninvasive optical imaging of firefly luciferase reporter gene expression in skeletal muscles of living mice. Mol. Ther. 4, 297–306 (2001).
Lee, A.S. et al. Effects of cell number on teratoma formation by human embryonic stem cells. Cell Cycle 8, 2608–2612 (2009).
Vodyanik, M.A. & Slukvin, II. Hematoendothelial differentiation of human embryonic stem cells. Curr. Protoc. Cell Biol. 36, 23.6.1–23.6.28 (2007).
Nagano, K., Yoshida, Y. & Isobe, T. Cell surface biomarkers of embryonic stem cells. Proteomics 8, 4025–4035 (2008).
Raman, R. et al. Advancing glycomics: implementation strategies at the consortium for functional glycomics. Glycobiology 16, 82R–90R (2006).
Torrado, J., Gutierrez Hoyos, A., Blasco, E., Larraz, J. & Fernandez Rivas, J.L. Immunohistological patterns of blood group ABO and type 1 chain (Lewis a Lewis b) and type 2 chain (H-2, Y) antigens in normal uterine cervix. Tissue Antigens 36, 8–11 (1990).
Rosler, E.S. et al. Long-term culture of human embryonic stem cells in feeder-free conditions. Dev. Dyn. 229, 259–274 (2004).
Mateizel, I. et al. Characterization of CD30 expression in human embryonic stem cell lines cultured in serum-free media and passaged mechanically. Hum. Reprod. 24, 2477–2489 (2009).
Draper, J.S., Pigott, C., Thomson, J.A. & Andrews, P.W. Surface antigens of human embryonic stem cells: changes upon differentiation in culture. J. Anat. 200, 249–258 (2002).
Yu, J. et al. Induced pluripotent stem cell lines derived from human somatic cells. Science 318, 1917–1920 (2007).
Sahoo, D. et al. MiDReG: a method of mining developmentally regulated genes using Boolean implications. Proc. Natl. Acad. Sci. USA 107, 5732–5737 (2010).
Inlay, M.A. et al. Ly6d marks the earliest stage of B-cell specification and identifies the branchpoint between B-cell and T-cell development. Genes Dev. 23, 2376–2381 (2009).
Solter, D. & Knowles, B.B. Monoclonal antibody defining a stage-specific mouse embryonic antigen (SSEA-1). Proc. Natl. Acad. Sci. USA 75, 5565–5569 (1978).
Hakomori, S. Glycosphingolipids in cellular interaction, differentiation, and oncogenesis. Annu. Rev. Biochem. 50, 733–764 (1981).
Liang, Y.J. et al. Switching of the core structures of glycosphingolipids from globo- and lacto- to ganglio-series upon human embryonic stem cell differentiation. Proc. Natl. Acad. Sci. USA 107, 22564–22569 (2010).
Sahoo, D., Dill, D.L., Gentles, A.J., Tibshirani, R. & Plevritis, S.K. Boolean implication networks derived from large scale, whole genome microarray datasets. Genome Biol. 9, R157 (2008).
Goldman, J.P. et al. Enhanced human cell engraftment in mice deficient in RAG2 and the common cytokine receptor gamma chain. Br. J. Haematol. 103, 335–342 (1998).
Acknowledgements
The authors acknowledge C. Contag for providing luciferase constructs, M. van de Rijn and K. Montgomery for their assistance scanning fetal array slides and providing online access to these slides, P. Chu for assistance with hematoxylin and eosin staining, C. Muscat and T. Naik for assistance with hybridoma culture, W. Zhang for assistance in cell culturing, the Consortium for Functional Glycomics for providing and testing glycan arrays, and T. Serwold and C. Bertozzi for critical advice. This work was supported by funds provided by the California Institute of Regenerative Medicine (CIRM) (Comprehensive grant RC1-00354-1). C.T. and A.S.L. are supported by the Howard Hughes Medical Institute Medical Fellows and the Stanford Medical Scholars Program, J.-P.V. is supported by the Deutsche Forschungsgemeinschaft, C.T., M.A.I., R.A. and M.D. are supported by CIRM (Comprehensive grant RC1-00354-1).
Author information
Authors and Affiliations
Contributions
C.T., J.-P.V., I.L.W. and M.D. designed the experiments and wrote the manuscript. C.T., A.S.L., J.-P.V., D.S., A.R.M., D.N., M.A.I. and M.D. performed the experiments and analyzed data. R.A., S.L.C., R.R.P., B.B. and J.C.W. provided samples and reagents. All authors endorse the full content of this work.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Table 1 and Supplementary Figures 1–7 (PDF 1998 kb)
Rights and permissions
About this article
Cite this article
Tang, C., Lee, A., Volkmer, JP. et al. An antibody against SSEA-5 glycan on human pluripotent stem cells enables removal of teratoma-forming cells. Nat Biotechnol 29, 829–834 (2011). https://doi.org/10.1038/nbt.1947
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.1947
This article is cited by
-
Optimizing Generation of Stem Cell-Derived Islet Cells
Stem Cell Reviews and Reports (2022)
-
A defined glycosylation regulatory network modulates total glycome dynamics during pluripotency state transition
Scientific Reports (2021)
-
The quest of cell surface markers for stem cell therapy
Cellular and Molecular Life Sciences (2021)
-
Mouse Pluripotent Stem Cell Differentiation Under Physiological Oxygen Reduces Residual Teratomas
Cellular and Molecular Bioengineering (2021)
-
Prevention of tumor risk associated with the reprogramming of human pluripotent stem cells
Journal of Experimental & Clinical Cancer Research (2020)