Abstract
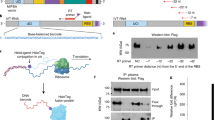
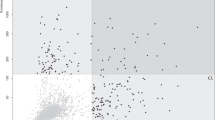
Immune responses targeting self-proteins (autoantigens) can lead to a variety of autoimmune diseases. Identification of these antigens is important for both diagnostic and therapeutic reasons. However, current approaches to characterize autoantigens have, in most cases, met only with limited success. Here we present a synthetic representation of the complete human proteome, the T7 peptidome phage display library (T7-Pep), and demonstrate its application to autoantigen discovery. T7-Pep is composed of >413,000 36-residue, overlapping peptides that cover all open reading frames in the human genome, and can be analyzed using high-throughput DNA sequencing. We developed a phage immunoprecipitation sequencing (PhIP-Seq) methodology to identify known and previously unreported autoantibodies contained in the spinal fluid of three individuals with paraneoplastic neurological syndromes. We also show how T7-Pep can be used more generally to identify peptide-protein interactions, suggesting the broader utility of our approach for proteomic research.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Graham, A.L. et al. Fitness correlates of heritable variation in antibody responsiveness in a wild mammal. Science 330, 662–665 (2010).
Faix, P.H. et al. Phage display of cDNA libraries: enrichment of cDNA expression using open reading frame selection. Biotechniques 36, 1018–1022 (2004).
Albert, M.L. & Darnell, R.B. Paraneoplastic neurological degenerations: keys to tumour immunity. Nat. Rev. Cancer 4, 36–44 (2004).
Wang, X. et al. Autoantibody signatures in prostate cancer. N. Engl. J. Med. 353, 1224–1235 (2005).
Anderson, K.S. et al. A protein microarray signature of autoantibody biomarkers for the early detection of breast cancer. J. Proteome Res. 10, 85–96 (2011).
Zacchi, P., Sblattero, D., Florian, F., Marzari, R. & Bradbury, A.R.M. Selecting open reading frames from DNA. Genome Res. 13, 980–990 (2003).
Kim, Y. et al. Identification of Hnrph3 as an autoantigen for acute anterior uveitis. Clin. Immunol. 138, 60–66 (2011).
Hughes, J.B., Hellmann, J.J., Ricketts, T.H. & Bohannan, B.J. Counting the uncountable: statistical approaches to estimating microbial diversity. Appl. Environ. Microbiol. 67, 4399–4406 (2001).
Swann, J.B. & Smyth, M.J. Immune surveillance of tumors. J. Clin. Invest. 117, 1137–1146 (2007).
Darnell, R.B. & Posner, J.B. Paraneoplastic syndromes involving the nervous system. N. Engl. J. Med. 349, 1543–1554 (2003).
Musunuru, K. & Kesari, S. Paraneoplastic opsoclonus-myoclonus ataxia associated with non-small-cell lung carcinoma. J. Neurooncol. 90, 213–216 (2008).
Srivastava, S. & Chen, L. A two-parameter generalized Poisson model to improve the analysis of RNA-seq data. Nucleic Acids Res. 38, e170 (2010).
Consul, P. & Shoukri, M. Maximum likelihood estimation for the generalized poisson distribution. Comm. Statist. Theory Methods 13, 1533–1547 (1984).
Bailey, T.L. & Elkan, C. Fitting a mixture model by expectation maximization to discover motifs in biopolymers. Proc. Int. Conf. Intell. Syst. Mol. Biol. 2, 28–36 (1994).
Almeida, L.G. et al. CTdatabase: a knowledge-base of high-throughput and curated data on cancer-testis antigens. Nucleic Acids Res. 37, D816–D819 (2009).
Rimoldi, D. et al. Efficient simultaneous presentation of NY-ESO-1/LAGE-1 primary and nonprimary open reading frame-derived CTL epitopes in melanoma. J. Immunol. 165, 7253–7261 (2000).
Chen, Y.T. et al. Identification of multiple cancer/testis antigens by allogeneic antibody screening of a melanoma cell line library. Proc. Natl. Acad. Sci. USA 95, 6919–6923 (1998).
Blanco-Arias, P., Sargent, C.A. & Affara, N.A. The human-specific Yp11.2/Xq21.3 homology block encodes a potentially functional testis-specific TGIF-like retroposon. Mamm. Genome 13, 463–468 (2002).
Berglund, L. et al. A genecentric human protein atlas for expression profiles based on antibodies. Mol. Cell. Proteomics 7, 2019–2027 (2008).
Li, L., Hagopian, W.A., Brashear, H.R., Daniels, T. & Lernmark, A. Identification of autoantibody epitopes of glutamic acid decarboxylase in stiff-man syndrome patients. J. Immunol. 152, 930–934 (1994).
Schwartz, H.L. et al. High-resolution autoreactive epitope mapping and structural modeling of the 65 kDa form of human glutamic acid decarboxylase. J. Mol. Biol. 287, 983–999 (1999).
Tanji, K. et al. TRIM9, a novel brain-specific E3 ubiquitin ligase, is repressed in the brain of Parkinson's disease and dementia with Lewy bodies. Neurobiol. Dis. 38, 210–218 (2010).
Rual, J.-F. et al. Towards a proteome-scale map of the human protein-protein interaction network. Nature 437, 1173–1178 (2005).
Ciccia, A. et al. The SIOD disorder protein SMARCAL1 is an RPA-interacting protein involved in replication fork restart. Genes Dev. 23, 2415–2425 (2009).
Mer, G. et al. Structural basis for the recognition of DNA repair proteins UNG2, XPA, and RAD52 by replication factor RPA. Cell 103, 449–456 (2000).
Barlow, D.J., Edwards, M.S. & Thornton, J.M. Continuous and discontinuous protein antigenic determinants. Nature 322, 747–748 (1986).
Jin, L., Fendly, B.M. & Wells, J.A. High resolution functional analysis of antibody-antigen interactions. J. Mol. Biol. 226, 851–865 (1992).
Miyazaki, K. et al. Analysis of in vivo role of alpha-fodrin autoantigen in primary Sjogren's syndrome. Am. J. Pathol. 167, 1051–1059 (2005).
Huang, M. et al. Detection of apoptosis-specific autoantibodies directed against granzyme B-induced cleavage fragments of the SS-B (La) autoantigen in sera from patients with primary Sjogren's syndrome. Clin. Exp. Immunol. 142, 148–154 (2005).
Robbins, D.C., Cooper, S.M., Fineberg, S.E. & Mead, P.M. Antibodies to covalent aggregates of insulin in blood of insulin-using diabetic patients. Diabetes 36, 838–841 (1987).
Papachroni, K.K. et al. Autoantibodies to alpha-synuclein in inherited Parkinson's disease. J. Neurochem. 101, 749–756 (2007).
Dalakas, M.C., Fujii, M., Li, M. & McElroy, B. The clinical spectrum of anti-GAD antibody-positive patients with stiff-person syndrome. Neurology 55, 1531–1535 (2000).
Derda, R. et al. Diversity of phage-displayed libraries of peptides during panning and amplification. Molecules 16, 1776–1803 (2011).
Lamesch, P. et al. hORFeome v3.1: a resource of human open reading frames representing over 10,000 human genes. Genomics 89, 307–315 (2007).
Acknowledgements
This work was supported in part by grants from the Department of Defense (W81XWH-10-1-0994 and W81XWH-04-1-0197) to S.J.E., and in part by the US National Institutes of Health (K08CA124804), The American Recovery and Reinvestment Act (3P30CA023100-25S8), Sontag Foundation Distinguished Scientist Award and a James S. McDonnell Foundation award to S.K. N.L.S. is a fellow of the Susan G. Komen for the Cure Foundation. S.J.E. is an investigator with the Howard Hughes Medical Institute. We would like to thank S. Gowrisankar, O. Iartchouk and L. Merrill for assistance with Illumina sequencing, and D. Šćepanović for statistical support.
Author information
Authors and Affiliations
Contributions
S.J.E. conceived the project, which was supervised by N.L.S. and S.J.E. Z.Z. designed the DNA sequences for synthesis. Oligo libraries were constructed by E.M.L. Cloning was performed by M.Z.L., M.A.M.G. and N.L.S. The T7-Pep, T7-NPep, and T7-CPep phage libraries were constructed by N.L.S. and characterized by N.L.S. and H.B.L. The PhIP-Seq protocol was developed and implemented by H.B.L. Clinical evaluations and patient sample acquisitions were performed by S.K. Statistical analysis of PhIP-Seq data was conceived by U.L. under the supervision of G.M.C. and implemented by H.B.L. PhIP-Seq candidates were confirmed by H.B.L. The RPA2 experiment was performed by A.C. The manuscript was prepared by H.B.L. and edited by N.L.S. and S.J.E.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Tables 1–3 and Supplementary Figs. 1–9 (PDF 5813 kb)
Rights and permissions
About this article
Cite this article
Larman, H., Zhao, Z., Laserson, U. et al. Autoantigen discovery with a synthetic human peptidome. Nat Biotechnol 29, 535–541 (2011). https://doi.org/10.1038/nbt.1856
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.1856
This article is cited by
-
Schistosoma mansoni vaccine candidates identified by unbiased phage display screening in self-cured rhesus macaques
npj Vaccines (2024)
-
Virome-wide detection of natural infection events and the associated antibody dynamics using longitudinal highly-multiplexed serology
Nature Communications (2023)
-
Human antibody profiling technologies for autoimmune disease
Immunologic Research (2023)
-
Detecting antibody reactivities in Phage ImmunoPrecipitation Sequencing data
BMC Genomics (2022)
-
PepSeq: a fully in vitro platform for highly multiplexed serology using customizable DNA-barcoded peptide libraries
Nature Protocols (2022)