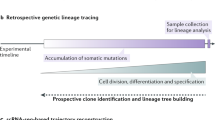
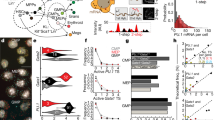
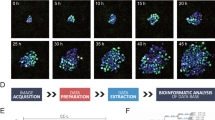
Abstract
The study of induced pluripotency often relies on experimental approaches that average measurements across a large population of cells, the majority of which do not become pluripotent. Here we used high-resolution, time-lapse imaging to trace the reprogramming process over 2 weeks from single mouse embryonic fibroblasts (MEFs) to pluripotency factor–positive colonies. This enabled us to calculate a normalized cell-of-origin reprogramming efficiency that takes into account only the initial MEFs that respond to form reprogrammed colonies rather than the larger number of final colonies. Furthermore, this retrospective analysis revealed that successfully reprogramming cells undergo a rapid shift in their proliferative rate that coincides with a reduction in cellular area. This event occurs as early as the first cell division and with similar kinetics in all cells that form induced pluripotent stem (iPS) cell colonies. These data contribute to the theoretical modeling of reprogramming and suggest that certain parts of the reprogramming process follow defined rather than stochastic steps.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
Nakagawa, M. et al. Generation of induced pluripotent stem cells without Myc from mouse and human fibroblasts. Nat. Biotechnol. 26, 101–106 (2008).
Wernig, M., Meissner, A., Cassady, J.P. & Jaenisch, R. C-Myc is dispensable for direct reprogramming of mouse fibroblasts. Cell Stem Cell 2, 10–12 (2008).
Yu, J. et al. Induced pluripotent stem cell lines derived from human somatic cells. Science 318, 1917–1920 (2007).
Feng, B. et al. Reprogramming of fibroblasts into induced pluripotent stem cells with orphan nuclear receptor Esrrb. Nat. Cell Biol. 11, 197–203 (2009).
Wernig, M. et al. In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature 448, 318–317 (2007).
Okita, K., Ichisaka, T. & Yamanaka, S. Generation of germline-competent induced pluripotent stem cells. Nature 448, 313–317 (2007).
Maherali, N. et al. Global epigenetic remodeling in directly reprogrammed fibroblasts. Cell Stem Cell 1, 55–70 (2007).
Boland, M.J. et al. Adult mice generated from induced pluripotent stem cells. Nature 461, 91–94 (2009).
Kang, L., Wang, J., Zhang, Y., Kou, Z. & Gao, S. iPS cells can support full-term development of tetraploid blastocyst-complemented embryos. Cell Stem Cell 5, 135–138 (2009).
Zhao, X.Y. et al. iPS cells produce viable mice through tetraploid complementation. Nature 461, 86–90 (2009).
Mikkelsen, T.S. et al. Dissecting direct reprogramming through integrative genomic analysis. Nature 454, 49–55 (2008).
Amabile, G. & Meissner, A. Induced pluripotent stem cells: current progress and potential for regenerative medicine. Trends Mol. Med. 15, 59–68 (2009).
Jaenisch, R. & Young, R. Stem cells, the molecular circuitry of pluripotency and nuclear reprogramming. Cell 132, 567–582 (2008).
Yamanaka, S. Elite and stochastic models for induced pluripotent stem cell generation. Nature 460, 49–52 (2009).
Hanna, J. et al. Direct cell reprogramming is a stochastic process amenable to acceleration. Nature 462, 595–601 (2009).
Wernig, M. et al. A drug-inducible transgenic system for direct reprogramming of multiple somatic cell types. Nat. Biotechnol. 26, 916–924 (2008).
Stadtfeld, M., Maherali, N., Breault, D. & Hochedlinger, K. Defining molecular cornerstones during fibroblast to iPS cell reprogramming in mouse. Cell Stem Cell 2, 230–240 (2008).
Orford, K.W. & Scadden, D.T. Deconstructing stem cell self-renewal: genetic insights into cell-cycle regulation. Nat. Rev. Genet. 9, 115–128 (2008).
Kamijo, T. et al. Tumor suppression at the mouse INK4a locus mediated by the alternative reading frame product p19ARF. Cell 91, 649–659 (1997).
Hong, H. et al. Suppression of induced pluripotent stem cell generation by the p53-p21 pathway. Nature 460, 1132–1135 (2009).
Kawamura, T. et al. Linking the p53 tumour suppressor pathway to somatic cell reprogramming. Nature 460, 1140–1144 (2009).
Li, H. et al. The Ink4/Arf locus is a barrier for iPS cell reprogramming. Nature 460, 1136–1139 (2009).
Marion, R.M. et al. A p53-mediated DNA damage response limits reprogramming to ensure iPS cell genomic integrity. Nature 460, 1149–1153 (2009).
Utikal, J. et al. Immortalization eliminates a roadblock during cellular reprogramming into iPS cells. Nature 460, 1145–1148 (2009).
Banito, A. et al. Senescence impairs successful reprogramming to pluripotent stem cells. Genes Dev. 23, 2134–2139 (2009).
Ventura, A. et al. Cre-lox-regulated conditional RNA interference from transgenes. Proc. Natl. Acad. Sci. USA 101, 10380–10385 (2004).
Brambrink, T. et al. Sequential expression of pluripotency markers during direct reprogramming of mouse somatic cells. Cell Stem Cell 2, 151–159 (2008).
Singh, A.M. & Dalton, S. The cell cycle and Myc intersect with mechanisms that regulate pluripotency and reprogramming. Cell Stem Cell 5, 141–149 (2009).
Carpenter, A.E. et al. Cell profiler: image analysis software for identifying and quantifying cell phenotypes. Genome Biol. 7, R100 (2006).
Acknowledgements
We thank A. Carpenter and M. Bray from the Broad Imaging Platform for help with the initial CellProfiler image analysis pipeline. We thank E.S. Lander and C. Bock and R.P. Koche for critical reading of the manuscript as well as M. Thomson, M. Staller, A. De Los Angeles and J. Dennett for technical assistance and intellectual input. I.N. was supported by a Merck postdoctoral fellowship and an Alon fellowship. A.R. was supported by a Career Award at the Scientific Interface from the Burroughs Wellcome Fund, an National Institutes of Health Pioneer Award and the Sloan Foundation. A.R. is an Early Career Scientist of the Howard Hughes Medical Institute and an Investigator of the Merkin Foundation for Stem Cell Research at the Broad Institute. A.M. was supported by the Pew Charitable Trust and a New Investigator grant by the Massachusetts Life Science Center (MLSC). This work was funded by the Pew and MLSC.
Author information
Authors and Affiliations
Contributions
Z.D.S., I.N., A.R. and A.M. conceived the experiments and wrote the manuscript. Z.D.S. generated all reagents and performed the experiments. Z.D.S. and I.N. performed the analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–10 (PDF 2399 kb)
Supplementary Movie 1
Time-lapse movie of a single (1/16 of the 4×4) monitored site (upper right corner of Fig. 1a,b,c and same as 1d). The movie follows the YFP labeled fibroblasts (#16 in Fig. 1b) as it divides and forms a colony over the course of 12 days. This MEF exhibits an immediate change in its proliferative behavior and size maintenance, rapidly forming many small cells that eventually give rise to multiple distinct iPS colonies within the formed cluster (shown in Figure 1d). Non responding induced MEFs are apparent in the upper right and lower left hand corner for immediate comparison to the reprogramming lineage; these cells divide more slowly and maintain large, asymmetrical mesenchymal characteristics. Time in days is shown and the arrow in the first frame points to the source cell. (MOV 2398 kb)
Supplementary Movie 2
Time-lapse movie of a global 5×5 field at 10× magnification in phase contrast and with GFP-labeled MEFs highlighted over a 14 day experiment. A single primary colony emerges at Day 4 while subsequent satellites continue to accumulate from Day 6 to the termination of the experiment at Day 14. (MOV 4536 kb)
Supplementary Movie 3
Time-lapse movie following a YFP-labeled MEF progressing to a lineage in which multiple iPS cell colonies (∼6) are formed (Fig. 2d shows selected images of this movie in grayscale). These subpopulations within the responding lineage can be clearly demarcated before unique iPS cell colonies are observed. Within the earliest time points, an additional YFP labeled responding population can be observed at the left, but these cells do not progress through the preliminary size reducing response, are not accountable for an iPS cell colony, and are lost or scored as non-proliferative by Day 6. Time in days is shown and the arrow in the first frame points to the source cell. (MOV 2760 kb)
Supplementary Movies 4a
Distinct morphologies of responding MEFs characterized from time-lapse imaging. a. Arrested/Apoptotic (A) responses in GFP, RFP, and YFP labeled MEFs. Several of these cells initially exhibit attributes of a positive response to factor induction as outlined in text but either arrest/apoptose or transition to a slower proliferative rate. (MOV 2429 kb)
Supplementary Movies 4b
Distinct morphologies of responding MEFs characterized from time-lapse imaging. b. Slow dividing (SD) YFP-labeled MEFs that do not reprogram, but slowly divide and maintain many of the morphological attributes of the original MEF population. (MOV 2353 kb)
Supplementary Movies 4c
Distinct morphologies of responding MEFs characterized from time-lapse imaging. c. Fast Dividing Fibroblast (FD) YFP labeled MEFs that demonstrate a transition to a rapid cell cycle but become more striated and grow as a mesenchymal monolayer. Note, that within this fast dividing fibroblast population, three ectopic, unlabeled satellite colonies emerge within the final 4 days of the 14 daytime series. (MOV 3332 kb)
Supplementary Movies 4d
Distinct morphologies of responding MEFs characterized from time-lapse imaging. d. iPS Forming (iPS) A RFP labeled MEF exhibiting an immediate change in proliferative rate and cell size, forming small clusters of dividing cells that lead to the formation of compacted subpopulations which give rise to pluripotency marker positive iPS cells. (MOV 2273 kb)
Supplementary Movie 5
Time-lapse movie of a MEF infected with an EGFP labeled p53 targeting shRNA (Upper Panels) compared to an internal RFP labeled control MEF (Lower Panels) within the same experiment (Fig. 4b shows selected images from these movies). Size and proliferative rate are nearly identical within the first four days, but only the RFP labeled MEF progeny continue along an expected trajectory to a compact iPS colony whereas the p53 depleted MEF slows its proliferative rate and forms a colony that is less compact compared to the wild type control. Note that this p53 KD derived colony remains negative for classical pluripotency markers at the final time point (14 days). Time in days is shown and the arrow in the first frame points to the source cell. (MOV 5070 kb)
Supplementary Movie 6
iPS cell colonies exhibit conserved formation dynamics. Shown are 12 independent iPS cell forming lineages imaged from the time of factor induction to termination after 14 days. As shown in Figure 5, each population is governed by near identical kinetics and concludes in a tightly distributed time of primary colony emergence that is unlikely to be dictated by a single low probability stochastic event. (MOV 3141 kb)
Supplementary Movie 7
Representative population wide overlays of RFP and YFP labeled inducible MEFs over a 12 day experiment and from which the YFP labeled fibroblast labeled #16 in Figure 1 was identified as a cell of origin for a primary iPS cell colony. Similar overlays were generated for every experiment and used to both identify primary colonies for downstream analysis and to gauge global reprogramming trends. (MOV 6515 kb)
Rights and permissions
About this article
Cite this article
Smith, Z., Nachman, I., Regev, A. et al. Dynamic single-cell imaging of direct reprogramming reveals an early specifying event. Nat Biotechnol 28, 521–526 (2010). https://doi.org/10.1038/nbt.1632
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.1632
This article is cited by
-
Functions of p53 in pluripotent stem cells
Protein & Cell (2020)
-
Tumorigenic and Immunogenic Properties of Induced Pluripotent Stem Cells: a Promising Cancer Vaccine
Stem Cell Reviews and Reports (2020)
-
Validation of Common Housekeeping Genes as Reference for qPCR Gene Expression Analysis During iPS Reprogramming Process
Scientific Reports (2018)
-
Single-cell transcriptional profiling: a window into embryonic cell-type specification
Nature Reviews Molecular Cell Biology (2018)
-
Ribosome Incorporation into Somatic Cells Promotes Lineage Transdifferentiation towards Multipotency
Scientific Reports (2018)