Abstract
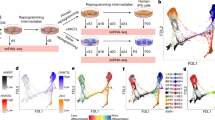
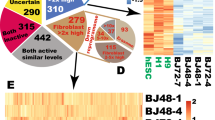
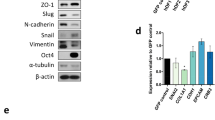
Somatic cells can be reprogrammed into induced pluripotent stem (iPS) cells by enforced expression of transcription factors. Using serial live imaging of human fibroblasts undergoing reprogramming, we identified distinct colony types that morphologically resemble embryonic stem (ES) cells yet differ in molecular phenotype and differentiation potential. By analyzing expression of pluripotency markers, methylation at the OCT4 and NANOG promoters and differentiation into teratomas, we determined that only one colony type represents true iPS cells, whereas the others represent reprogramming intermediates. Proviral silencing and expression of TRA-1-60, DNMT3B and REX1 can be used to distinguish the fully reprogrammed state, whereas alkaline phosphatase, SSEA-4, GDF3, hTERT and NANOG are insufficient as markers. We also show that reprogramming using chemically defined medium favors formation of fully reprogrammed over partially reprogrammed colonies. Our data define molecular markers of the fully reprogrammed state and highlight the need for rigorous characterization and standardization of putative iPS cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Dimos, J.T. et al. Induced pluripotent stem cells generated from patients with ALS can be differentiated into motor neurons. Science 321, 1218–1221 (2008).
Park, I.H. et al. Disease-specific induced pluripotent stem cells. Cell 134, 877–886 (2008).
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
Okita, K., Ichisaka, T. & Yamanaka, S. Generation of germline-competent induced pluripotent stem cells. Nature 448, 313–317 (2007).
Wernig, M. et al. In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature 448, 318–324 (2007).
Maherali, N. et al. Directly reprogrammed fibroblasts show global epigenetic remodeling and widespread tissue contribution. Cell Stem Cell 1, 55–70 (2007).
Park, I.H. et al. Reprogramming of human somatic cells to pluripotency with defined factors. Nature 451, 141–146 (2008).
Maherali, N. et al. A high-efficiency system for the generation and study of human induced pluripotent stem cells. Cell Stem Cell 3, 340–345 (2008).
Hockemeyer, D. et al. A drug-inducible system for direct reprogramming of human somatic cells to pluripotency. Cell Stem Cell 3, 346–353 (2008).
Park, I.H., Lerou, P.H., Zhao, R., Huo, H. & Daley, G.Q. Generation of human-induced pluripotent stem cells. Nat. Protoc. 3, 1180–1186 (2008).
Lowry, W.E. et al. Generation of human induced pluripotent stem cells from dermal fibroblasts. Proc. Natl. Acad. Sci. USA 105, 2883–2888 (2008).
Apati, A. et al. High level functional expression of the ABCG2 multidrug transporter in undifferentiated human embryonic stem cells. Biochim. Biophys. Acta 1778, 2700–2709 (2008).
Brambrink, T. et al. Sequential expression of pluripotency markers during direct reprogramming of mouse somatic cells. Cell Stem Cell 2, 151–159 (2008).
Stadtfeld, M., Maherali, N., Breault, D.T. & Hochedlinger, K. Defining molecular cornerstones during fibroblast to iPS cell reprogramming in mouse. Cell Stem Cell 2, 230–240 (2008).
Takahashi, K. et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 (2007).
Yu, J. et al. Induced pluripotent stem cell lines derived from human somatic cells. Science 318, 1917–1920 (2007).
Okita, K., Nakagawa, M., Hyenjong, H., Ichisaka, T. & Yamanaka, S. Generation of mouse induced pluripotent stem cells without viral vectors. Science 322, 949–953 (2008).
Yu, J. et al. Human induced pluripotent stem cells free of vector and transgene sequences. Science 324, 797–801 (2009).
Maherali, N. & Hochedlinger, K. Guidelines and techniques for the generation of induced pluripotent stem cells. Cell Stem Cell 3, 595–605 (2008).
Daley, G.Q. et al. Broader implications of defining standards for the pluripotency of iPSCs. Cell Stem Cell 4, 200–201, author reply 202 (2009).
Ellis, J. et al. Alternative induced pluripotent stem cell characterization criteria for in vitro applications. Cell Stem Cell 4, 198–199, author reply 202 (2009).
Feng, B. et al. Reprogramming of fibroblasts into induced pluripotent stem cells with orphan nuclear receptor Esrrb. Nat. Cell Biol. 11, 197–203 (2009).
Acknowledgements
G.Q.D. was supported by grants from the National Institutes of Health (NIH), the NIH Director's Pioneer Award of the NIH Roadmap for Medical Research, and private funds contributed to the Harvard Stem Cell Institute and the Children's Hospital Stem Cell Program. G.Q.D. is a recipient of Clinical Scientist Awards in Translational Research from the Burroughs Wellcome Fund and the Leukemia and Lymphoma Society, and is an investigator of the Howard Hughes Medical Institute. T.M.S. was supported by the Harvard Stem Cell Institute and the Children's Hospital Stem Cell Program. Y.-H.L. is funded by the Agency of Science, Technology and Research International Fellowship and the Institute of Medical Biology, Singapore. E.M.C. was funded by The Canadian Institutes of Health Research. T.A.I. was supported by Breast Cancer Research Foundation, NY, and Department of Defense, Congressionally Directed Medical Research Programs (CDMRPs) and Breast Cancer Research Program (BCRP). We thank T. Woo (BWH) for her assistance with immnuhistopathological stains of the teratoma sections.
Author information
Authors and Affiliations
Contributions
Co-direction of the project: G.Q.D. and T.M.S. Study concept and design: T.M.S., G.Q.D., E.M.C., S.R. and I.-H.P. Live imaging reprogramming studies: T.M.S., E.M.C., S.R. and P.D.M. Isolation of cell lines: E.M.C. and S.R. Characterization of cell lines: E.M.C., S.R., I.H.P., Y.H.-L., H.H. and J.R. Human iPS cell reprogramming comparison on chemically defined conditions on Matrigel versus KOSR media on feeders: S.R., J.D.M. and O.H. Teratoma interpretation and characterization: T.A.I.
Corresponding authors
Ethics declarations
Competing interests
G.Q.D. is founder of iPierian, a biotechnology company that is using iPS cells in drug discovery in neurodegenerative diseases.
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–2 (ZIP 8425 kb)
Supplementary Text and Figures
Supplementary Figs. 3–4 (ZIP 6840 kb)
Supplementary Text and Figures
Supplementary Figs. 5–7 (ZIP 24781 kb)
Supplementary Text and Figures
Supplementary Figs. 8–11 (ZIP 22387 kb)
Supplementary Text and Figures
Supplementary Tables 1–4 (ZIP 924 kb)
Rights and permissions
About this article
Cite this article
Chan, E., Ratanasirintrawoot, S., Park, IH. et al. Live cell imaging distinguishes bona fide human iPS cells from partially reprogrammed cells. Nat Biotechnol 27, 1033–1037 (2009). https://doi.org/10.1038/nbt.1580
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.1580
This article is cited by
-
Genome-wide ATAC-see screening identifies TFDP1 as a modulator of global chromatin accessibility
Nature Genetics (2024)
-
AF10 (MLLT10) prevents somatic cell reprogramming through regulation of DOT1L-mediated H3K79 methylation
Epigenetics & Chromatin (2021)
-
Novel live cell fluorescent probe for human-induced pluripotent stem cells highlights early reprogramming population
Stem Cell Research & Therapy (2021)
-
Loss of the fragile X syndrome protein FMRP results in misregulation of nonsense-mediated mRNA decay
Nature Cell Biology (2021)
-
Mental health dished up—the use of iPSC models in neuropsychiatric research
Journal of Neural Transmission (2020)