Abstract
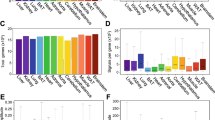
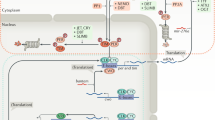
Many mammalian peripheral tissues have circadian clocks1,2,3,4; endogenous oscillators that generate transcriptional rhythms thought to be important for the daily timing of physiological processes5,6. The extent of circadian gene regulation in peripheral tissues is unclear, and to what degree circadian regulation in different tissues involves common or specialized pathways is unknown. Here we report a comparative analysis of circadian gene expression in vivo in mouse liver and heart using oligonucleotide arrays representing 12,488 genes. We find that peripheral circadian gene regulation is extensive (≥8–10% of the genes expressed in each tissue), that the distributions of circadian phases in the two tissues are markedly different, and that very few genes show circadian regulation in both tissues. This specificity of circadian regulation cannot be accounted for by tissue-specific gene expression. Despite this divergence, the clock-regulated genes in liver and heart participate in overlapping, extremely diverse processes. A core set of 37 genes with similar circadian regulation in both tissues includes candidates for new clock genes and output genes, and it contains genes responsive to circulating factors with circadian or diurnal rhythms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Balsalobre, A., Damiola, F. & Schibler, U. A serum shock induces circadian gene expression in mammalian tissue culture cells. Cell 93, 929–937 (1998)
Yamazaki, S. et al. Resetting central and peripheral circadian oscillators in transgenic rats. Science 288, 682–685 (2000)
Balsalobre, A. et al. Resetting of circadian time in peripheral tissues by glucocorticoid signaling. Science 289, 2344–2347 (2000)
McNamara, P. et al. Regulation of CLOCK and MOP4 by nuclear hormone receptors in the vasculature: a humoral mechanism to reset a peripheral clock. Cell 105, 877–889 (2001)
Reppert, S. M. & Weaver, D. R. Molecular analysis of mammalian circadian rhythms. Annu. Rev. Physiol. 63, 647–676 (2001)
Ripperger, J. A. & Schibler, U. Circadian regulation of gene expression in animals. Curr. Opin. Cell Biol. 13, 357–362 (2001)
Lockhart, D. J. et al. Expression monitoring by hybridization to high-density oligonucleotide arrays. Nature Biotechnol. 14, 1675–1680 (1996)
Li, C. & Wong, W. H. Model-based analysis of oligonucleotide arrays: expression index computation and outlier detection. Proc. Natl Acad. Sci. USA 98, 31–36 (2001)
Claverie, J.-M. Gene number: what if there are only 30,000 human genes? Science 291, 1255–1257 (2001)
Murphy, W. J., Stanyon, R. & O'Brien, S. J. Evolution of mammalian genome organization inferred from comparative gene mapping. Genome Biol. (Review) 2, 0005.1–0005.8 (2001)
Kornmann, B., Preitner, N., Rifat, D., Fleury-Olela, F. & Schibler, U. Analysis of circadian liver gene expression by ADDER, a highly sensitive method for the display of differentially expressed mRNAs. Nucleic Acids Res. 29, E51 (2001)
Velculescu, V. E. et al. Analysis of human transcriptomes. Nature Genet. 23, 387–388 (1999)
Klein, D. C., Moore, R. Y. & Reppert, S. M. (eds) Suprachiasmatic Nucleus: The Mind's Clock (Oxford Univ. Press, New York, 1991)
Grundschober, C. et al. Circadian regulation of diverse gene products revealed by mRNA expression profiling of synchronized fibroblasts. J. Biol. Chem. 276, 46751–46758 (2001)
Ashburner, M. et al. Gene ontology: tool for the unification of biology. The Gene Ontology Consortium. Nature Genet. 25, 25–29 (2000)
Torra, I. P. et al. Circadian and glucocorticoid regulation of Rev-erb-alpha expression in liver. Endocrinology 141, 3799–3806 (2000)
Ripperger, J. A., Shearman, L. P., Reppert, S. M. & Schibler, U. CLOCK, an essential pacemaker component, controls expression of the circadian transcription factor DBP. Genes Dev. 14, 679–689 (2000)
Young, M. W. & Kay, S. A. Time zones: a comparative genetics of circadian clocks. Nature Rev. Genet. 2, 702–715 (2001)
Tata, J. R., Baker, B. S., Machuca, I., Rabelo, E. M. & Yamauchi, K. Autoinduction of nuclear receptor genes and its significance. J. Steroid Biochem. Mol. Biol. 46, 105–119 (1993)
Kalsbeek, A., Fliers, E., Franke, A. N., Worte, J. & Buijs, R. M. Functional connections between the suprachiasmatic nucleus and the thyroid gland as revealed by lesioning and viral tracing techniques in the rat. Endocrinology 141, 3832–3841 (2000)
Harris, H. J., Kotelevtsev, Y., Mullins, J. J., Seckl, J. R. & Holmes, M. C. Intracellular regeneration of glucocorticoids by 11beta-hydroxysteroid dehydrogenase (11beta-HSD)-1 plays a key role in regulation of the hypothalamic-pituitary-adrenal axis: analysis of 11beta-HSD-1-deficient mice. Endocrinology 142, 114–120 (2001)
Chen, J., Maltby, K. M. & Miano, J. M. A novel retinoid-response gene set in vascular smooth muscle cells. Biochem. Biophys. Res. Commun. 281, 475–482 (2001)
Wolf, F. W. et al. B94, a primary response gene inducible by tumour necrosis factor-alpha, is expressed in developing hematopoietic tissues and the sperm acrosome. J. Biol. Chem. 269, 3633–3640 (1994)
Cooper, P., Potter, S., Mueck, B., Yousefi, S. & Jarai, G. Identification of genes induced by inflammatory cytokines in airway epithelium. Am. J. Physiol. Lung Cell. Mol. Physiol. 280, L841–L852 (2001)
Buchan, P. et al. Repeated topical administration of all-trans-retinoic acid and plasma levels of retinoic acids in humans. J. Am. Acad. Dermatol. 30, 428–434 (1994)
Petrovsky, N. & Harrison, L. C. The chronobiology of human cytokine production. Int. Rev. Immunol. 16, 635–649 (1998)
Harmer, S. L. et al. Orchestrated transcription of key pathways in Arabidopsis by the circadian clock. Science 290, 2110–2113 (2000)
Claridge-Chang, A. et al. Circadian regulation of gene expression systems in the Drosophila head. Neuron 32, 657–671 (2001)
McDonald, M. J. & Rosbash, M. Microarray analysis and organization of circadian gene expression in Drosophila. Cell 107, 567–578 (2001)
Panda, S. J. et al. Coordinated transcription of key pathways in the mouse by the circadian clock. Cell [online 2 April 2001] 10.1016/S0092867402007225 (2002)
Acknowledgements
This work was supported by the Edward R. and Anne G. Lefler Center, Harvard Medical School and the National Eye Institute (C.J.W.), a Deutsche Forschungsgemeinschaft Postdoctoral Fellowship (K-F.S.), the National Institute of Child Health and Human Development (F.C.D. and N.V.), and the National Human Genome Research Institute (W.H.W.). We thank S. Meng and the Harvard Center for Genomics Research for technical assistance and M.-C. Kao for programming.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Storch, KF., Lipan, O., Leykin, I. et al. Extensive and divergent circadian gene expression in liver and heart. Nature 417, 78–83 (2002). https://doi.org/10.1038/nature744
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature744
This article is cited by
-
Circadian rhythm in cardiovascular diseases: a bibliometric analysis of the past, present, and future
European Journal of Medical Research (2023)
-
Astrocytic insulin receptor controls circadian behavior via dopamine signaling in a sexually dimorphic manner
Nature Communications (2023)
-
Application and limitation of a biological clock-based method for estimating time of death in forensic practices
Scientific Reports (2023)
-
Sexually dimorphic role of circadian clock genes in alcohol drinking behavior
Psychopharmacology (2023)
-
Aging disrupts circadian gene regulation and function in macrophages
Nature Immunology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.