Abstract
Metal halide perovskites are of great interest for various high-performance optoelectronic applications1. The ability to tune the perovskite bandgap continuously by modifying the chemical composition opens up applications for perovskites as coloured emitters, in building-integrated photovoltaics, and as components of tandem photovoltaics to increase the power conversion efficiency2,3,4. Nevertheless, performance is limited by non-radiative losses, with luminescence yields in state-of-the-art perovskite solar cells still far from 100 per cent under standard solar illumination conditions5,6,7. Furthermore, in mixed halide perovskite systems designed for continuous bandgap tunability2 (bandgaps of approximately 1.7 to 1.9 electronvolts), photoinduced ion segregation leads to bandgap instabilities8,9. Here we demonstrate substantial mitigation of both non-radiative losses and photoinduced ion migration in perovskite films and interfaces by decorating the surfaces and grain boundaries with passivating potassium halide layers. We demonstrate external photoluminescence quantum yields of 66 per cent, which translate to internal yields that exceed 95 per cent. The high luminescence yields are achieved while maintaining high mobilities of more than 40 square centimetres per volt per second, providing the elusive combination of both high luminescence and excellent charge transport10. When interfaced with electrodes in a solar cell device stack, the external luminescence yield—a quantity that must be maximized to obtain high efficiency—remains as high as 15 per cent, indicating very clean interfaces. We also demonstrate the inhibition of transient photoinduced ion-migration processes across a wide range of mixed halide perovskite bandgaps in materials that exhibit bandgap instabilities when unpassivated. We validate these results in fully operating solar cells. Our work represents an important advance in the construction of tunable metal halide perovskite films and interfaces that can approach the efficiency limits in tandem solar cells, coloured-light-emitting diodes and other optoelectronic applications.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others

References
Stranks, S. D. & Snaith, H. J. Metal-halide perovskites for photovoltaic and light-emitting devices. Nat. Nanotechnol. 10, 391–402 (2015)
Eperon, G. E. et al. Perovskite–perovskite tandem photovoltaics with optimized band gaps. Science 354, 861–865 (2016)
McMeekin, D. P. et al. A mixed-cation lead mixed-halide perovskite absorber for tandem solar cells. Science 351, 151–155 (2016)
Bush, K. A. et al. 23.6%-efficient monolithic perovskite/silicon tandem solar cells with improved stability. Nat. Energy 2, 17009 (2017)
Saliba, M. et al. Incorporation of rubidium cations into perovskite solar cells improves photovoltaic performance. Science 354, 206–209 (2016)
Momblona, C. et al. Efficient vacuum deposited p-i-n and n-i-p perovskite solar cells employing doped charge transport layers. Energy Environ. Sci. 9, 3456–3463 (2016)
Stranks, S. D. Nonradiative losses in metal halide perovskites. ACS Energy Lett. 2, 1515–1525 (2017)
Hoke, E. T. et al. Reversible photo-induced trap formation in mixed-halide hybrid perovskites for photovoltaics. Chem. Sci. 6, 613–617 (2015)
Barker, A. J. et al. Defect-assisted photoinduced halide segregation in mixed-halide perovskite thin films. ACS Energy Lett. 2, 1416–1424 (2017)
Stoddard, R. J., Eickemeyer, F. T., Katahara, J. K. & Hillhouse, H. W. Correlation between photoluminescence and carrier transport and a simple in situ passivation method for high-bandgap hybrid perovskites. J. Phys. Chem. Lett. 8, 3289–3298 (2017)
Saliba, M. et al. Cesium-containing triple cation perovskite solar cells: improved stability, reproducibility and high efficiency. Energy Environ. Sci. 9, 1989–1997 (2016)
Miller, O. D., Yablonovitch, E. & Kurtz, S. R. Strong internal and external luminescence as solar cells approach the Shockley–Queisser limit. IEEE J. Photovoltaics 2, 303–311 (2012)
Stranks, S. D. et al. Recombination kinetics in organic–inorganic perovskites: excitons, free charge, and subgap states. Phys. Rev. Appl. 2, 034007 (2014)
Kim, J., Lee, S. H., Lee, J. H. & Hong, K. H. The role of intrinsic defects in methylammonium lead iodide perovskite. J. Phys. Chem. Lett. 5, 1312–1317 (2014)
Agiorgousis, M. L., Sun, Y.-Y., Zeng, H. & Zhang, S. Strong covalency-induced recombination centers in perovskite solar cell material CH3NH3PbI3 . J. Am. Chem. Soc. 136, 14570–14575 (2014)
Richter, J. M. et al. Enhancing photoluminescence yields in lead halide perovskites by photon recycling and light out-coupling. Nat. Commun. 7, 13941 (2016)
Deschler, F. et al. High photoluminescence efficiency and optically pumped lasing in solution-processed mixed halide perovskite semiconductors. J. Phys. Chem. Lett. 5, 1421–1426 (2014)
Hutter, E. M. et al. Direct–indirect character of the bandgap in methylammonium lead iodide perovskite. Nat. Mater. 16, 115–120 (2017)
Reid, O. G., Yang, M., Kopidakis, N., Zhu, K. & Rumbles, G. Grain-size-limited mobility in methylammonium lead iodide perovskite thin films. ACS Energy Lett. 1, 561–565 (2016)
deQuilettes, D. W. et al. Photo-induced halide redistribution in organic–inorganic perovskite films. Nat. Commun. 7, 11683 (2016)
Pauca, V. P., Piper, J. & Plemmons, R. J. Nonnegative matrix factorization for spectral data analysis. Linear Algebra Appl. 416, 29–47 (2006)
Abdi-Jalebi, M. et al. Impact of monovalent cation halide additives on the structural and optoelectronic properties of CH3NH3PbI3 perovskite. Adv. Energy Mater. 6, 1502472 (2016)
Tang, Z. et al. Hysteresis-free perovskite solar cells made of potassium-doped organometal halide perovskite. Sci. Rep. 7, 12183 (2017)
Zhao, P. et al. Improved carriers injection capacity in perovskite solar cells by introducing A-site interstitial defects. J. Mater. Chem. A 5, 7905–7911 (2017)
Nam, J. K. et al. Potassium incorporation for enhanced performance and stability of fully inorganic cesium lead halide perovskite solar cells. Nano Lett. 17, 2028–2033 (2017)
Kubicki, D. J. et al. Phase segregation in Cs-, Rb- and K-doped mixed-cation (MA)x(FA)1−xPbI3 hybrid perovskites from solid-state NMR. J. Am. Chem. Soc. 139, 14173–14180 (2017)
Ibrahim Dar, M. et al. Understanding the impact of bromide on the photovoltaic performance of CH3NH3PbI3 solar cells. Adv. Mater. 27, 7221–7228 (2015)
Sutter-Fella, C. M. et al. High photoluminescence quantum yield in band gap tunable bromide containing mixed halide perovskites. Nano Lett. 16, 800–806 (2016)
Stranks, S. D. et al. Electron–hole diffusion lengths exceeding 1 micrometer in an organometal trihalide perovskite absorber. Science 342, 341–344 (2013)
Yablonovitch, E. & Miller, O. D. The opto-electronics which broke the efficiency record in solar cells. In Proc. Conference on Lasers and Electro-Optics 2012 CF2J.1 (Optical Society of America, 2012)
Pazos-Outón, L. M. et al. Photon recycling in lead iodide perovskite solar cells. Science 351, 1430–1433 (2016)
Abdi-Jalebi, M. et al. Impact of a mesoporous titania–perovskite interface on the performance of hybrid organic–inorganic perovskite solar cells. J. Phys. Chem. Lett. 7, 3264–3269 (2016)
de Mello, J. C., Wittman, H. F. & Friend, R. H. An improved experimental determination of external photoluminescence quantum efficiency. Adv. Mater. 9, 230–232 (1997)
Park, B. et al. Chemical engineering of methylammonium lead iodide/bromide perovskites: tuning of opto-electronic properties and photovoltaic performance. J. Mater. Chem. A 3, 21760–21771 (2015)
Hutter, E. M., Eperon, G. E., Stranks, S. D. & Savenije, T. J. Charge carriers in planar and meso-structured organic–inorganic perovskites: mobilities, lifetimes, and concentrations of trap states. J. Phys. Chem. Lett. 6, 3082–3090 (2015)
Philippe, B. et al. Chemical distribution of multiple cation (Rb+, Cs+, MA+, and FA+) perovskite materials by photoelectron spectroscopy. Chem. Mater. 29, 3589–3596 (2017)
Acknowledgements
M.A.-J. thanks Nava Technology Limited and Nyak Technology Limited for their funding and technical support. Z.A.-G. acknowledges funding from a Winton Studentship, and ICON Studentship from the Lloyd’s Register Foundation. This project has received funding from the European Union’s Seventh Framework Programme (FP7/2007-2013) under REA grant agreement number PIOF-GA-2013-622630, the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation programme (grant agreement number 756962), and the Royal Society and Tata Group (UF150033). We thank the Engineering and Physical Sciences Research Council (EPSRC) for support. XMaS is a mid-range facility at the European Synchrotron Radiation Facility supported by the EPSRC and we are grateful to the XMaS beamline team staff for their support. We thank Diamond Light Source for access to beamline I09 and staff member T.-L. Lee as well as U. Cappel for assistance during the HAXPES measurements. S.C., C.D. and G.D. acknowledge funding from the ERC under grant number 25961976 PHOTO EM and financial support from the European Union under grant number 77 312483 ESTEEM2. M.A. thanks the president of the UAE’s Distinguished Student Scholarship Program, granted by the Ministry of Presidential Affairs. H.R. and B.P. acknowledge support from the Swedish research council (2014-6019) and the Swedish foundation for strategic research. E.M.H. and T.J.S. were supported by the Netherlands Organization for Scientific Research under the Echo grant number 712.014.007.
Author information
Authors and Affiliations
Contributions
M.A.-J. and S.D.S. conceived and planned the experiments with additional input from R.H.F. M.A.-J. fabricated all samples and devices and performed and analysed the PLQE experiments, the photoluminescence stability experiments, the time-resolved photoluminescence, absorption and photothermal deflection spectroscopies, the SEM and the device characterisation measurements. M.A.-J., Z.A.-G. and C.S. obtained and analysed the confocal photoluminescence maps. S.C., G.D. and C.D. performed and analysed the STEM–EDX measurements. M.A. and S.L. performed the GIWAXS experiments and analysed the data. E.M.H. and T.J.S. performed the TRMC measurements and analysed the data. M.A.-J., B.P. and H.R. performed and analysed HAXPES measurements. M.A.-J. and E.P.B. performed X-ray diffraction measurements and J.M.R. calculated the internal PLQE and assisted with time-resolved photoluminescence measurement and analysis. M.A.-J. and A.J.P. carried out the device stability tests. M.A.-J. and S.D.S. took the lead in drafting the manuscript and compiled the figures. All authors discussed the results and provided feedback on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Figure 1 Stoichiometries of the precursor solutions, and thickness measurements and scanning electron microscopy images of the (Cs,FA,MA)Pb(I0.85Br0.15)3 films.
a, Calculated values of [K]/([K]+[A]) as a function of potassium iodide volume ratio added to the (Cs,FA,MA)Pb(I0.85Br0.15)3 perovskite precursor solution. b, The halide to lead ratio ([X]/[Pb]) for different fractions of potassium (x) in perovskite films based on stoichiometric calculations. c, The thickness of the films deposited on compact-TiO2/thin-mesoporous TiO2 electrodes for different fractions of potassium. d, The calculated ratios between different elements in the (Cs,FA,MA)Pb(I0.85Br0.15)3 films at different fractions of potassium (x); note that X represents halide (that is, the sum of iodide and bromide). e–j, Scanning electron micrographs of (Cs,FA,MA)Pb(I0.85Br0.15)3 perovskite thin films, with potassium fractions (x) ranging from 0.0 to 0.6. The coloured insets show images at higher magnification.
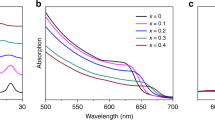
Extended Data Figure 2 Absorption, photoluminescence and X-ray diffraction characteristics of the (Cs,FA,MA)Pb(I0.85Br0.15)3 (bromide-containing) and (Cs,FA,MA)PbI3 (pure-iodide) films.
a, b, Normalized UV-vis absorption (a) and photoluminescence (PL, 532-nm continuous-wave excitation) (b) of the (Cs,FA,MA)(I0.85Br0.15)3 perovskite films with different fractions of potassium (x). The inset of b is a schematic of the change in energy level of the conduction band with increasing values of x. CB, conduction band; VB, valence band. c, The absorption spectra of (Cs,FA,MA)Pb(I0.85Br0.15)3 perovskite thin films with different potassium fractions measured by photothermal deflection spectroscopy, showing a decreased sub-gap density of states with potassium passivation. d, X-ray diffraction spectra of the (Cs,FA,MA)Pb(I0.85Br0.15)3 thin films, with new peaks arising from increasing potassium labelled with an asterisk. e, Calculated lattice parameters using a Le Bail analysis on the X-ray diffraction data. f, PLQE as a function of excitation power measured by a 532-nm continuous-wave laser for (Cs,FA,MA)Pb(I0.85Br0.15)3 perovskite thin films in an ambient atmosphere. g–i, Normalized UV-vis absorption (g), photoluminescence (532-nm continuous-wave excitation) (h) and photothermal deflection (i) spectra of the (Cs,FA,MA)PbI3 perovskite films with different potassium content. j, Calculated lattice parameters, determined using a Le Bail analysis on X-ray diffraction data, for the (Cs,FA,MA)PbI3 perovskite thin films (black squares) compared to (Cs,FA,MA)(I0.85Br0.15)3 (red circles). k, PLQE of different passivated perovskite thin films with and without bromide measured under illumination with a 532-nm laser at an excitation intensity equivalent to approximately 1 sun (60 mW cm−2) in ambient air.
Extended Data Figure 3 Confocal photoluminescence maps of passivated (Cs,FA,MA)Pb(I0.85Br0.15)3 films.
a–d, Confocal photoluminescence intensity maps with 405-nm excitation measured in ambient atmosphere for (Cs,FA,MA)Pb(I0.85Br0.15)3 perovskite thin films with x = 0.0 (a), x = 0.10 (b), x = 0.20 (c) and x = 0.40 (d). e–h, Centre-of-mass photoluminescence wavelength of the films for x = 0.0 (e), x = 0.10 (f), x = 0.20 (g) and x = 0.40 (h). i, j, Histograms of the absolute photoluminescence intensities (i) and photoluminescence wavelength (j) extracted from the respective maps for x = 0–0.40. k, Correlation between the local photoluminescence intensity and mean wavelength for x = 0.20.
Extended Data Figure 4 Time-resolved photoluminescence measurements and photostability of passivated (Cs,FA,MA)Pb(I1−yBry)3 films.
a–d, Intensity dependent time-resolved photoluminescence decays of the (Cs,MA,FA)Pb(I0.85Br0.15)3 perovskite films with different fractions of potassium. The pulse fluences of the 407-nm excitation are quoted on the graphs. e–j, Photoluminescence from (Cs,FA,MA)Pb(I1−yBry)3 films with (e, f) y = 0.4 passivated (x = 0.4, e) and unpassivated (x = 0, f), (g, h) y = 0.8 passivated (x = 0.4, g) and unpassivated (x = 0, h), (i, j) y = 1 passivated (x = 0.4, i) and unpassivated (x = 0, j). The samples were illuminated and the photoluminescence acquired continuously with a 532-nm laser at an excitation intensity equivalent to approximately 1 sun (60 mW cm−2) in an ambient atmosphere. k, Centre of mass for the photoluminescence wavelength of the passivated perovskite films (x = 0.4).
Extended Data Figure 5 Time-resolved microwave conductivity measurements on passivated (Cs,FA,MA)Pb(I0.85Br0.15)3 films.
a–e, Time-resolved microwave conductivity measurements for (Cs,FA,MA)Pb(I0.85Br0.15)3 perovskite films with different fractions of potassium (x = 0–0.4) showing the change in photoconductance after pulsed excitation at 600 nm, with excitation densities (photons per cm2) as quoted on the graphs. f, Half-life values extracted from the decays, with the excitation density quoted after accounting for the absorbed fraction and film thickness of each sample.
Extended Data Figure 6 STEM–EDX chemical maps of the passivated and unpassivated (Cs,FA,MA)Pb(I0.85Br0.15)3 samples.
a, The HAADF image and corresponding STEM–EDX quantitative maps for iodine, lead, potassium and bromide in a (Cs,FA,MA)Pb(I0.85Br0.15)3 perovskite specimen with a potassium fraction (x) of 0.20. b, STEM-HAADF cross-sectional image of an unpassivated (Cs,FA,MA)Pb(I0.85Br0.15)3 perovskite thin film (x = 0). c, d, NMF decomposition results in factor 1 associated with the perovskite layer (c) and in factor 2 indicating the presence of a lead and bromide-rich phase (d). e, f, STEM–EDX profiles of factor 1 (e) and factor 2 (f).
Extended Data Figure 7 GIWAXS measurements of passivated (Cs,FA,MA)Pb(I0.85Br0.15)3 films.
a–e, The diffraction patterns of thin (Cs,FA,MA)Pb(I0.85Br0.15)3 films collected at low angle using GIWAXS for x = 0.00 (a), x = 0.05 (b), x = 0.10 (c), x = 0.20 (d) and x = 0.40 (e). f, g, The high-resolution line profiles azimuthally integrated over the entire GIWAX profile for different fractions of potassium at 0.5 ≤ q ≤ 0.8 (f) and 0.8 ≤ q ≤ 1.1 (g).
Extended Data Figure 8 HAXPES spectra for passivated (Cs,FA,MA)Pb(I0.85Br0.15)3 perovskite thin films.
a–c, HAXPES spectra for (Cs,FA,MA)Pb(I0.85Br0.15)3 perovskite thin films (0 ≤ x ≤ 0.20) over a binding-energy range of 0 to 85 eV, recorded with a photon energy of 758 eV (a), 2,200 eV (b) and 6,600 eV (c). d, Intensity ratio between core levels ([Cs]/[Pb] and [K]/[Pb]) calculated from the experimental results as a function of photon energy (measurements at 758, 2,200 and 6,600 eV). e, f, Intensity ratios between different core levels of the perovskite thin films with different fractions of potassium (0.0 ≤ x ≤ 0.20), calculated from experimental results: I/Pb (e) and Br/I (f). We used Pb 5d, K 2p, Cs 4d, I 4d and Br 3d core levels for all energies, with the exception of 6,600 eV, for which K 1s was used.
Extended Data Figure 9 Excitation-dependent photoluminescence quantum efficiency and time-resolved photoluminescence measurements of (Cs,FA,MA)Pb(I0.85Br0.15)3 device stacks.
a–c, PLQE of reference (x = 0) and passivated (x = 0.1) (Cs,FA,MA)Pb(I0.85Br0.15)3 perovskite thin films with n-type contact (a), p-type contact (b) and both contacts (c), each measured under illumination with a 532-nm laser at different excitation intensities. d–f, Time-resolved photoluminescence decays of encapsulated (Cs,FA,MA)Pb(I0.85Br0.15)3 films (x = 0 and x = 0.1) with excitation at 400 nm and pulse fluence of 0.17 μJ cm−2 (5 × 1015 cm−3, equivalent to around 3 sun) when the perovskite is interfaced with an n-type electron-collecting electrode (compact-TiO2/thin-mesoporous TiO2) (d), a p-type hole-collecting electrode (spiro-OMeTAD) (e), and both electrodes in a full device stack (f).
Extended Data Figure 10 Current–voltage curves of passivated (Cs,FA,MA)Pb(I0.85Br0.15)3 devices and tabulated results for the best-performing (Cs,FA,MA)Pb(I0.85Br0.15)3 and (Cs,FA,MA)Pb(I0.4Br0.6)3 devices.
a–d, Forward (open symbols) and reverse (closed symbols) J–V curves of the best-performing solar cells with (Cs,FA,MA)Pb(I0.85Br0.15)3 absorbers with x = 0.00 (a), x = 0.05 (b), x = 0.10 (c) and x = 0.20 (d) measured under full simulated solar illumination conditions (AM1.5, 100 mW cm−2) with a scan rate of 15 mV s−1. The corresponding dark J–V curves are also shown. e, f, Preliminary stability tests of (Cs,FA,MA)Pb(I0.85Br0.15)3 perovskite devices. Shelf-life of devices for x = 0.00 and x = 0.10 stored in a nitrogen glove box over a month and tested regularly under full AM1.5 simulated sunlight (e), and stability of a device with x = 0.10 aged at 0.8 V under continuous ultraviolet-filtered simulated sunlight in a nitrogen atmosphere for over 350 hours (f). g, Device parameters for the passivated (Cs,FA,MA)Pb(I0.85Br0.15)3 (upper) and (Cs,FA,MA)Pb(I0.4Br0.6)3 (lower) perovskite solar cells measured under full simulated solar illumination conditions (AM1.5, 100 mW cm−2).
Extended Data Figure 11 Device statistics.
a–h, Box and whisker plots to summarize the statistics of photovoltaic parameters of ten devices with passivated (Cs,FA,MA)Pb(I0.85Br0.15)3 solar cells (a–d) and eight devices of passivated (Cs,FA,MA)Pb(I0.4Br0.6)3 solar cells (e–h), each measured under full simulated solar illumination conditions (AM1.5, 100 mW cm−2) and scanned at a rate of 15 mV s−1. The boxes represent the interquartile range, with the median represented by the line dividing the boxes, and the whiskers are determined by the 5th and 95th percentiles. The mean is given by the open square symbols, and the cross symbols represent the maximum and minimum values.
Rights and permissions
About this article
Cite this article
Abdi-Jalebi, M., Andaji-Garmaroudi, Z., Cacovich, S. et al. Maximizing and stabilizing luminescence from halide perovskites with potassium passivation. Nature 555, 497–501 (2018). https://doi.org/10.1038/nature25989
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature25989
This article is cited by
-
Effect of sub-bandgap defects on radiative and non-radiative open-circuit voltage losses in perovskite solar cells
Nature Communications (2024)
-
Redox mediator-stabilized wide-bandgap perovskites for monolithic perovskite-organic tandem solar cells
Nature Energy (2024)
-
Efficient blue electroluminescence from reduced-dimensional perovskites
Nature Photonics (2024)
-
Suppression of phase segregation in wide-bandgap perovskites with thiocyanate ions for perovskite/organic tandems with 25.06% efficiency
Nature Energy (2024)
-
Optimizing Carbon-Based Perovskite Solar Cells with Pyramidal Core–Shell Nanoparticles for High Efficiency
Plasmonics (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.