Abstract
Collective cell migration is essential for morphogenesis, tissue remodelling and cancer invasion1,2. In vivo, groups of cells move in an orchestrated way through tissues. This movement involves mechanical as well as molecular interactions between cells and their environment. While the role of molecular signals in collective cell migration is comparatively well understood1,2, how tissue mechanics influence collective cell migration in vivo remains unknown. Here we investigated the importance of mechanical cues in the collective migration of the Xenopus laevis neural crest cells, an embryonic cell population whose migratory behaviour has been likened to cancer invasion3. We found that, during morphogenesis, the head mesoderm underlying the cephalic neural crest stiffens. This stiffening initiates an epithelial-to-mesenchymal transition in neural crest cells and triggers their collective migration. To detect changes in their mechanical environment, neural crest cells use mechanosensation mediated by the integrin–vinculin–talin complex. By performing mechanical and molecular manipulations, we show that mesoderm stiffening is necessary and sufficient to trigger neural crest migration. Finally, we demonstrate that convergent extension of the mesoderm, which starts during gastrulation, leads to increased mesoderm stiffness by increasing the cell density underneath the neural crest. These results show that convergent extension of the mesoderm has a role as a mechanical coordinator of morphogenesis, and reveal a link between two apparently unconnected processes—gastrulation and neural crest migration—via changes in tissue mechanics. Overall, we demonstrate that changes in substrate stiffness can trigger collective cell migration by promoting epithelial-to-mesenchymal transition in vivo. More broadly, our results raise the idea that tissue mechanics combines with molecular effectors to coordinate morphogenesis4.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Roca-Cusachs, P., Sunyer, R. & Trepat, X. Mechanical guidance of cell migration: lessons from chemotaxis. Curr. Opin. Cell Biol. 25, 543–549 (2013)
Mayor, R. & Etienne-Manneville, S. The front and rear of collective cell migration. Nat. Rev. Mol. Cell Biol. 17, 97–109 (2016)
Kerosuo, L. & Bronner-Fraser, M. What is bad in cancer is good in the embryo: importance of EMT in neural crest development. Semin. Cell Dev. Biol. 23, 320–332 (2012)
Gilmour, D., Rembold, M. & Leptin, M. From morphogen to morphogenesis and back. Nature 541, 311–320 (2017)
Nieto, M. A., Huang, R. Y., Jackson, R. A. & Thiery, J. P. EMT: 2016. Cell 166, 21–45 (2016)
Betancur, P., Bronner-Fraser, M. & Sauka-Spengler, T. Assembling neural crest regulatory circuits into a gene regulatory network. Annu. Rev. Cell Dev. Biol. 26, 581–603 (2010)
Nieuwkoop, P. D. & Faber, J. Normal Table of Xenopus laevis (Daudin): a Systematical and Chronological Survey of the Development from the Fertilized Egg till the End of Metamorphosis 2nd edn (North-Holland, 1967)
Theveneau, E. et al. Collective chemotaxis requires contact-dependent cell polarity. Dev. Cell 19, 39–53 (2010)
Alfandari, D., Cousin, H., Gaultier, A., Hoffstrom, B. G. & DeSimone, D. W. Integrin α5β1 supports the migration of Xenopus cranial neural crest on fibronectin. Dev. Biol. 260, 449–464 (2003)
Wei, S. C. et al. Matrix stiffness drives epithelial–mesenchymal transition and tumour metastasis through a TWIST1–G3BP2 mechanotransduction pathway. Nat. Cell Biol. 17, 678–688 (2015)
Sunyer, R. et al. Collective cell durotaxis emerges from long-range intercellular force transmission. Science 353, 1157–1161 (2016)
Koser, D. E. et al. Mechanosensing is critical for axon growth in the developing brain. Nat. Neurosci. 19, 1592–1598 (2016)
Zhou, J., Kim, H. Y. & Davidson, L. A. Actomyosin stiffens the vertebrate embryo during crucial stages of elongation and neural tube closure. Development 136, 677–688 (2009)
Barriga, E. H., Maxwell, P. H., Reyes, A. E. & Mayor, R. The hypoxia factor Hif-1α controls neural crest chemotaxis and epithelial to mesenchymal transition. J. Cell Biol. 201, 759–776 (2013)
Rauzi, M., Verant, P., Lecuit, T. & Lenne, P. F. Nature and anisotropy of cortical forces orienting Drosophila tissue morphogenesis. Nat. Cell Biol. 10, 1401–1410 (2008)
Bjerke, M. A., Dzamba, B. J., Wang, C. & DeSimone, D. W. FAK is required for tension-dependent organization of collective cell movements in Xenopus mesendoderm. Dev. Biol. 394, 340–356 (2014)
Wang, N. et al. Mechanical behavior in living cells consistent with the tensegrity model. Proc. Natl Acad. Sci. USA 98, 7765–7770 (2001)
Storm, C., Pastore, J. J., MacKintosh, F. C., Lubensky, T. C. & Janmey, P. A. Nonlinear elasticity in biological gels. Nature 435, 191–194 (2005)
Pogoda, K. et al. Compression stiffening of brain and its effect on mechanosensing by glioma cells. New J. Phys. 16, 075002 (2014)
Charras, G. & Sahai, E. Physical influences of the extracellular environment on cell migration. Nat. Rev. Mol. Cell Biol. 15, 813–824 (2014)
Moeendarbary, E. et al. The soft mechanical signature of glial scars in the central nervous system. Nat. Commun. 8, 14787 (2017)
Chevalier, N. R. et al. How tissue mechanical properties affect enteric neural crest cell migration. Sci. Rep. 6, 20927 (2016)
Seufert, D. W., Hanken, J. & Klymkowsky, M. W. Type II collagen distribution during cranial development in Xenopus laevis. Anat. Embryol. (Berl.) 189, 81–89 (1994)
Shindo, A. & Wallingford, J. B. PCP and septins compartmentalize cortical actomyosin to direct collective cell movement. Science 343, 649–652 (2014)
Koser, D. E., Moeendarbary, E., Hanne, J., Kuerten, S. & Franze, K. CNS cell distribution and axon orientation determine local spinal cord mechanical properties. Biophys. J. 108, 2137–2147 (2015)
Shyer, A. E. et al. Emergent cellular self-organization and mechanosensation initiate follicle pattern in the avian skin. Science 357, 811–815 (2017)
Dzamba, B. J., Jakab, K. R., Marsden, M., Schwartz, M. A. & DeSimone, D. W. Cadherin adhesion, tissue tension, and noncanonical Wnt signaling regulate fibronectin matrix organization. Dev. Cell 16, 421–432 (2009)
Weber, G. F., Bjerke, M. A. & DeSimone, D. W. A mechanoresponsive cadherin–keratin complex directs polarized protrusive behavior and collective cell migration. Dev. Cell 22, 104–115 (2012)
Wrighton, P. J. & Kiessling, L. L. Forces of change: mechanics underlying formation of functional 3D organ buds. Cell Stem Cell 16, 453–454 (2015)
Poh, Y. C. et al. Generation of organized germ layers from a single mouse embryonic stem cell. Nat. Commun. 5, 4000 (2014)
Mayor, R., Morgan, R. & Sargent, M. G. Induction of the prospective neural crest of Xenopus. Development 121, 767–777 (1995)
Harland, R. M. In situ hybridization: an improved whole-mount method for Xenopus embryos. Methods Cell Biol. 36, 685–695 (1991)
Mancilla, A. & Mayor, R. Neural crest formation in Xenopus laevis: mechanisms of Xslug induction. Dev. Biol. 177, 580–589 (1996)
Iioka, H., Iemura, S., Natsume, T. & Kinoshita, N. Wnt signalling regulates paxillin ubiquitination essential for mesodermal cell motility. Nat. Cell Biol. 9, 813–821 (2007)
Morita, H. et al. Cell movements of the deep layer of non-neural ectoderm underlie complete neural tube closure in Xenopus. Development 139, 1417–1426 (2012)
Weiser, D. C., Row, R. H. & Kimelman, D. Rho-regulated myosin phosphatase establishes the level of protrusive activity required for cell movements during zebrafish gastrulation. Development 136, 2375–2384 (2009)
Di Ciano-Oliveira, C. et al. Is myosin light-chain phosphorylation a regulatory signal for the osmotic activation of the Na+–K+–2Cl− cotransporter? Am. J Physiol. Cell. Physiol. 289, C68 –C81 (2005)
Theveneau, E. & Mayor, R. Beads on the run: beads as alternative tools for chemotaxis assays. Methods Mol. Biol. 769, 449–460 (2011)
Hutter, J. & Bechhoefer, J. Calibration of atomic-force microscope tips. Rev. Sci. Instrum. 64, 1868–1873 (1993)
Iwashita, M., Kataoka, N., Toida, K. & Kosodo, Y. Systematic profiling of spatiotemporal tissue and cellular stiffness in the developing brain. Development 141, 3793–3798 (2014)
Benko, R. & Brodland, G. W. Measurement of in vivo stress resultants in neurulation-stage amphibian embryos. Ann. Biomed. Eng. 35, 672–681 (2007)
Goto, T., Davidson, L., Asashima, M. & Keller, R. Planar cell polarity genes regulate polarized extracellular matrix deposition during frog gastrulation. Curr. Biol. 15, 787–793 (2005)
Acknowledgements
We thank A. J. Thompson for assistance with the AFM. This study was supported by grants to R.M. from the Medical Research Council (J000655), Biotechnology and Biological Sciences Research Council (M008517) and Wellcome Trust; to K.F. from the Medical Research Council (Career Development Award G1100312/1) and from the Eunice Kennedy Shriver National Institute of Child Health & Human Development of the National Institutes of Health (R21HD080585); to G.C. from the European Research Council (Consolidator grant MolCellTissMech, agreement 647186); and by postdoctoral fellowships to E.H.B. from EMBO (LTF-971) and Marie Skłodowska Curie (IF-2014_ST VivoMechCollMigra, agreement 658536). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
E.H.B. and R.M. conceived the project, E.H.B., K.F., G.C. and R.M. designed the experiments. All the experiments and analyses were performed by E.H.B. E.H.B. and R.M. wrote the manuscript. All the authors edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer Information Nature thanks L. A. Davidson, N. Wang and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Figure 1 Neural crest migration is controlled by environmental factors.
a–d, Heterochronic graft controls. In all grafts, we completely removed the host neural crest before implanting the graft, and all grafted cells migrated 20 h post graft, as expected for embryos reaching the migratory stage7 (stage 23). a, Labelled pre-migratory neural crest grafted into unlabelled non- and pre-migratory hosts. Ten hours post graft, representative examples show that pre-migratory neural crest grafted into non-migratory hosts does not migrate and that pre-migratory neural crest grafted into pre-migratory hosts does migrate. b, Labelled non-migratory neural crest grafted into unlabelled non- and pre-migratory hosts. Ten hours post graft, representative examples show that non-migratory neural crests grafted into non-migratory hosts do not migrate and that pre-migratory neural crests grafted into pre-migratory hosts do migrate. Twenty hours post-graft panels in a and b show that neural crests migrated in all conditions after 20 h. c, Normalized neural crest migration. Histograms show mean, error bars represent s.d.; n = 80 measurements from 20 embryos (one-way ANOVA, P < 0.0001; two-tailed t-test, **P = 0.0026, ****P < 0.0001, CI = 95%). d, Dorsal views of embryos hybridized with a probe against snail2 after dissection of the host neural crest, showing that endogenous neural crest was completely removed before grafting. a, b and d show representative examples of three independent experiments; scale bar, 250 μm.
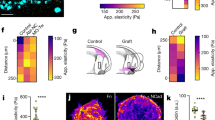
Extended Data Figure 2 Fibronectin expression under the neural crest in vivo and contribution to tissue stiffness, stiffness measurements across epidermis, and ex vivo system characterization.
a–d, Analysis of fibronectin expression under the neural crest. a, Schematic showing the imaging method used to access fibronectin under the neural crest. b, Immunostaining for fibronectin in embryos at non- and pre-migratory stages. 20× panels, yellow outline highlights the anatomical position of the neural crest; green box highlights the region shown in the zoom panels. Zoom panels show heat maps of fibronectin signal (representative of five independent samples). c, Fluorescence signal intensity. Histograms show mean, error bars represent s.d. (two-tailed t-test, CI = 95%). d, Fibronectin signal thickness in the mesoderm at pre- and non-migratory stages. Green lines show median and red whiskers represent interquartile range; n = 10 embryos (c), n = 26 sections from three independent experiments (d) (two-tailed t-test, CI = 95%). Scale bars: 50 μm (20× panels), 20 μm (zoom panels). e, f, Apparent elasticity was measured before and after mechanical removal of the ECM by gently scraping the surface of the tissue with a hair loop. e, Apparent elasticity was measured directly on the mesoderm in the presence (Fn(+)) or absence (Fn(−)) of fibronectin (Fn, green) in embryos at stage 21 (pre-migratory). Graphs plot spread of data; green lines show median; red whiskers represent interquartile range. Two-tailed Mann–Whitney U-test, P as indicated, CI = 95%; n = number of AFM indentations: n = 124 (Fn(+)), n = 157 (Fn(−)). Mean indentation depth = 8 μm. f, Representative immunostaining against fibronectin (Fn), conditions as indicated, n = 7 embryos. Scale bar, 100 μm. g, h, AFM controls. g, Schematic of iAFM measurement. To check that the dissection of the epidermis did not modify the mesodermal elasticity, we measured the elastic modulus through the epidermis, and no differences were found (compare with Fig. 1h). h, Apparent elasticity. Box plots show the median, box edges represent the 25th and 75th percentiles, and whiskers show the spread of data (excluding outliers). Two-tailed Mann–Whitney U-test, ***P = 0.0006, CI = 95%; n = number of embryos, δ = mean indentation depth. i, j, Representative examples of force curves obtained from measurements performed using cantilevers coated with either 37-μm beads (i, n = 2386) or 90-μm beads (j, n = 28). Red lines represent cantilever extension and dark blue lines represent retraction. Indentation depth (δ) for each curve is provided. k–m, Ex vivo system characterization. k, Cross-sectional confocal image of stiff or soft PAA hydrogels, confirming that gels (red) of varying stiffness are evenly functionalized with fibronectin (green). l, Fibronectin thickness does not change between soft and stiff gels. Histograms represent mean and bars show s.d.; n = 8 gels; two-tailed t-test, CI = 95%. Results in k and l are representative of gels from three independent experiments. m, AFM measurements. Spread of data, green lines show median, red whiskers represent interquartile range; two-tailed Mann–Whitney U-test, ****P < 0.0001, CI = 95%; n = 60 (stiff), n = 50 (soft) from 10 gels. Scale bars, 50 μm (k).
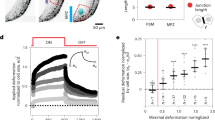
Extended Data Figure 3 Neural crest motility, dispersion and EMT are controlled by substrate mechanics.
a–d, Protrusion and chemotaxis analysis of collective and single neural crest cells plated on soft and stiff substrates. a, Confocal projections of labelled neural crest collectives plated on soft or stiff gels. b, Confocal projections of labelled single neural crest cells plated on soft or stiff gels. Protrusion area (c) and chemotaxis quantifications (d) for collective and single neural crest cells plated on soft or stiff gels, box plots show median, box edges represent 25th and 75th percentiles and whiskers show the spread of data (excluding outliers). n = 63 cells (protrusion area), n = 79 cells (chemotaxis); one-way ANOVA, P < 0.0001; two-tailed t-test, ****P < 0.0001, CI = 95%. e–h, Neural crest dispersion analysis on gels of varying stiffness. e, Neural crest cells labelled with nRFP and plated on stiff or soft gels are shown at 0 h and 17 h after plating. f–h, Quantification of cell dispersion. f, Colour-coded Delaunay triangulation shown at 17 h after plating to facilitate visualization of the distances between neighbour cells. g, Quantification of Delaunay triangulation. Green lines show median, red whiskers represent the interquartile range; two-tailed Mann–Whitney U-test, ****P < 0.0001, CI = 95%; n = 730 triangles from 31 explants. h, Percentage of dispersing explants. Histograms show mean and error bars represent s.e.m.; two-tailed t-test, ****P < 0.0001, CI = 95%; n = 31 explants. g and h show data from four independent experiments (soft) and three independent experiments (stiff). i, j, Immunostaining for E-cadherin (i) and N-cadherin (j) on neural crests plated on substrates of varying stiffness. a, b, e, i and j show representative examples from three independent experiments. Scale bars: 20 μm (a, b), 250 μm (e), 50 μm (i, j, main), 10 μm (i, j, zoom).
Extended Data Figure 4 Posterior ablations, mesoderm targeted injections and extrinsic compression characterization.
a, b, Ablation controls. Mechanical tension in Xenopus is higher in the anterior region of the embryo than in posterior territories12,41. Consistent with this observation, no effect on neural crest migration or tissue stiffness was observed when the ablation was done in posterior regions. a, Schematic showing embryos ablated at non-migratory stages, and neural crest migration at stage 23 (migratory)7. b, Lateral views of control and posterior-ablated embryos hybridized with a probe against snail2. Scale bar, 250 μm. c, d, Targeted injections into the head mesoderm. c, Cartoon shows that injecting two dorso-vegetal blastomeres targets the head mesoderm. d, Confocal images of targeted injections showing nuclear RFP expression in the mesoderm. Scale bar, 100 μm. e–g, Compression controls. e, Images of embryos being compressed from non-migratory to pre-migratory stages with a 90-μm bead attached to an AFM cantilever (bead, red circumference). Neural plate border highlighted with red lines and neural crest position shown in light blue; stages as labelled. f, Magnitude maps from a particle image velocity (PIV) analysis indicates the x–y extent of the deformation induced by AFM indentation. g, Maximum projection of a cross-section showing the z-deformation generated by the AFM compression. Epidermis (ep) and mesoderm (ms) are being deformed, mean maximum indentation depth (δ) is provided (nuclei are visible in the bead region owing to a maximum projection effect). h, i, Result of extrinsic stress experiments. h, Dorsal view (anterior to top) of embryos hybridized with a probe against snail2; black arrows point to migrating neural crest. i, Percentage of embryos displaying neural crest migration. Histograms show mean, error bars represent s.e.m.; n = 13 embryos; two-tailed t-test, **P = 0.001, CI = 95%. All experiments repeated three times. Scale bars: 200 μm (e, f), 100 μm (g), 200 μm (h).
Extended Data Figure 5 Mesodermal stiffening promotes neural crest CCM in a non-autonomous manner in vivo.
a–d, Non-autonomous effect of mesodermal myosin manipulations on neural crest migration in vivo. a, Wild-type labelled pre-migratory neural crest (cyan) grafted into pre-migratory wild-type, myl9-MO or CA-MYPT-injected hosts (mesoderm in red). b, Representative embryos showing normal neural crest migration in wild-type hosts and inhibited neural crest migration in myl9-MO or CA-MYPT hosts. Scale bar, 200 μm. c, d, Normalized neural crest migration (c, n = 22 embryos) and percentage of embryos displaying neural crest migration (d, n = 38 embryos) (c, d; one-way ANOVA, P < 0.0001; two-tailed t-test, ***P < 0.0006, ****P < 0.0001, CI = 95%). Histograms in c, d and g show mean, error bars represent s.d. (c) or s.e.m. (d, g). e–g, Effect of premature mesodermal stiffening on neural crest migration in vivo. e, Wild-type labelled pre-migratory neural crest grafted into non-migratory wild-type or CA-MLC hosts. f, Representative embryos showing premature neural crest migration in CA-MLC hosts, whereas no migration is observed in control embryos at this stage. Scale bar, 100 μm. g, Percentage of embryos displaying neural crest migration. Two-tailed t-test, ****P < 0.0001, CI = 95%, n = 20 embryos. h–k, Non-autonomous effect of DshDEP+ on neural crest migration. h, Wild-type labelled pre-migratory neural crest (cyan) was grafted into pre-migratory wild-type or DshDEP+ hosts. i, Representative examples showing neural crest migration in wild-type hosts and inhibited neural crest migration in DshDEP+-injected hosts (mesoderm in red). Scale bar, 150 μm. Normalized neural crest migration (j) and percentage of embryos displaying neural crest migration (k) (j, k; two-tailed t-test, ***P < 0.0002, ****P < 0.0001, CI = 95%, n = 14 embryos). Histograms in j and k show mean, error bars represent s.d. (j) and s.e.m. (k). b, f and i are representative examples of three independent experiments.
Extended Data Figure 6 Head mesoderm manipulations did not affect fibronectin deposition and organization under the neural crest.
As it has been previously shown that inhibition of myosin and PCP can affect fibronectin deposition and organization in the dorsal mesoderm27,28,42; we analysed the effect of these treatments on the lateral mesoderm underlying the premigratory neural crest. Our results show no effect of inhibition of myosin or PCP on fibronectin deposition or organization in the lateral mesoderm, indicating that the effect of our treatments on neural crest migration are not due to an indirect effect on fibronectin, but rather a consequence of affecting mesoderm stiffness. a–c, Transverse sections showing fibronectin expression with different treatments used to manipulate the apparent elasticity of head mesoderm. a, Schematic showing the area presented in b and c. b, c, Immunostaining against fibronectin (green) and DAPI stain (blue) showing the fibronectin signal underneath the neural crest, treatments as labelled in each panel. Scale bar, 30 μm. d–g, Effect of mesoderm manipulation on fibronectin assembly. d, Dorsal view of a flat-mounted embryo. White square indicates the area shown in e and f. e, Immunostaining against fibronectin (green) in non-migratory embryos (stage 13), showing fibronectin assembly underneath the neural crest, treatments as indicated. f, Immunostaining against fibronectin (green) in pre-migratory embryos (stage 20) showing fibronectin assembly underneath the neural crest, treatments as indicated. Control to show that inhibition of PCP signalling (DshDEP+) affects fibronectin assembly in dorsal mesoderm42 (g) but not lateral mesoderm (f). Scale bars, 30 μm. b, c and e–g are representative examples of three independent experiments.
Extended Data Figure 7 Neural crest senses mesoderm stiffening by using an integrin–vinculin–talin-mediated mechanoresponsive mechanism.
a–c, Analysis of neural crest migration after integrin β1, vinculin or talin knockdown. a, Lateral views of embryos hybridized with a probe against snail2. Control embryos migrate normally from dorsal to ventral. However, neural crest migration is drastically inhibited in embryos treated with itgβ1-MO or the dominant-negative mutants vinculin-Cter and talin-Nter. Embryos in a are representative examples of three independent experiments. b, c, Quantification of neural crest migration showing percentage of embryos (b) and normalized neural crest migrated distances (c). Histograms in b and c show mean, error bars represent s.e.m. (b) and s.d. (c); one-way ANOVA, P < 0.0001; two-tailed t-test, ***P < 0.0002 ****P < 0.0001, CI = 95%; n = 39 embryos. Scale bar, 200 μm.
Extended Data Figure 8 Actomyosin expression in the head mesoderm does not change from non- to pre-migratory stages.
a–e, Analysis of actin and phospho-myosin expression under the neural crest. As actomyosin contractility has been proposed to contribute to paraxial mesoderm stiffness in Xenopus13, we assessed actomyosin activity in the head mesoderm, the substrate of the neural crest. Note that the expression levels of either actin or phospho-myosin remained unchanged at pre-migratory stages compared to non-migratory stages. a, Schematic showing the imaging method used to access the head mesoderm under the neural crest. b, Phalloidin stain was used to label actin on embryos at non- and pre-migratory stages. d, Immunostaining against phospho-myosin on embryos at non- and pre-migratory stages. c, e, Quantification of fluorescence intensity across the membrane in b and d, respectively, represented by a continuous line. Points show mean and error bars represent s.d.; n, number of measurements per data (time) point; n = 16 (c), n = 15 (e); from three independent experiments; non-significant differences were observed, P value is indicated in each chart (two-tailed t-test, CI = 95%). Scale bars, 20 μm.
Extended Data Figure 9 Myosin contractility is not required to maintain mesodermal stiffness.
a–f, Blebbistatin incubation experiments. To functionally test the effect of actomyosin contractility on mesoderm stiffness, we applied blebbistatin to pre-migratory embryos. Note that blebbistatin treatment at this stage did not affect mesoderm stiffness, cell density or neural crest migration, in spite of a marked reduction in myosin phosphorylation. a, Lateral views of embryos hybridized with a probe against snail2, showing no differences in neural crest migration after 4 h of incubation with DMSO or blebbistatin (BB). Embryos were incubated as indicated. Scale bar, 150 μm. Normalized neural crest migration (b) and percentage of embryos (c). In b and c, histograms show mean and error bars represent s.d. (b) and s.e.m. (c); one-way ANOVA, P < 0.0001; two-tailed t-test **P < 0.008, ***P < 0.002, CI = 95%, n = 19 embryos. d, iAFM measurement direct on mesoderm. Spread of data plotted for each condition; green lines show median, red whiskers represent the interquartile range (two-tailed Mann–Whitney U-test, P value as indicated, CI = 95%; n = number of AFM indentations, n = 116 (DMSO), n = 204 (BB_pre). e, f, As blebbistatin treatment could potentially affect cell–cell adhesion, thus promoting cell dispersion, we measured its effect on cell dispersion by calculating the distance between neighbouring mesoderm cells. e, Chart comparing the mean area of the triangle formed by each mesoderm cell with its neighbours, calculated by Delaunay triangulation (Mann–Whitney U-test, P value is shown). n = number of embryos, δ = mean maximum indentation depth. f, Images showing representative neural crest and mesoderm cells (treated as indicated) and triangles from which the areas in e were calculated. Scale bar, 200 μm. g, Immunostaining for phospho-myosin; dorsal view of a flat-mounted embryo and colour-coded intensity of zoom images at the indicated time points are shown (scale in arbitrary units, a.u.). Scale bars, 100 μm (whole), 15 μm (zoom). h, Fluorescence intensity in a.u. across the white line in the zoom panels in g. All experiments repeated at least three times. Images in a, f and g are representative of three independent experiments.
Extended Data Figure 10 Extrinsic compression modifies the environment of PCP-depleted embryos and triggers premature neural crest CCM in vivo.
a, Magnitude maps from a particle image velocity (PIV) analysis showing the x–y extent of the stress field generated by the extrinsic compression experiment. Neural plate border is indicated by red lines, neural crest is shown in light blue. Scale bar, 200 μm. b, c, Extrinsic stress-induced premature neural crest migration. b, Lateral views of embryos hybridized with a probe against snail2. Note that premature neural crest migration is observed in the DshDEP+/stressed panel. Normalized neural crest migration (c) and percentage of embryos displaying each phenotype (d). Histograms show mean, error bars show s.d.(c) and s.e.m. (d); one-way ANOVA, P < 0.0001; two-tailed t-test, ****P < 0.0001, CI = 95%, n = 18 embryos. a and b show representative images of three independent experiments. Scale bar, 150 μm.
Supplementary information
Chemotaxis assay testing the collective response of non- and pre-migratory neural crest clusters towards Sdf-1 on glass
Neural crest labelled with nuclear (magenta) and membrane (cyan) markers. Note that both populations directionally migrate towards Sdf-1. Time-lapse setting was 1 picture every 5 min; 49 frames are shown; 20x objective. (AVI 1233 kb)
Chemotaxis assay testing the collective response of neural crest clusters to Sdf-1 on substrates of varying stiffness
Neural crest labelled with nuclear (magenta) and membrane (cyan) markers. Note that cells migrating on soft gels do not migrate towards Sdf-1. Time-lapse setting was 1 picture every 5 min; 50 frames are shown; 63x water-immersion objective. (AVI 869 kb)
Confocal time-lapse of neural crest clusters migrating toward Sdf-1 on substrates of varying stiffness
Neural crest labelled with nuclear (magenta) and membrane (cyan) markers. Sdf-1 located to the right side of the NC. Note the lack of polarity in cells plated on soft gels. Time-lapse setting was 1 picture every 16 seconds; 29 frames are shown; 63x objective. (AVI 1543 kb)
Dispersion assay for neural crest clusters explanted on substrates of varying stiffness
Neural crest labelled with nuclear (magenta) and membrane (cyan) markers. Notice that cells migrating on soft gels do not disperse. Time-lapse setting was 1 picture every 5 min; 250 frames are shown; 20x objective. (AVI 5669 kb)
Source data
Rights and permissions
About this article
Cite this article
Barriga, E., Franze, K., Charras, G. et al. Tissue stiffening coordinates morphogenesis by triggering collective cell migration in vivo. Nature 554, 523–527 (2018). https://doi.org/10.1038/nature25742
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature25742
This article is cited by
-
Mechanical state transitions in the regulation of tissue form and function
Nature Reviews Molecular Cell Biology (2024)
-
Ultrasound-assisted tissue engineering
Nature Reviews Bioengineering (2024)
-
Adherens junctions as molecular regulators of emergent tissue mechanics
Nature Reviews Molecular Cell Biology (2024)
-
Profiling native pulmonary basement membrane stiffness using atomic force microscopy
Nature Protocols (2024)
-
Bioelectric stimulation controls tissue shape and size
Nature Communications (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.