Abstract
Both microbial and host genetic factors contribute to the pathogenesis of autoimmune diseases1,2,3,4. There is accumulating evidence that microbial species that potentiate chronic inflammation, as in inflammatory bowel disease, often also colonize healthy individuals. These microorganisms, including the Helicobacter species, can induce pathogenic T cells and are collectively referred to as pathobionts4,5,6. However, how such T cells are constrained in healthy individuals is not yet understood. Here we report that host tolerance to a potentially pathogenic bacterium, Helicobacter hepaticus, is mediated by the induction of RORγt+FOXP3+ regulatory T (iTreg) cells that selectively restrain pro-inflammatory T helper 17 (TH17) cells and whose function is dependent on the transcription factor c-MAF. Whereas colonization of wild-type mice by H. hepaticus promoted differentiation of RORγt-expressing microorganism-specific iTreg cells in the large intestine, in disease-susceptible IL-10-deficient mice, there was instead expansion of colitogenic TH17 cells. Inactivation of c-MAF in the Treg cell compartment impaired differentiation and function, including IL-10 production, of bacteria-specific iTreg cells, and resulted in the accumulation of H. hepaticus-specific inflammatory TH17 cells and spontaneous colitis. By contrast, RORγt inactivation in Treg cells had only a minor effect on the bacteria-specific Treg and TH17 cell balance, and did not result in inflammation. Our results suggest that pathobiont-dependent inflammatory bowel disease is driven by microbiota-reactive T cells that have escaped this c-MAF-dependent mechanism of iTreg–TH17 homeostasis.
This is a preview of subscription content, access via your institution
Access options




Similar content being viewed by others
References
Round, J. L. & Mazmanian, S. K. The gut microbiota shapes intestinal immune responses during health and disease. Nat. Rev. Immunol. 9, 313–323 (2009)
Hooper, L. V., Littman, D. R. & Macpherson, A. J. Interactions between the microbiota and the immune system. Science 336, 1268–1273 (2012)
Kamada, N., Seo, S. U., Chen, G. Y. & Núñez, G. Role of the gut microbiota in immunity and inflammatory disease. Nat. Rev. Immunol. 13, 321–335 (2013)
Chow, J., Tang, H. & Mazmanian, S. K. Pathobionts of the gastrointestinal microbiota and inflammatory disease. Curr. Opin. Immunol. 23, 473–480 (2011)
Kullberg, M. C. et al. IL-23 plays a key role in Helicobacter hepaticus-induced T cell-dependent colitis. J. Exp. Med. 203, 2485–2494 (2006)
Hue, S. et al. Interleukin-23 drives innate and T cell-mediated intestinal inflammation. J. Exp. Med. 203, 2473–2483 (2006)
Sanderson, S., Campbell, D. J. & Shastri, N. Identification of a CD4+ T cell-stimulating antigen of pathogenic bacteria by expression cloning. J. Exp. Med. 182, 1751–1757 (1995)
Yang, Y. et al. Focused specificity of intestinal TH17 cells towards commensal bacterial antigens. Nature 510, 152–156 (2014)
Kearney, E. R., Pape, K. A., Loh, D. Y. & Jenkins, M. K. Visualization of peptide-specific T cell immunity and peripheral tolerance induction in vivo. Immunity 1, 327–339 (1994)
Moon, J. J. et al. Naive CD4+ T cell frequency varies for different epitopes and predicts repertoire diversity and response magnitude. Immunity 27, 203–213 (2007)
Sefik, E. et al. Individual intestinal symbionts induce a distinct population of RORγ+ regulatory T cells. Science 349, 993–997 (2015)
Ohnmacht, C. et al. The microbiota regulates type 2 immunity through RORγt+ T cells. Science 349, 989–993 (2015)
Schiering, C. et al. The alarmin IL-33 promotes regulatory T-cell function in the intestine. Nature 513, 564–568 (2014)
Hirota, K. et al. Fate mapping of IL-17-producing T cells in inflammatory responses. Nat. Immunol. 12, 255–263 (2011)
Hand, T. W. et al. Acute gastrointestinal infection induces long-lived microbiota-specific T cell responses. Science 337, 1553–1556 (2012)
Chai, J. N. et al. Helicobacter species are potent drivers of colonic T cell responses in homeostasis and inflammation. Sci. Immunol. 2, eaal5068 (2017)
Yang, B. H. et al. Foxp3+ T cells expressing RORγt represent a stable regulatory T-cell effector lineage with enhanced suppressive capacity during intestinal inflammation. Mucosal Immunol. 9, 444–457 (2016)
Ciofani, M. et al. A validated regulatory network for Th17 cell specification. Cell 151, 289–303 (2012)
Apetoh, L. et al. The aryl hydrocarbon receptor interacts with c-Maf to promote the differentiation of type 1 regulatory T cells induced by IL-27. Nat. Immunol. 11, 854–861 (2010)
Hoshi, N. et al. MyD88 signalling in colonic mononuclear phagocytes drives colitis in IL-10-deficient mice. Nat. Commun. 3, 1120 (2012)
Ivanov, I. I. et al. Specific microbiota direct the differentiation of IL-17-producing T-helper cells in the mucosa of the small intestine. Cell Host Microbe 4, 337–349 (2008)
Gorelik, L. & Flavell, R. A. Abrogation of TGFβ signaling in T cells leads to spontaneous T cell differentiation and autoimmune disease. Immunity 12, 171–181 (2000)
Chaudhry, A. et al. CD4+ regulatory T cells control TH17 responses in a Stat3-dependent manner. Science 326, 986–991 (2009)
Haribhai, D. et al. A requisite role for induced regulatory T cells in tolerance based on expanding antigen receptor diversity. Immunity 35, 109–122 (2011)
Round, J. L. et al. The Toll-like receptor 2 pathway establishes colonization by a commensal of the human microbiota. Science 332, 974–977 (2011)
Kim, K. S. et al. Dietary antigens limit mucosal immunity by inducing regulatory T cells in the small intestine. Science 351, 858–863 (2016)
Atarashi, K. et al. Treg induction by a rationally selected mixture of Clostridia strains from the human microbiota. Nature 500, 232–236 (2013)
Rubtsov, Y. P. et al. Regulatory T cell-derived interleukin-10 limits inflammation at environmental interfaces. Immunity 28, 546–558 (2008)
Wende, H. et al. The transcription factor c-Maf controls touch receptor development and function. Science 335, 1373–1376 (2012)
Awasthi, A. et al. Cutting edge: IL-23 receptor GFP reporter mice reveal distinct populations of IL-17-producing cells. J. Immunol. 182, 5904–5908 (2009)
Dash, P. et al. Paired analysis of TCRα and TCRβ chains at the single-cell level in mice. J. Clin. Invest. 121, 288–295 (2011)
Lefranc, M. P. et al. IMGT, the international ImMunoGeneTics information system. Nucleic Acids Res. 37, D1006–D1012 (2009)
Ise, W. et al. CTLA-4 suppresses the pathogenicity of self antigen-specific T cells by cell-intrinsic and cell-extrinsic mechanisms. Nat. Immunol. 11, 129–135 (2010)
Mach, N. et al. Differences in dendritic cells stimulated in vivo by tumors engineered to secrete granulocyte-macrophage colony-stimulating factor or Flt3-ligand. Cancer Res. 60, 3239–3246 (2000)
Reche, P. A. & Reinherz, E. L. Prediction of peptide–MHC binding using profiles. Methods Mol. Biol. 409, 185–200 (2007)
Kouskoff, V., Signorelli, K., Benoist, C. & Mathis, D. Cassette vectors directing expression of T cell receptor genes in transgenic mice. J. Immunol. Methods 180, 273–280 (1995)
Altman, J. D. et al. Phenotypic analysis of antigen-specific T lymphocytes. Science 274, 94–96 (1996)
Ostanin, D. V. et al. T cell transfer model of chronic colitis: concepts, considerations, and tricks of the trade. Am. J. Physiol. Gastrointest. Liver Physiol. 296, G135–G146 (2009)
Read, S., Malmström, V. & Powrie, F. Cytotoxic T lymphocyte-associated antigen 4 plays an essential role in the function of CD25+CD4+ regulatory cells that control intestinal inflammation. J. Exp. Med. 192, 295–302 (2000)
Dobin, A . et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013)
Liao, Y., Smyth, G. K. & Shi, W. featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30, 923–930 (2014)
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014)
Johnson, W. E., Li, C. & Rabinovic, A. Adjusting batch effects in microarray expression data using empirical Bayes methods. Biostatistics 8, 118–127 (2007)
R Development Core Team. R: A Language And Environment For Statistical Computing ; https://www.r-project.org/ (R Foundation for Statistical Computing, Vienna, Austria, 2016)
Acknowledgements
We thank S. Y. Kim and the NYU Rodent Genetic Engineering Laboratory (RGEL) for generating TCR transgenic mice, A. Heguy and colleagues at the NYU School of Medicine’s Genome Technology Center (GTC) for preparation of RNA-seq libraries and RNA sequencing, the NIH Tetramer Core Facility for generating MHC class II tetramers, K. Murphy for providing the 58α−β− hybridoma line, D. E. Levy for providing the Stat3fl/fl;Cd4cre mice, J. Fox for providing the H. hepaticus strain, P. Dash and P. G. Thomas for advice on single-cell TCR cloning, and J. A. Hall, J. Muller and J. Lafaille for suggestions on the manuscript. The Experimental Pathology Research Laboratory of NYU Medical Center is supported by National Institutes of Health Shared Instrumentation grants S10OD010584-01A1 and S10OD018338-01. The GTC is partially supported by the Cancer Center Support grant P30CA016087 at the Laura and Isaac Perlmutter Cancer Center. This work was supported by the Irvington Institute fellowship program of the Cancer Research Institute (M.X.); the training program in Immunology and Inflammation 5T32AI100853 (M.P.); the Helen and Martin Kimmel Center for Biology and Medicine (D.R.L.); the Colton Center for Autoimmunity (D.R.L.); and National Institutes of Health grant R01DK103358 (R.B. and D.R.L.). D.R.L. is an Investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
M.X. and M.P. designed and performed all experiments and analysed the data. Y.D. performed blinded histology scoring on colitis sections. C.A. and C.G. assisted with in vivo and in vitro experiments. R.Y. and M.P. performed RNA-seq analysis. O.J.H. and Y.B. analysed the Gata3∆Treg mouse phenotype. R.B. supervised RNA-seq analysis. M.X., M.P. and D.R.L. wrote the manuscript with input from the co-authors. D.R.L. supervised the research and contributed to experimental design.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer Information Nature thanks C. Ohnmacht and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Figure 1 Cloning and characterization of H. hepaticus-specific TH17 TCRs, and generation of TCRtg mice and MHC class II tetramers.
a, IL-23R–GFP expression in CD4+ T cells from the large intestines of mice with and without H. hepaticus colonization and after IL-10RA blockade. Data are from one of five independent experiments. b, Experimental scheme for cloning H. hepaticus-induced single IL-23R–GFP+ (predominantly TH17) cell TCRs under IL-10RA blockade. c, Summary of the 12 dominant H. hepaticus-induced TH17 TCRs. d, In vitro activation of CFSE-labelled naive HH7-2tg and HH5-1tg cells by indicated stimuli in the presence of antigen-presenting cells. Data are from one of two independent experiments. e, f, Expansion of donor-derived HH7-2tg (e) and HH5-1tg (f) (CD45.2) cells in the large intestine of H. hepaticus-colonized or -free CD45.1 mice, gated on total CD4+ T cells. Data are from one of three independent experiments. g, HH-E2 tetramer staining of CD4+ T cells from the large intestine of H. hepaticus-colonized or -free mice. Data are from one of six independent experiments.
Extended Data Figure 2 Extended characterization of SFB- and H. hepaticus-specific T cells in distinct anatomical sites in bacteria-colonized wild-type mice.
a, Representative flow cytometry plots of donor-derived HH7-2tg (CD45.1/45.2) and 7B8tg (CD45.2/45.2) T cells in indicated tissues of mice colonized with SFB and H. hepaticus, gated on total CD4+ T cells (CD4+CD3+) (n = 15). b, Proportions of donor-derived HH7-2tg and 7B8tg T cells among total CD4+ T cells in indicated tissues. Data in a and b are from 1 of 3 experiments, with a total of 15 mice in the 3 experiments. c, Representative flow cytometry plots of RORγt, FOXP3, BCL6 and CXCR5 expression in CD4+ T cells from the host and from HH7-2tg and 7B8tg donors in different tissues (n = 15). d, Frequencies of Treg (FOXP3+), TH17 (FOXP3-RORγt+) and TFH (BCL6+CXCR5+) cells among donor-derived HH7-2tg and 7B8tg cells in different tissues. Data are from 1 of 3 experiments, with a total of 15 mice in the 3 experiments. e, Representative flow cytometry plots of FOXP3, RORγt, GATA3 and ST2 expression in CD4+ T cells from the host (blue) and from HH7-2tg donors (red) in the LILP (n = 5). f, Representative flow cytometry plots of FOXP3, RORγt, GATA3 and ST2 expression in total CD4+ (green) and HH-E2 tetramer+ (red) T cells in the LILP (n = 5). Spl, spleen.
Extended Data Figure 3 Extended characterization of H. hepaticus-specific TCRtg cell differentiation.
a, HH7-2tg;Rag1−/− mice do not develop Treg cells in the thymus. Representative flow cytometry plots of Treg (FOXP3+CD25+) frequency in indicated tissues of H. hepaticus-free HH7-2tg;Rag1+/− (n = 3) or HH7-2tg;Rag1−/− (n = 3) mice. b, c, HH7-2tg;Rag1−/− and HH7-2tg;Rag1+/− donor-derived T cells differentiated into equal frequencies of RORγt+ Treg cells in the large intestine of wild-type mice. Equal numbers (2,000) of congenic isotype-labelled HH7-2tg;Rag1+/− (CD45.1/45.1) and Rag1−/− (CD45.1/45.2) naive T cells were co-transferred into H. hepaticus-colonized wild-type B6 mice. Cells from the LILP were analysed two weeks after transfer. Data summarize two independent experiments (n = 6). b, Representative flow cytometry plots of donor and recipient T cell frequency (left), and RORγt and FOXP3 expression (right) (n = 6). c, Frequencies of Treg (FOXP3+), TH17 (FOXP3−RORγt+) and TFH (BCL6+CXCR5+) cells among HH7-2tg Rag1+/− (n = 6) and Rag1−/− (n = 6) donor-derived T cells. d, e, Approximately 2,000 naive HH5-1tg cells (CD45.1/45.2) were adoptively transferred into wild-type B6 mice (CD45.2/45.2) colonized with H. hepaticus. Cells from LILP and the caecal patch were analysed two weeks after transfer. d, Representative flow cytometry plots are shown for RORγt, FOXP3, BCL6 and CXCR5 expression in donor-derived and recipient CD4+ T cells in the indicated tissues. e, Frequencies of Treg (FOXP3+), TH17 (FOXP3−RORγt+) and TFH (BCL6+CXCR5+) among HH5-1tg donor T cells (n = 8). Data are a summary of eight mice from two independent experiments. All statistics were calculated by unpaired two-sided Welch’s t-test. Error bars denote mean ± s.d. P values are indicated on the figure.
Extended Data Figure 4 Differentiation of SFB- and H. hepaticus-specific T cells in Il10+/− and Il10−/− mice.
a–d, Equal numbers (10,000) of congenic isotype-labelled HH7-2tg (CD45.1/45.2) and 7B8tg (CD45.1/45.1) T cells were co-transferred into Il10−/− and Il10+/− mice (CD45.2/45.2) colonized with both H. hepaticus and SFB. Intestinal T cells were examined two weeks later. a, Representative flow cytometry plots of FOXP3, RORγt and T-bet expression in total and FOXP3− host CD4+ T cells in the SILP and LILP of Il10+/− (n = 10) and Il10−/− (n = 8) mice that received TCR transgenic T cell transplants. b, Frequencies of Treg (FOXP3+) and TH17 (FOXP3−RORγt+) cells among SILP 7B8tg donor-derived cells in Il10+/− (n = 10) and Il10−/− (n = 8) mice. Data for a and b are a summary of four independent experiments. c, Representative flow cytometry plots of IL-10, IL-17A and IFNγ expression in transferred 7B8tg and HH7-2tg cells from LILP and SILP of Il10+/− and Il10−/− mice after re-stimulation (n = 5 or 6). d, Proportions of transferred 7B8tg and HH7-2tg cells in the SILP and LILP of Il10+/− and Il10−/− mice that express IL-10, IL-17A and IFNγ after re-stimulation (n = 5 or 6). Data for c and d are a summary of two independent experiments. e, 2,000 naive HH5-1tg cells (CD45.1/45.2) were adoptively transferred into Il10+/− and Il10−/− mice colonized with H. hepaticus. Cells from the LILP were analysed two weeks after transfer (n = 5). Representative flow cytometry plots of RORγt and FOXP3 expression in HH5-1tg donor cells are shown (left), along with a compilation of frequencies of Treg (FOXP3+) and TH17 (FOXP3−RORγt+). f, g, RORγt and FOXP3 expression in total CD4+ and HH-E2 tetramer+ T cells (f) and frequencies (above) and absolute numbers (below) of Treg (FOXP3+) and TH17 (FOXP3-RORγt+) cells among HH-E2 tetramer+ T cells (g) in the LILP of Il10+/− (day 25, n = 8) and Il10−/− (day 12 n = 8, day 25 n = 7, day 42 n = 8) mice colonized with H. hepaticus for indicated times. All statistics were calculated by unpaired two-sided Welch’s t-test. Error bars denote mean ± s.d. P values are as follows: b, i = 0.062 and ii = 0.063. e, i = 1.46 × 10−3 and ii = 3.10 × 10−4. g, Top, i = 7.82 × 10−4, ii = 0.014, iii = 0.088, iv = 1.48 × 10−4, v = 1.47 × 10−3 and vi = 0.016; bottom, i = 3.85 × 10−6, ii = 9.63 × 10−7, iii = 1.31 × 10−6, iv = 8.91 × 10−7, v = 1.15 × 10−5 and vi = 1.56 × 10−7.
Extended Data Figure 5 Differentiation of H. hepaticus-specific T cells in colitis models.
a, b, Naive HH7-2tg T cells were adoptively transferred into H. hepaticus-colonized Rag1−/− mice to induce colitis (n = 7). Data summarize two independent experiments. Representative expression of FOXP3, RORγt, and T-bet (a), and a compilation of frequencies of Treg (FOXP3+), TH17 (FOXP3-RORγt+), TH1 (FOXP3-T-bet+) and TH17/TH1 (FOXP3-RORγt+T-bet+) cells in HH7-2tg donor-derived cells in the LILP of recipient mice was analysed 4 weeks after transfer. c–e, Analysis of H. hepaticus-specific T cell differentiation during C. rodentium-induced colonic inflammation. Data summarize two independent experiments. c, Schematic of experimental design. d, e, Representative flow cytometry plots of FOXP3, RORγt and T-bet expression in total CD4+ and HH-E2 tetramer+ T cells (d) and frequencies of Treg (FOXP3+) and TH17 (FOXP3−RORγt+) cells among HH-E2 tetramer+ T cells (e) in the LILP of C. rodentium-infected (n = 5) and -uninfected mice (n = 5). f–h, Analysis of H. hepaticus-specific T cell differentiation during DSS-induced colitis. Data are a summary of two independent experiments. f, Schematic of experimental design. g, h, Representative flow cytometry plots of FOXP3, RORγt and T-bet expression in total CD4+ and HH-E2 tetramer+ T cells (g) and a compilation of frequencies of Treg (FOXP3+) and TH17 (FOXP3-RORγt+) cells among HH-E2 tetramer+ cells (h) in the LILP of DSS-treated (n = 10) and -untreated mice (n = 10). All statistics were calculated by unpaired two-sided Welch’s t-test. Error bars denote mean ± s.d. P values are indicated on the figure.
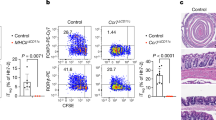
Extended Data Figure 6 Extended characterization of Maf∆Treg, Rorc∆Treg and Gata3∆Treg mice.
a, Expression of c-MAF in the indicated CD4+ T cell subsets in the LILP. b, Incomplete depletion of c-MAF protein in RORγt+ Treg cells in Maf∆Treg mice shown by a representative flow cytometry graph from 3 independent experiments (left), and a compilation of mean fluorescence intensities (MFI) in RORγt− Treg cells and residual RORγt+ Treg cells (right). c, Absolute numbers of indicated CD4+ T cell populations in the LILP of indicated mice. Data are a summary of 3 independent experiments for Rorc∆Treg (n = 7) and littermate controls (n = 7) and 4 independent experiments for Maf∆Treg (n = 11) and littermate controls (n = 8). d, e, Representative flow cytometry plots of FOXP3, RORγt and GATA3 expression in total and FOXP3+ CD4+ T cells (d) and a compilation of frequencies of RORγt+ and RORγt− Treg (FOXP3+) cells and TH17 (FOXP3−RORγt+) cells among total CD4+ T cells (e) in the LILP of Gata3∆Treg (n = 8) and littermate controls (n = 7). Data summarize two independent experiments. f, Absolute numbers of indicated HH-E2 tetramer+ T cell populations in the LILP of indicated mice. Data are a summary of 3 independent experiments for Rorc∆Treg (n = 7) and littermate controls (n = 6) and 4 independent experiments for Maf∆Treg (n = 11) and littermate controls (n = 8). g, Representative flow cytometry plots of TFH markers BCL6 and CXCR5 among total CD4+ and HH-E2 tetramer+ cells from the caecal patch of Maf∆Treg mice and littermate controls (n = 4). h, i, SFB-specific T cells did not adopt the pro-inflammatory TH17–TH1 phenotype or expand in Maf∆Treg mice. Data summarize two experiments, Maf∆Treg (n = 5) and littermate controls (n = 6). Representative flow cytometry plots of FOXP3, RORγt and T-bet expression in total CD4+ and SFB-tetramer+ T cells (h) and absolute number of SFB-tetramer+ cells (i) in the SILP. All statistics were calculated by unpaired two-sided Welch’s t-test. Error bars denote mean ± s.d. P values are indicated on the figure or as follows: c, i = 0.42, ii = 0.73, iii = 6.38 × 10−3, iv = 2.28 × 10−4, v = 7 × 10−11, vi = 7.10 × 10−3, vii = 2.99 × 10−2 and viii = 0.83. e, i = 0.081, ii = 0.102 and iii = 0.16. f, i = 0.65, ii = 0.41, iii = 0.045, iv = 0.12, v = 0.29 and vi = 6.28 × 10−3.
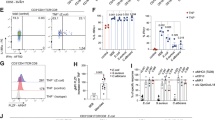
Extended Data Figure 7 Analysis of c-MAF function in RORγt+ iTreg cells.
a–d, Equal numbers of congenic isotype-labelled naive Maf+/+;Foxp3cre (ctrl, CD45.1/45.2) and Maffl/fl;Foxp3cre (CD45.2/45.2) HH7-2tg cells were co-transferred into H. hepaticus-colonized wild-type CD45.1 mice. Cells from the LILP, mLNs and spleen were analysed 15 days after transfer. a, Schematic of experimental design. b, Flow cytometry plot depicting ratio of pooled co-transferred naive T cells before transfer. c, d, Left, ratios of Maf∆Treg versus control HH7-2tg donor-derived cells in the mLNs and spleen (n = 10). Dashed line represents ratio of co-transferred cells before transfer. Right, frequencies of Treg (FOXP3+) and TH17 (FOXP3−RORγt+) cells among donor-derived cells (n = 10). Statistics were calculated by unpaired two-sided Welch’s t-test. Error bars denote mean ± s.d. P values are indicated on the figure. e–h, Isolation of Maf-deficient and -sufficient iTreg cells for RNA-seq through a T cell reconstitution system. Two replicates represent two independent experiments. e, Schematic of experimental design. f, Flow cytometry plots indicating the sorting gates from two independent experiments. g, Flow cytometry plots showing FOXP3 and RORγt expression in sorted FOXP3–YFP+ cells from two independent experiments. h, Gene set enrichment analysis performed on RNA-seq dataset of c-MAF-sufficient versus -deficient iTreg (FOXP3–YFP+) cells (n = 2 independent experiments) with a gene set of 33 RORγt-dependent transcripts identified previously11. i, Top, representative flow cytometry plot of c-MAF expression in TH17 cells (FOXP3−RORγt+) from LILP of control (black) and Maf∆Treg (red) mice. The c-MAF negative population is defined by gating on FOXP3+RORγt− Treg cells from Maf∆Treg mice (solid grey). Bottom, frequency of c-MAF expression in TH17 cells in control (n = 6) and Maf∆Treg (n = 9) mice from 3 independent experiments.
Extended Data Figure 8 Transcriptional profiling of conventional TH17 and H. hepaticus-specific T effector cells.
a–f, RNA-seq was performed on 2 biological replicates of each indicated condition. a, Flow cytometry analysis of HH7-2tg Teff cells from H. hepaticus-colonized mice and conventional IL-23R–GFP+ (predominantly SFB-specific TH17) cells from SFB-colonized mice. GFP+ gates in the bottom panel were used for sorting to perform RNA-seq. b, Principal component analysis of RNA-seq data from sorted cell populations. Coloured dots represent individual samples (n = 2). c, e, f, Differentially expressed genes were calculated in DESeq2 using the Wald test with Benjamini–Hochberg correction to determine the FDR. Genes were considered differentially expressed when FDR < 0.1 and log2 fold change > 1.5. c, Venn diagram depicting differentially expressed genes between indicated comparisons. d, Significantly enriched disease pathways in the set of 149 shared genes upregulated in HH7-2tg Maf∆Treg and HH7-2tg from anti-IL-10RA-treated mice compared to conventional large intestine TH17 cells. P values calculated by ingenuity pathway analysis using Fisher’s exact test. e, Comparison of transcriptomes of H. hepaticus-specific TH17 cells from mice treated with IL-10RA blockade or Maf∆Treg and conventional TH17 cells. Scatter plot depicting log2 fold change of gene expression. Blue, red and purple dots indicate significant differences. f, Heat map depicting the 347 shared genes differentially expressed between pathogenic HH7-2 and conventional TH17 cells (purple dots in e). Data for each condition are the mean of 2 biological replicates. Scale bar represents z-scored variance stabilized data (VSD) counts.
Extended Data Figure 9 STAT3 and TGFβ signal synergistically to promote c-MAF expression.
a, Top, representative flow cytometry plots depicting RORγt and FOXP3 expression in CD4+ T cells in the LILP of mice with a dominant-negative mutant of TGFβ receptors (CD4-dnTGFbRII) and littermate controls (n = 3). Bottom, representative plot of c-MAF expression in FOXP3+ cells from the indicated mice in the top panel. b, Top, representative flow cytometry plots depicting RORγt and FOXP3 expression in CD4+ T cells in the LILP of Stat3fl/fl;Cd4cre and Stat3fl/fl littermate controls (n = 4). Bottom, representative plot of c-MAF staining in FOXP3+ cells from indicated mice in the top panel. c, Mean fluorescence intensity of c-MAF staining in in vitro differentiated CD4+ T cells. Naive CD4+ T cells from wild-type, Stat3fl/fl;Cd4cre and Maffl/fl;Cd4cre mice were activated for 48 h with anti-CD3ε/anti-CD28 antibodies under indicated conditions. Dashed line represents the MFI of c-MAF in Maffl/fl;Cd4cre T cells. Data are from one of two independent experiments.
Extended Data Figure 10 c-MAF-deficient nTreg cells retain suppressive function.
a, Equivalent inhibitory function of nTreg cells from Maf∆Treg and control mice in the in vitro proliferative response of CD4+ T cells (Teff). Three data points are from one of two independent replicates. b, Activity of nTreg cells in the transfer-mediated colitis model. Percentage weight change (left) and colitis histology scores (right) of Rag1−/− mice adoptively transferred with naive T cells alone (n = 8), or naive T cells in combination with nTreg cells from Maf∆Treg (n = 10) or littermate control Foxp3creYFP (n = 9) mice. Data are a summary of two independent experiments. All statistics were calculated by unpaired two-sided Welch’s t-test. Error bars denote mean ± s.d. P values are indicated on the figure.
Supplementary information
Rights and permissions
About this article
Cite this article
Xu, M., Pokrovskii, M., Ding, Y. et al. c-MAF-dependent regulatory T cells mediate immunological tolerance to a gut pathobiont. Nature 554, 373–377 (2018). https://doi.org/10.1038/nature25500
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature25500
This article is cited by
-
Efficacy and safety of gut microbiota-based therapies in autoimmune and rheumatic diseases: a systematic review and meta-analysis of 80 randomized controlled trials
BMC Medicine (2024)
-
Immune microniches shape intestinal Treg function
Nature (2024)
-
The emerging family of RORγt+ antigen-presenting cells
Nature Reviews Immunology (2024)
-
Blimp-1 and c-Maf regulate immune gene networks to protect against distinct pathways of pathobiont-induced colitis
Nature Immunology (2024)
-
Ferritin heavy chain supports stability and function of the regulatory T cell lineage
The EMBO Journal (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.