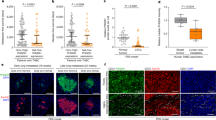
Abstract
Using a functional model of breast cancer heterogeneity, we previously showed that clonal sub-populations proficient at generating circulating tumour cells were not all equally capable of forming metastases at secondary sites1. A combination of differential expression and focused in vitro and in vivo RNA interference screens revealed candidate drivers of metastasis that discriminated metastatic clones. Among these, asparagine synthetase expression in a patient’s primary tumour was most strongly correlated with later metastatic relapse. Here we show that asparagine bioavailability strongly influences metastatic potential. Limiting asparagine by knockdown of asparagine synthetase, treatment with l-asparaginase, or dietary asparagine restriction reduces metastasis without affecting growth of the primary tumour, whereas increased dietary asparagine or enforced asparagine synthetase expression promotes metastatic progression. Altering asparagine availability in vitro strongly influences invasive potential, which is correlated with an effect on proteins that promote the epithelial-to-mesenchymal transition. This provides at least one potential mechanism for how the bioavailability of a single amino acid could regulate metastatic progression.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
04 April 2018
Please see accompanying Erratum (https://doi.org/10.1038/nature26162). In Fig. 3d, the blue bars should be ‘L-asparaginase’ rather than ‘L-asparagine; ‘orthotropic’ should be ‘orthotopic’ in the Extended Data Fig. 6 legend; and the legend to Supplementary Table 4 was repeated for Supplementary Table 5 in the HTML. These and some other minor wording changes have been corrected online.
References
Wagenblast, E. et al. A model of breast cancer heterogeneity reveals vascular mimicry as a driver of metastasis. Nature 520, 358–362 (2015)
Miller, F. R., Miller, B. E. & Heppner, G. H. Characterization of metastatic heterogeneity among subpopulations of a single mouse mammary tumor: heterogeneity in phenotypic stability. Invasion Metastasis 3, 22–31 (1983)
Harrell, J. C. et al. Genomic analysis identifies unique signatures predictive of brain, lung, and liver relapse. Breast Cancer Res. Treat. 132, 523–535 (2012)
Knott, S. R. V. et al. A computational algorithm to predict shRNA potency. Mol. Cell 56, 796–807 (2014)
Zhang, J. et al. Asparagine plays a critical role in regulating cellular adaptation to glutamine depletion. Mol. Cell 56, 205–218 (2014)
Krall, A. S., Xu, S., Graeber, T. G., Braas, D. & Christofk, H. R. Asparagine promotes cancer cell proliferation through use as an amino acid exchange factor. Nat. Commun. 7, 11457 (2016)
Richards, N. G. & Schuster, S. M. Mechanistic issues in asparagine synthetase catalysis. Adv. Enzymol. 72, 145–198 (1998)
Richards, N. G. J. & Kilberg, M. S. Asparagine synthetase chemotherapy. Annu. Rev. Biochem. 75, 629–654 (2006)
Tallal, L. et al. E. coli l-asparaginase in the treatment of leukemia and solid tumors in 131 children. Cancer 25, 306–320 (1970)
Stams, W. A. G. et al. Asparagine synthetase expression is linked with l-asparaginase resistance in TEL-AML1-negative but not TEL-AML1-positive pediatric acute lymphoblastic leukemia. Blood 105, 4223–4225 (2005)
GTEx Consortium et al. Genetic effects on gene expression across human tissues. Nature 550, 204–213 (2017)
Loayza-Puch, F. et al. Tumour-specific proline vulnerability uncovered by differential ribosome codon reading. Nature 530, 490–494 (2016)
Taube, J. H. et al. Core epithelial-to-mesenchymal transition interactome gene-expression signature is associated with claudin-low and metaplastic breast cancer subtypes. Proc. Natl Acad. Sci. USA 107, 15449–15454 (2010)
Gowen, B. G. et al. A forward genetic screen reveals novel independent regulators of ULBP1, an activating ligand for natural killer cells. eLife 4, 1876 (2015)
Al-Baghdadi, R. J. T. et al. Role of activating transcription factor 4 in the hepatic response to amino acid depletion by asparaginase. Sci. Rep. 7, 1272 (2017)
McEarchern, J. A. et al. Invasion and metastasis of a mammary tumor involves TGF-β signaling. Int. J. Cancer 91, 76–82 (2001)
Eden, E., Navon, R., Steinfeld, I., Lipson, D. & Yakhini, Z. GOrilla: a tool for discovery and visualization of enriched GO terms in ranked gene lists. BMC Bioinformatics 10, 48 (2009)
Langmead, B. & Salzberg, S. L. Fast gapped-read alignment with Bowtie 2. Nat. Methods 9, 357–359 (2012)
Anders, S., Pyl, P. T. & Huber, W. HTSeq—a Python framework to work with high-throughput sequencing data. Bioinformatics 31, 166–169 (2015)
Anders, S. & Huber, W. Differential expression analysis for sequence count data. Genome Biol. 11, R106 (2010)
Ruifrok, A. C. & Johnston, D. A. Quantification of histochemical staining by color deconvolution. Anal. Quant. Cytol. Histol. 23, 291–299 (2001)
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet.journal 17, 10 (2011)
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013)
Vizcaíno, et al. 2016 update of the PRIDE database and related tools. Nucleic Acids Res 44, D447–D456 (2016)
Acknowledgements
This work was performed with assistance from CSHL Shared Resources, which are funded, in part, by the Cancer Center Support Grant 5P30CA045508. We thank M. Mosquera, M. Cahn, J. Coblentz, and L. Bianco for support with mouse work; K. Cheng, J. Bourbonniere, D. Hoppe, A. Nourjanova, and R. Puzis for support with histology; E. Hodges and E. Lee for support with next-generation sequencing; and J. Johnson for assistance with HPLC. This work was also performed with the assistance of the Cancer Research UK, Cambridge Institute Proteomics Core Facility. S.R.V.K. is supported by a fellowship from The Hope Funds for Cancer Research. E.W. is supported by a long-term fellowship from the Human Frontier Science Program. L.A.C. is supported by the Susan G. Komen Foundation (SAC110006) and the NCI Breast SPORE program (P50-CA58223-09A1). J.C.H. and C.M.P. are supported by funds from the NCI Breast SPORE program (P50-CA58223-09A1), the Breast Cancer Research Foundation, and the Triple Negative Breast Cancer Foundation. H.G. is supported by a grant from the National Institutes of Health (NIH) (NCI R00 CA194077). Work in the G.P. laboratory is supported by the Institute of Cancer Research, London and a Cancer Research UK grand challenge award (C59824/A25044). G.J.H. is the Royal Society Wolfson Research Professor and is supported by core funding from Cancer Research UK, by a Program Project grant from the NIH (5 P01 CA013106-44), and by a grant from the Department of Defense Breast Cancer Research Program (W81XWH-12-1-0300).
Author information
Authors and Affiliations
Contributions
S.R.V.K., E.W., and G.J.H. conceived the project, supervised research, and wrote the paper. S.R.V.K. and E.W. analysed experiments. S.R.V.K., E.W., S.K., S.Y.K., and M.S. performed in vitro and in vivo experiments. N.E., A.L.G., A.R.M., and S.D. assisted with virus production, shRNA cloning, and library preparation. L.A.C., J.C.H., and C.M.P. assisted with human expression data. J.E.W. performed histological analyses. E.K.P. and C.S.D. assisted with proteomic analyses. L.F. and H.G. assisted with ribosomal profiling analyses. M.W., M.T., and G.P. performed metabolite profiling experiments.
Corresponding author
Ethics declarations
Competing interests
C.M.P. is an equity stock holder of BioClassifier LLC and University Genomics, and has filed a patent on the PAM50 subtyping assay. S.R.V.K., E.W., and G.J.H. are seeking patent protection on the manipulation of asparagine availability for patient benefit in the metastatic setting. The remaining authors declare no competing financial interests.
Additional information
Reviewer Information Nature thanks R. Agami and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Figure 1 Analysis of ASNS expression levels in patient data.
a, Expression level of genes identified as overexpressed in 4T1-T compared with 4T1-E in the primary tumours of patients with different disease subtypes (edges of the box are the 25th and 75th percentiles and error bars extend to the values q3 + w(q3 − q1) and q1 − w(q3 − q1), in which w is 1.5 and q1 and q3 are the 25th and 75th percentiles, which is also true for b, d, and e, ANOVA P < 0.0001). b, Expression level of the same genes in disease-free survivors and patients with relapse to the lung (rank-sum P < 0.01). c, For each gene that was identified in the screen, a prognostic value was calculated using three different datasets. One consisted of gene expression measurements in three patient-matched basal tumour and metastasis pairs (patients A1, A7, and A11). Here genes were classified as correlated with progression if expression was higher in each of the metastases and negatively correlated if expression was higher in each of the primaries. The other two datasets consisted of primary tumour gene-expression profiles with matched outcomes. For the UNC254 patient dataset, the site of relapse was not available and genes were deemed positively correlated with progression if they had significant (Cox P < 0.05) relapse-free survival hazard ratios greater than 1, and negatively correlated if these ratios were significant (Cox P < 0.05) and less than 1. As the UNC855 dataset also had site of relapse information, here both relapse-free and lung relapse-free survival (RFS and LRFS) hazard ratios were used to classify genes as positively or negatively correlated with progression based on the same criteria that were used for the UNC254 data. d, Expression level of ASNS in the primary tumours of patients with different disease subtypes (ANOVA P < 0.0001). e, Expression level of ASNS in the primary tumours of patients with non-specific relapse and relapse to the lymph node, bone, brain, liver, or lung compared with expression levels in patients without relapse to each corresponding site (rank-sum P < 0.005). f, Analysis of ASNS in three additional sets from patients with breast cancer (MDACC, METRABIC, and TCGA). Shown are survival plots and relevant statistics (Cox P < 0.01). g, Analysis of ASNS in the TCGA Pan-Cancer expression data. Shown are survival plots and relevant statistics for the ten non-breast solid tumours represented in the dataset (Cox P < 0.05 for colon, squamous head and neck, renal clear cell, and endometrial cancers). h, Analysis of ASNS across all tumours represented in the TCGA Pan-Cancer dataset (Cox P = 1.5 × 10−12).
Extended Data Figure 2 Primary validation of Asns as a driver of invasion and metastasis.
a, Representative images of the lungs of mice that were intravenously injected with Asns-silenced or -expressing 4T1-T cells as described in Fig. 2a. b, Quantification of Matrigel invasion capacity for Asns-silenced and -expressing 4T1-T cells (n = 3 replicates per cell line). c, Quantification of mCherry-positive 4T1-T cells after roughly 50% of cells were infected with mCherry-expressing constructs harbouring shRNAs targeting Renilla luciferase and Asns. Cells were grown during the 24-h period that the Matrigel invasion assay described in Fig. 2b was being performed (n = 3 replicates per cell line). d, Violet cell-labelling intensity of Asns-silenced and -expressing 4T1-T cells, relative to the initial population. Cells were grown during the 24-h period that the Matrigel invasion assay described in Fig. 2b was being performed (n = 3 replicates per cell line). e, Free amino-acid quantification by HPLC for each amino acid in Asns-expressing and -silenced cells. Shown are the log-fold changes for each amino acid (n = 3 replicates per cell line). f, Quantification of mCherry-positive 4T1-T cells after roughly 50% of cells were infected with mCherry-expressing constructs harbouring shRNAs targeting Renilla luciferase and Asns. After infection, cells were grown in medium supplemented with l-asparagine or d-asparagine and mCherry percentages were measured at 48 and 96 h (n = 3 replicates per cell line). g, Quantification of Matrigel invasion for Asns-silenced and -expressing cells when assayed in medium supplemented with and without l-asparagine (n = 3 invasion chambers per cell line).
Extended Data Figure 3 Secondary validation of Asns as a driver of invasion and metastasis.
a, Volume measurements of tumours resulting from orthotopic injection of Asns-silenced and -expressing parental 4T1 cells (n = 10 mice per cell line, edges of the box are the 25th and 75th percentiles and error bars extend to the values q3 + w(q3 − q1) and q1 − w(q3 − q1), in which w is 1.5 and q1 and q3 are the 25th and 75th percentiles, which is also the case for b–g). b, Quantification of lung metastases corresponding to the tumours described in a (rank-sum P < 0.002). c, Volume measurements of tumours resulting from orthotopic injection of parental 4T1 cells with basal (Empty) or enforced expression of Asns (n = 10 mice per cell line). d, Quantification of lung metastases corresponding to the tumours described in c (rank-sum P < 5.0 × 10−5). e, Average diameters of the metastases of each mouse described in d (rank-sum P < 0.001). f, Volume measurements for tumours resulting from orthotopic injection of MDA-MB-231 cells with basal (Empty) or enforced expression of ASNS (n = 10 mice per cell line). g, Quantification of lung metastases corresponding to the tumours described in f (rank-sum P < 0.005). h, Quantification of Matrigel invasion for the MDA-MB-231-derived cell lines described in f (n = 3 invasion chambers per cell line). i, Representative images of the collection wells for the invasion assays described in h. See Source Data.
Extended Data Figure 4 Primary validation that extracellular asparagine availability affects invasion and metastasis.
a, HPLC quantification of cellular free amino-acid percentages for parental 4T1 cells when the medium is supplemented with each of the NEAAs lacking in the DMEM culture medium (n = 3 replicates per cell line). b, Quantification of MDA-MB-231 Matrigel invasion rates under the same conditions as described in Fig. 3a (n = 5 invasion chambers per condition, rank-sum P < 0.001). c, HPLC quantification of cellular free amino-acid percentages for MDA-MB-231 cells when cultured in the medium conditions described in a (n = 3 replicates per cell line). d, Violet cell-labelling intensity of parental 4T1 cells when grown in asparagine-lacking or -supplemented medium for the same period that the Matrigel invasion assay described in Fig. 3a was being performed (n = 3 replicates per cell line). e, Violet cell-labelling intensity of MDA-MB-231 cells when grown in asparagine-lacking or -supplemented medium for the same period that the Matrigel invasion assay described in b was being performed (n = 3 replicates per cell line).
Extended Data Figure 5 Secondary validation that extracellular asparagine availability affects invasion and metastasis.
a, Tumour volumes resulting from the orthotopic injection of parental 4T1 cells. Half of the mice received l-asparaginase and the other half received an equivalent volume of PBS at the same injection rate (n = 10 mice per condition, edges of the box are the 25th and 75th percentiles and error bars extend to the values q3 + w(q3 − q1) and q1 − w(q3 − q1), in which w is 1.5 and q1 and q3 are the 25th and 75th percentiles, which is also the case for b–g). b, Quantification of lung metastases detected in the animals described in a (rank-sum P < 0.001). c, Representative H&E-stained lung sections as described in b. d, Quantification of the lung metastases described in Fig. 3b, in which Asns-silenced and -expressing 4T1-T cells were injected into mice. Half of the mice received l-asparaginase and the other half received an equivalent volume of PBS at the same injection rate (n = 10 mice per condition, rank-sum P < 0.0005 for l-asparaginase versus control for each line and for Asns-silenced versus -unsilenced cells in each drug condition). e, Tumour volumes corresponding to the lung metastases described in d (rank-sum P < 0.005 for Asns-silenced versus -expressing cells in l-asparaginase-treated mice). f, Lung metastases resulting from the orthotopic injection of ASNS-silenced and -expressing MDA-MB-231 cells and subsequent treatment of the injected animals with l-asparaginase or PBS (n = 10 mice per cell line, rank-sum P < 0.05 for ASNS-silenced versus -expressing cells in both conditions and for silenced cells in treated versus untreated mice). g, Tumour volumes corresponding to the mice described in f (rank-sum P < 0.05 for Asns-silenced versus -expressing cells under both treatments and for PBS versus l-asparaginase-treated animals for each cell line). See Source Data.
Extended Data Figure 6 Tertiary validation that extracellular asparagine availability affects invasion and metastasis.
a, Asparagine content in the serum free amino-acid pool, for mice fed 0%, 0.6%, or 4% asparagine diets (n = 5 mice per diet, edges of the box are the 25th and 75th percentiles and error bars extend to the values q3 + w(q3 − q1) and q1 − w(q3 − q1), in which w is 1.5 and q1 and q3 are the 25th and 75th percentiles, which is also the case for b, d, e and h, rank-sum P < 0.05 between each diet). b, Volumes of orthotopic tumours corresponding to the lung metastases described in Fig. 3c, in which Asns-silenced and -expressing 4T1-T cells were orthotopic injected into mice fed with 0%, 0.6%, and 4% asparagine diets (n = 10 mice per condition). c, Representative images of the lung metastases described for Fig. 3c, which also correspond to the mice described in b. d, Volumes of tumours resulting from the orthotopic injection of parental 4T1 cells into mice fed with 0%, 0.6%, or 4% asparagine diets (n = 10 mice per diet). e, Quantification of metastases in the lungs of the animals described in d (rank-sum P < 0.05 for mice receiving 0% versus 0.6% and 0% versus 4% diets). f, Representative images of H&E-stained sections of the lungs described in e. g, Relative expression of Asns in the mammary gland, serum, and lungs of mice treated with l-asparaginase or PBS, as measured by qPCR with two primer pairs P1 and P2 (n = 3 per condition). h, Transcripts per million (TPM) expression measurements for ASNS in human breast, lung, and whole-blood samples (n > 114 for each tissue, rank-sum P < 2.8 × 10−37 for blood versus breast and blood versus lung). See Source Data.
Extended Data Figure 7 Primary validation that asparagine availability regulates EMT.
a, Protein-level changes between Asns-silenced and -expressing cells when genes are stratified by transcription-level changes (top and bottom 10% of genes based on log-fold change in Asns-silenced cells, gene-up and -down, respectively) and asparagine content (top and bottom 10% of genes based on asparagine content, Asp-high and -low, respectively), edges of the box are the 25th and 75th percentiles and error bars extend to the values q3 + w(q3 − q1) and q1 − w(q3 − q1), in which w is 1.5 and q1 and q3 are the 25th and 75th percentiles, which is also the case for d and e, rank-sum P < 5.0 × 10−24 for both individual variables, and rank-sum P < 0.005 for interacting variables). b, Amino acid enrichment analysis of downregulated genes (bottom 25% based on log-fold change) on the basis of RNA and protein levels in Asns-expressing versus -silenced 4T1-T cells. Negative correlations indicate the amino acid is depleted in the downregulated genes, whereas positive correlations indicate the amino acid is enriched. For protein minus RNA level expression changes, amino acids with positive correlations are enriched in proteins in which depletion levels exceed what is predicted by corresponding RNA changes. Negative correlations indicate the amino acid is enriched in proteins in which depletion levels are less than what is predicted by corresponding RNA changes (rank-sum P < 1.0 × 10−5 for asparagine in protein and protein–RNA). c, Amino-acid enrichment in mouse and human EMT-up proteins (rank-sum P < 0.01 for both human and mouse). d, Position 15 asparagine codon enrichment in ribosome protected fragments from PC-3 cells grown with and without l-asparaginase, when all genes or only EMT-up genes are analysed (outliers were not plotted to improve interpretability, which is also the case for e, rank-sum P < 0.05 for EMT-up versus all genes in both untreated and l-asparaginase-treated cells). e, Increase in asparagine codon representation in ribosome protected fragments, when PC-3 cells are grown in l-asparaginase (relative to without), and all genes or EMT-up genes are analysed (rank-sum P < 0.05). See Source Data.
Extended Data Figure 8 Conservation of asparagine enrichment in EMT promoting proteins.
Asparagine enrichment analysis of EMT-promoting protein orthologues in the 126 species listed in the Orthologous MAtrix database that harbour at least 10 orthologues (sign-rank P < 1.0 × 10−13 for all species and rank-sum P < 9.0 × 10−9 for mammals versus other species).
Extended Data Figure 9 Secondary validation that asparagine availability regulates EMT.
a, Transcription-level changes in EMT-up and -down genes that occur in response to Asns silencing in 4T1-T cells (n = 2 replicates per condition, edges of the box are the 25th and 75th percentiles and error bars extend to the values q3 + w(q3 − q1) and q1 − w(q3 − q1), in which w is 1.5 and q1 and q3 are the 25th and 75th percentiles, which is also the case for b–e, h and i, rank-sum P < 0.001 for EMT-up genes, DESeq false discovery rate < 0.05 for Twist1 and Cdh1). b, Transcription-level changes in EMT-up and -down genes that occur in response to the medium of Asns-silenced 4T1-T cells being supplemented with l-asparagine (n = 2 replicates per condition, rank-sum P < 0.005 for EMT-up genes). c, Gene expression changes in EMT-up and -down genes that result from ATF4 knockout in near haploid KBM-7 chronic myelogenous leukaemia (HAP-1 cells, rank-sum P < 0.05 for EMT-up genes). d, Gene expression changes in EMT-up and -down genes, which result in the liver cells of homozygous ATF4-deleted mice when treated with l-asparaginase (rank-sum P < 0.05 for EMT-down genes in wild-type mice treated with l-asparaginase (WT+L-asp) mice, and both EMT-up and -down genes in ATF4 mice treated with l-asparaginase (ATF4+L-asp) mice). e, Volumes of tumours resulting from orthotopic injection of Tgf-β-silenced and -expressing 4T1-T cells (n = 10 mice per cell line). f, Percentage of Twist1-positive regions based on IHC staining of sections from tumours described in e (n = 5 tumour sections per cell line, rank-sum P < 0.01). g, Percentage of Cdh1-positive regions based on IHC staining of sections from tumours described in e (n = 5 tumour sections per cell line, rank-sum P < 0.01). h, Quantification of metastases resulting from the tumours described in e (rank-sum P < 0.05). i, Quantification of metastases resulting from intravenous injection of Tgf-β-silenced and -expressing cells (n = 10 mice per cell line, rank-sum P < 0.05). See Source Data.
Extended Data Figure 10 Tertiary validation that asparagine availability regulates EMT.
a, Representative H&E-stained sections of the tumours described in Fig. 2c, in which Asns-silenced and -expressing 4T1-T cells were orthotopically injected into NSG mice. b, Images of cultured cells after they were isolated from the tumours and lungs of mice injected orthotopically with Asns-silenced and -expressing 4T1-T cells. c, Relative Twist1 expression, as measured by qPCR, which were sorted from the tumours and lungs of mice injected orthotopically with Asns-silenced and -expressing 4T1-T cells (n = 3 tumours and lungs per cell line). d, Relative Cdh1 expression, as measured by qPCR, in the tumours and lungs described in c (n = 3 tumours and lungs per cell line).
Extended Data Figure 11 Quaternary validation that asparagine availability regulates EMT.
a, Representative images of IHC staining for Twist1 and Cdh1 on sections from lungs described in Fig. 4e, in which mice were injected orthotopically with Asns-silenced and -expressing 4T1-T cells. b, Quantification of all Twist1 stainings, described in Fig. 4e and a (n = 5 tumour sections and n > 5 lung metastases, edges of the box are the 25th and 75th percentiles and error bars extend to the values q3 + w(q3 − q1) and q1 − w(q3 − q1), in which w is 1.5 and q1 and q3 are the 25th and 75th percentiles, which is also the case for c–g, rank-sum P < 0.01 and P < 0.05 for Asns-silenced versus -expressing tumours and metastases, respectively). c, Quantification of all Cdh1 stainings, described in b (n = 5 tumour sections and n = 9 lung metastases, rank-sum P < 0.01 and P < 0.05 for Asns-silenced versus -expressing tumours and metastases, respectively). d, Quantification of Twist1-positive regions in the tumours resulting from orthotopic injection of Asns-expressing and -silenced 4T1-T cells into animals treated with PBS or l-asparaginase (n = 5 tumour sections per condition, rank-sum P < 0.01 for Asns-silenced versus -unsilenced cells and rank-sum P < 0.05 for each cell line in treated versus untreated mice). e, Quantification of Cdh1-positive regions in the tumours described in d (n = 5 tumour sections per condition, rank-sum P < 0.01 for Asns-silenced versus -unsilenced cells and rank-sum P < 0.05 for each cell line in treated versus untreated mice). f, Quantification of Twist1-positive regions in tumours resulting from orthotopic injection of Asns-expressing and -silenced cells into mice fed a 0%, 0.6%, or 4% asparagine diet (n = 5 tumour sections per condition, rank-sum P < 0.01 between Asns-silenced and -expressing cells and between diets). g, Quantification of Cdh1-positive regions in the tumours described in f (n = 5 tumour sections per condition, rank-sum P < 0.01 between Asns-silenced and -expressing cells and between diets).
Supplementary information
Supplementary Table 1
Genes identified as over-expressed in 4T1-T. Genes identified as over-expressed in 4T1-T cells as compared to 4T1-E cells by differential expression analysis of RNA sequencing data. For each line, cells were grown in vitro and two biological replicates were processed for RNA sequencing. Genes were identified in 4T1-T using a fold-change cutoff of 2 and a DESeq FDR cut-off of 0.05. (XLSX 56 kb)
Supplementary Table 2
Gene Ontology analysis of genes over-expressed in 4T1-T. The genes listed in Supplementary Table 1 were analysed to identify enriched cellular processes, functions and components. Each Gene Ontology term that was associated with the genes in the subset was compared in its number of associates within the list to its number of associations to the entire murine Refseq gene list via a hypergeometic test. Ontology terms that were enriched with a q-value < 0.05 are listed. (XLSX 34 kb)
Supplementary Table 3
Raw RNAi screening data and shRNA depletion scores. Represented are the Illumina sequence reads that were assigned to each of ~6 shRNAs targeting protein coding members of the genes listed in Supplementary Table 1, both in the pre-injection infected cell population and in the cell populations that were removed from the lungs of mice in the in vivo screen or were removed from the matrigel invaded cells in the in vitro screen. Also listed are the log-fold enrichment and depletion scores of each shRNA and false-discovery rates as assigned using an Emperical-bayes moderated t-test. (XLSX 188 kb)
Supplementary Table 4
Expression changes induced by shRNA silencing or cDNA induced over-expression. Relative expression values for each cell line produced using shRNAs for silencing or cDNAs for enforcing expression. (XLSX 38 kb)
Supplementary Table 5
Amino acid composition of serum with and without ʟ-asparaginase treatment. 4T1-T cells harbouring the non-targeting Renilla shRNA were injected into immunocompromised mice. Five mice each were either injected with 60 U l -asparaginase or PBS 5 days per week. After blood collection and serum isolation, free amino acids were quantified using High Performance Liquid Chromatography (HPLC) and a fluorometric detector. (XLSX 52 kb)
Supplementary Table 6
Mouse orthologues of human genes that were identified as differentially expressed during EMT. Listed are genes that are up- or down-regulated when cells were enforced for the expression of Tgf-ß, Twist, Gsc or Snail or when E-cadherin was silenced (EMT-up and –down genes, respectively). EMT-up genes whose protein-level log-fold changes in Asns-silenced cells fell within the bottom or top 10% are annotated as Down-regulated and Up-regulated, respectively. (XLSX 14 kb)
Rights and permissions
About this article
Cite this article
Knott, S., Wagenblast, E., Khan, S. et al. Asparagine bioavailability governs metastasis in a model of breast cancer. Nature 554, 378–381 (2018). https://doi.org/10.1038/nature25465
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature25465
This article is cited by
-
Metabolism of asparagine in the physiological state and cancer
Cell Communication and Signaling (2024)
-
Selective enhanced cytotoxicity of amino acid deprivation for cancer therapy using thermozyme functionalized nanocatalyst
Journal of Nanobiotechnology (2024)
-
AURKA emerges as a vulnerable target for KEAP1-deficient non-small cell lung cancer by activation of asparagine synthesis
Cell Death & Disease (2024)
-
Amino acid metabolism in tumor biology and therapy
Cell Death & Disease (2024)
-
Glutamine analogs for pancreatic cancer therapy
Nature Cancer (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.