Abstract
The spreading of pathology within and between brain areas is a hallmark of neurodegenerative disorders. In patients with Alzheimer’s disease, deposition of amyloid-β is accompanied by activation of the innate immune system and involves inflammasome-dependent formation of ASC specks in microglia. ASC specks released by microglia bind rapidly to amyloid-β and increase the formation of amyloid-β oligomers and aggregates, acting as an inflammation-driven cross-seed for amyloid-β pathology. Here we show that intrahippocampal injection of ASC specks resulted in spreading of amyloid-β pathology in transgenic double-mutant APPSwePSEN1dE9 mice. By contrast, homogenates from brains of APPSwePSEN1dE9 mice failed to induce seeding and spreading of amyloid-β pathology in ASC-deficient APPSwePSEN1dE9 mice. Moreover, co-application of an anti-ASC antibody blocked the increase in amyloid-β pathology in APPSwePSEN1dE9 mice. These findings support the concept that inflammasome activation is connected to seeding and spreading of amyloid-β pathology in patients with Alzheimer’s disease.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Heneka, M. T., Kummer, M. P. & Latz, E. Innate immune activation in neurodegenerative disease. Nat. Rev. Immunol. 14, 463–477 (2014)
Lambert, J. C. et al. Meta-analysis of 74,046 individuals identifies 11 new susceptibility loci for Alzheimer’s disease. Nat. Genet. 45, 1452–1458 (2013)
Yokoyama, J. S. et al. Association between genetic traits for immune-mediated diseases and Alzheimer disease. JAMA Neurol. 73, 691–697 (2016)
Gjoneska, E. et al. Conserved epigenomic signals in mice and humans reveal immune basis of Alzheimer’s disease. Nature 518, 365–369 (2015)
Zhang, B. et al. Integrated systems approach identifies genetic nodes and networks in late-onset Alzheimer’s disease. Cell 153, 707–720 (2013)
Raj, T. et al. Polarization of the effects of autoimmune and neurodegenerative risk alleles in leukocytes. Science 344, 519–523 (2014)
Karch, C. M., Cruchaga, C. & Goate, A. M. Alzheimer’s disease genetics: from the bench to the clinic. Neuron 83, 11–26 (2014)
Musiek, E. S. & Holtzman, D. M. Three dimensions of the amyloid hypothesis: time, space and ‘wingmen’. Nat. Neurosci. 18, 800–806 (2015)
Jucker, M. & Walker, L. C. Self-propagation of pathogenic protein aggregates in neurodegenerative diseases. Nature 501, 45–51 (2013)
Jack, C. R. Jr et al. Tracking pathophysiological processes in Alzheimer’s disease: an updated hypothetical model of dynamic biomarkers. Lancet Neurol. 12, 207–216 (2013)
Heneka, M. T., Golenbock, D. T. & Latz, E. Innate immunity in Alzheimer’s disease. Nat. Immunol. 16, 229–236 (2015)
Heneka, M. T. et al. NLRP3 is activated in Alzheimer’s disease and contributes to pathology in APP/PS1 mice. Nature 493, 674–678 (2013)
Lu, A. et al. Unified polymerization mechanism for the assembly of ASC-dependent inflammasomes. Cell 156, 1193–1206 (2014)
Masumoto, J. et al. ASC, a novel 22-kDa protein, aggregates during apoptosis of human promyelocytic leukemia HL-60 cells. J. Biol. Chem. 274, 33835–33838 (1999)
Cai, X. et al. Prion-like polymerization underlies signal transduction in antiviral immune defense and inflammasome activation. Cell 156, 1207–1222 (2014)
Franklin, B. S. et al. The adaptor ASC has extracellular and ‘prionoid’ activities that propagate inflammation. Nat. Immunol. 15, 727–737 (2014)
Baroja-Mazo, A. et al. The NLRP3 inflammasome is released as a particulate danger signal that amplifies the inflammatory response. Nat. Immunol. 15, 738–748 (2014)
Dick, M. S., Sborgi, L., Rühl, S., Hiller, S. & Broz, P. ASC filament formation serves as a signal amplification mechanism for inflammasomes. Nat. Commun. 7, 11929 (2016)
Tomiyama, T. et al. A new amyloid β variant favoring oligomerization in Alzheimer’s-type dementia. Ann. Neurol. 63, 377–387 (2008)
Borchelt, D. R. et al. Familial Alzheimer’s disease-linked presenilin 1 variants elevate Aβ1–42/1–40 ratio in vitro and in vivo. Neuron 17, 1005–1013 (1996)
Scheuner, D. et al. Secreted amyloid β-protein similar to that in the senile plaques of Alzheimer’s disease is increased in vivo by the presenilin 1 and 2 and APP mutations linked to familial Alzheimer’s disease. Nat. Med. 2, 864–870 (1996)
Youm, Y.-H. et al. Canonical Nlrp3 inflammasome links systemic low-grade inflammation to functional decline in aging. Cell Metab. 18, 519–532 (2013)
Meyer-Luehmann, M. et al. Exogenous induction of cerebral β-amyloidogenesis is governed by agent and host. Science 313, 1781–1784 (2006)
Elinav, E. et al. NLRP6 inflammasome regulates colonic microbial ecology and risk for colitis. Cell 145, 745–757 (2011)
Nuvolone, M., Sorce, S., Schwarz, P. & Aguzzi, A. Prion pathogenesis in the absence of NLRP3/ASC inflammasomes. PLoS ONE 10, e0117208 (2015)
Yamanaka, M. et al. PPARγ/RXRα-induced and CD36-mediated microglial amyloid-β phagocytosis results in cognitive improvement in amyloid precursor protein/presenilin 1 mice. J. Neurosci. 32, 17321–17331 (2012)
Fernandes-Alnemri, T. & Alnemri, E. S. Assembly, purification, and assay of the activity of the ASC pyroptosome. Methods Enzymol. 442, 251–270 (2008)
Fernandes-Alnemri, T. et al. The pyroptosome: a supramolecular assembly of ASC dimers mediating inflammatory cell death via caspase-1 activation. Cell Death Differ. 14, 1590–1604 (2007)
Wahle, T. et al. GGA1 is expressed in the human brain and affects the generation of amyloid β-peptide. J. Neurosci. 26, 12838–12846 (2006)
Kumar, S. et al. Extracellular phosphorylation of the amyloid β-peptide promotes formation of toxic aggregates during the pathogenesis of Alzheimer’s disease. EMBO J. 30, 2255–2265 (2011)
Rostagno, A. & Ghiso, J. Isolation and biochemical characterization of amyloid plaques and paired helical filaments. Curr. Protoc. Cell. Biol. 44, 3.33.1–3.33.33 (2009)
Fritschi, S. K. et al. Aβ seeds resist inactivation by formaldehyde. Acta Neuropathol. 128, 477–484 (2014)
Jäger, S . et al.. α-secretase mediated conversion of the amyloid precursor protein derived membrane stub C99 to C83 limits Aβ generation. J. Neurochem. 111, 1369–1382 (2009)
Acknowledgements
This work was funded by the Deutsche Forschungsgemeinschaft through the Cluster of Excellence “Immunosensation” (to M.T.H., E.L., M.G. and B.S.F.), the Clinical Research Group (KFO177; to M.T.H., E.L. and J.W.), the SFB670 (E.L.), grant WA1477/6 (J.W.), ERC InflammAct (E.L.), ERC PLAT-IL-1 (B.S.F.), the ERA-NET consortium TracInflam (M.T.H.) and JPND consortium InCure (M.T.H.).
Author information
Authors and Affiliations
Contributions
C.V., S.K., B.S.F., T.D., R.B., D.T., A.V.-S., S.S., F.S., A.G., D.R., M.G., M.B., E.L. and M.T.H. performed experiments and analysed data. S.K., M.P.K., E.G., D.T.G., J.W. and M.T.H. designed the study and wrote the paper. Results were discussed and the manuscript was commented on by all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer Information Nature thanks R. Ransohoff, G. Nunez and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Figure 1 Characteristics of microglial ASC-speck formation in mice and humans.
a, b, Immunohistochemistry of the microglial marker CD11b and ASC in sections derived from brains of patients with AD (a) or controls without dementia (Con) (b) (top), and omitting either the primary (middle) or secondary (bottom) antibody. c, Percentage of ASC specks detected by immunohistochemistry in- and outside of microglial cells in sections derived from brains of patients with AD (AD-in and AD-ex) and age-matched controls without dementia (Con-in and Con-ex). n = 10 human cases. One-way ANOVA, Tukey test, ***P < 0.0001. d, Percentage of ASC specks detected by immunohistochemistry in- and outside (-in and -ex, respectively, on y axis) of microglial cells in sections derived from the hippocampus of APP/PS1 mice at the indicated ages given in months (m). n = 10 mice. Two-tailed Student’s t-test, ***P < 0.0001. e, Number of ASC specks bound to Aβ deposits per visual field observed. n = 5 mice. Two-tailed Student’s t-test, *P = 0.0216. f, ASC expression in brain lysates derived from wild type (WT) and APP/PS1 transgenic mice at 4, 8, 12 and 24 months of age. g, Hippocampal sections of 8-month-old wild-type (WT), Asc−/−, APP/PS1 and APP/PS1;Asc−/− mice were stained for the microglial marker CD11b and ASC in the presence of primary and secondary antibodies (left) or in the absence of the respective primary antibody (right). h, Hippocampal sections of 8-month-old wild-type, Asc−/−, APP/PS1 and APP/PS1;Asc−/− mice were stained for the microglial marker CD11b and ASC in the presence of the primary and secondary antibodies (left, same panels as shown in g) or in the absence of the respective secondary antibody (right). Experiments were performed independently two (f) or three (a, b, g, h) times or were performed once (c–e). Scale bars, 15 μm (a, b, g, h). Data are mean ± s.e.m. (c–e).
Extended Data Figure 2 Experimental ASC-speck formation in primary mouse microglia and human THP-1 cells.
a, Flow cytometry analysis of conditioned medium from primary mouse microglia using 2- and 6-μm fluorescent beads for gating ASC specks. b, Confocal imaging of primary mouse microglia exposed to control solvent (Con), LPS alone or LPS followed by nigericin (LPS + Nig) or ATP (LPS + ATP). Cells were stained with anti-ASC antibody followed by an Alexa Fluor 488 conjugate. Arrowheads show extracellular ASC specks. Scale bars, 24 μm; insets are 4× (bottom left two panels) and 8× (bottom right two panels) magnifications of the areas shown in the squares. c, Gating strategy for the detection of ASC specks in cell-free supernatants of untreated (−) or LPS-primed, nigericin-activated (10 μM) (LPS + Nig) ASC–mCerulean-expressing THP-1 cells. d, Confocal imaging of LPS-primed, nigericin-treated THP-1 cells showing green fluorescent ASC specks in the extracellular space. Scale bars, 38 μm (left) and 8 μm (inset). e, Quantification of extracellular specks in cell-free supernatants. n = 3 technical replicates, data mean ± s.d., representative of two independent experiments. f–j, Images of THP-1 cells in the absence of TAMRA–Aβ1–42 (f), showing TAMRA–Aβ surface binding and early incorporation (g), subsequent upregulation of ASC (green) (h), early ASC-speck formation in a cell, which has incorporated TAMRA–Aβ1–42 (i) and ASC specks formed within a cell (j). Scale bars, 5 μm (f–j). Experiments were independently performed three (a–c, f–j) or two (d, e) times.
Extended Data Figure 3 Qualitative and quantitative description of Aβ–ASC binding.
a, Experimental design and timeline: 3 h LPS and 1 h nigericin induces a highly inflammatory form of programmed cell death (pyroptosis) causing release of ASC specks. Supernatants containing ASC specks were subsequently incubated with Aβ1–42 for 6 h and then analysed by flow cytometry. b, Top, immunoprecipitation and immunoblot detection of ASC in unstimulated, immune activated wild-type (WT) and Asc−/− macrophages. ASC monomer detection is restricted to supernatants of immune activated, ASC-competent wild-type cells and absent in unstimulated wild-type cells or Asc−/− macrophages. Bottom, immunoprecipitation of ASC followed by immunoblot detection of Aβ as in the top panel. ASC-bound Aβ is exclusively detected in supernatants derived from immune activated wild-type macrophages but not from unstimulated wild-type or Asc−/− cells. c, Top, immunoprecipitation of ASC and immunoblot detection of ASC in unstimulated, immune activated wild-type and Asc−/− microglia. ASC monomer detection is restricted to supernatants of immune-activated ASC-competent wild-type cells and absent in unstimulated wild-type or Asc−/− microglia. Bottom, immunoprecipitation of ASC followed by immunoblot detection of Aβ as in the top panel. ASC-bound Aβ is exclusively detected in supernatants derived from immune-activated wild-type microglia but not from unstimulated wild-type or Asc−/− cells. d, Gating strategy and control group: gated on debris to exclude remaining cells and larger particles. Recombinant ASC labelled with CFP and Aβ1–42 labelled with TAMRA signal in independent quadrants (Q1 and Q3). When incubated together, the molecules accumulate and signal in Q2. n = 3 biologically independent samples. One-way ANOVA, Tukey test, ***P < 0.0001. e, Experimental groups: ASC–mCerulean-expressing, immortalized macrophages show simular results. Asc−/− macrophages show no ASC-speck formation and no Aβ1–42 accumulation. Con, control; activ., activated. n = 3 biologically independent samples. One-way ANOVA, Tukey test, activated + Aβ1–42 versus control: ***P = 0.0002; activated + Aβ1–42 versus control + Aβ1–42: ***P = 0.0009; activated + Aβ1–42 versus activated: ***P = 0.0002. f, Flow cytometry quantification of ASC–TAMRA-labelled Aβ1–40 after immune stimulation of microglia. Experimental design and timeline: 3 h LPS and 1 h ATP induce a highly inflammatory form of programmed cell death (pyroptosis) through which ASC specks are released. Supernatants containing ASC specks were subsequently incubated with Aβ1–40 for 6 h and analysed by FACS. Experiments were performed independently three times (b, c). Data are mean ± s.e.m. (d, e).
Extended Data Figure 4 An immunoprecipitation and enzymatic cleavage-based method for the generation of highly pure ASC specks.
a, Schematic of ASC-speck formation upon inflammasome assembly and purification of ASC specks via immunoprecipitation and enzymatic cleavage. Immortalized, ASC-deficient macrophages were transduced with a construct containing ASC–mCerulean with a Flag-tag, and a precision site for the TEV protease between ASC and mCerulean. Inflammasome activation in these cells results in ASC aggregation and formation of an ASC speck. The ASC speck containing the mCerulean and Flag-tag can be immunopurified, followed by proteolytic cleavage of the mCerulean–Flag-tag by the TEV protease to generate pure ASC specks. b, Immunoblotting analysis of ASC specks isolated from ASC–mCerulean–Flag macrophages before (-) or after immunoprecipitation using anti-GFP antibodies followed by enzymatic cleavage using TEV protease. IP, immunoprecipitation; IB, immunoblot. c, Confocal imaging after immunostaining of ASC and GFP in untreated versus immunoprecipitated and TEV-treated ASC specks. Scale bars, 3.8 μm (top), 6.3 μm (middle) and 9 μm (bottom). d, Flow cytometry analysis of anti-ASC–Alexa Fluor 488 and anti-GFP–Alexa Fluor 647 double-stained ASC specks isolated from ASC–mCerulean–Flag macrophages (top). Anti-mouse IgGs conjugated to Alexa Fluor 488 or 647 were used as controls (bottom). Experiments were performed independently four times (b–d).
Extended Data Figure 5 ASC specks increase the tendency of Aβ peptides to aggregate in a time- and concentration-dependent manner.
a, ThT fluorescence assay of ASC specks and Aβ1–40 co-incubation showing cross-seeding potency of ASC specks in a time-dependent manner. b, Western blot detection of time-dependent, ASC-speck-induced aggregation of Aβ1–40. Co-incubation of Aβ1–40 with ASC specks increases the tendency to aggregate and increased the formation of high-molecular-weight Aβ oligomers and protofibrils. c, Quantification at the indicated time points. n = 4 biologically independent samples. Two-tailed Student’s t-test, 6 h: ***P = 0.0002; 4 h and 24 h: ***P < 0.0001. d, Western blot analysis of Aβ1–42 co-incubated with increasing concentrations of ASC specks (0–1.75 μM) at 0 and 24 h. e, Western blot analysis of Aβ1–40 co-incubated with increasing concentrations of ASC specks (0–1.75 μM) at 0 and 24 h. For both Aβ peptide variants, co-incubation with ASC specks increased the tendency to aggregate and increased the formation of high-molecular-weight oligomers. Note that for Aβ1–42 the increase in oligomer formation is paralleled by a reduction in the levels of Aβ monomers and dimers. f, Electron microscopy of Aβ1–42, ASC and ASC–Aβ1–42 aggregation after 96 h of incubation. Scale bars, 200 nm. g, Confirmation of ASC-speck-increased Aβ1–40 and Aβ1–42 aggregation by turbidity assay. n = 3 biologically independent samples. h, ThT fluorescence assay of ASC specks and Aβ42–1 co-incubation showing no cross-seeding potential of ASC specks for the reversed peptide. i, ThT fluorescence assay of Aβ1–40 co-incubation with ASC specks and two different concentrations with bovine serum albumin (BSA). Although ASC specks cross-seed Aβ1–40 in a time-dependent manner, neither 0.22 μM nor 0.66 μM BSA affected Aβ1–40 aggregation. Experiments were performed independently four (a, b, d, e) or three (f, h, i) times. Data are mean ± s.e.m. (c, g).
Extended Data Figure 6 The ASC PYD domain is critical for Aβ cross-seeding.
a, Immunoblots were probed for Aβ using antibody 82E1, revealing time-dependent aggregation of Aβ1–40. Co-incubation of Aβ1–40 with recASC specks promotes aggregation and increases the formation of high-molecular-weight Aβ oligomers. Notably, formation of intermediate Aβ oligomers (from 28 to 62 kDa bands) is observed and increased with incubation time. b, Immunoblot for ASC, revealing time-dependent auto-aggregation. c, Immunoblots were probed for Aβ using antibody 82E1, revealing time-dependent aggregation of Aβ1–42. Co-incubation of Aβ1–42 with recASC specks promotes aggregation and increases the formation of high-molecular-weight Aβ oligomers. Notably, formation of intermediate Aβ oligomers (from 28 to 62 kDa bands) is observed and increased with incubation time. d, Immunoblots were probed for ASC, revealing time-dependent auto-aggregation. e, Recombinant mutant ASC was generated by introducing point mutations at residues K21A, K22A and K26A in the ASC PYD domain. Purified recombinant mutant ASC specks were used for the Aβ aggregation assay. Immunoblots were probed for Aβ using antibody 82E1, revealing time-dependent aggregation of Aβ. Co-incubation of recombinant mutant ASC specks (recASC; K21A, K22A and K26A) failed to increase high-molecular-weight Aβ oligomer levels. No intermediate Aβ oligomers (from 28 to 62 kDa bands) are seen in Aβ supplemented with recombinant mutant ASC specks. f, Immunoblots were stained for ASC revealing no auto-aggregation of recombinant mutant ASC specks. g, Purified recombinant mutant ASC generated by introducing point mutations at residues D134R and Y187E in the ASC CARD domain were used for the Aβ aggregation assay. Immunoblot was probed for Aβ using antibody 82E1, revealing time-dependent aggregation of Aβ1–40. Increased levels of high-molecular-weight Aβ oligomers are evident after 2 h of incubation in Aβ samples upon addition of recombinant mutant ASC (recASC; D134R and Y187E) specks. The levels of Aβ oligomers increased with incubation time. Formation of intermediate Aβ oligomers (from 28 to 62 kDa bands) is also apparent and increased with incubation time. h, Immunoblot stained for ASC revealing auto-aggregation of recombinant ASC–CARD mutant ASC (D134R and Y187E) specks. i, Quantification of Aβ1–40 at the indicated time points. n = 3 biologically independent samples. Two-tailed Student’s t-test, 2 h: **P = 0.0012; 4 h: **P = 0.0052; 6 h: **P = 0.0032; 12 h: **P = 0.0033; 48 h: ***P = 0.0003; 24 h and 72h: ***P < 0.0001. j, Quantification of Aβ1–42 at the indicated time points. n = 3 biologically independent samples. Two-tailed Student’s t-test, 2 h: *P = 0.0212; 4 h: **P = 0.0012; 6 h: *P = 0.0240; 12 h: **P = 0.0018; 24 h: **P = 0.0069; 48 h: **P = 0.0031; 72 h: ***P = 0.0002. k, Ribbon diagrams displaying the positions of the respective mutations in the PYD and CARD domains of ASC. Experiments were performed independently three times (a–h). Data are mean ± s.e.m. (i, j).
Extended Data Figure 7 ThT fluorescence analysis of samples from co-sedimentation assay of Aβ peptides and ASC specks.
a, b, ThT fluorescence spectra of the supernatant (a) and pellet (b) fractions of Aβ1–40 (Aβ1–40 alone or in combination with ASC (Aβ1–40 + ASC)) at 0 and 6 h post incubation monitored at λ emission between 460 and 605 nm with excitation at 446 nm. Excitation and emission slit was set at 10 nm. a.u., arbitrary units. c, Quantification of the λmax values (485 nm) and statistical analysis. sup, supernatant; pel, pellet. n = 3 biologically independent samples. Two-tailed Student’s t-test, **P = 0.0011, P = ***0.0003. d, e, ThT fluorescence spectra of supernatant (d) and pellet (e) fractions of Aβ1–42 (Aβ1–42 alone or in combination with ASC (Aβ1–42 + ASC)) at 0 and 6 h post incubation obtained as in a, b. f, Quantification of the λmax values (485 nm) and statistical analysis. n = 3 biologically independent samples. Two-tailed Student’s t-test, **P = 0.0023, ***P < 0.0001. g, h, Aβ1–40 (g) or Aβ1–42 (h) in the presence or absence of ASC specks with anti-Aβ antibody (82E1) (1, Aβ alone; 2, Aβ and ASC; 3, ASC). Experiments were performed independently three times (g, h). Data are mean ±s.e.m. (a–f).
Extended Data Figure 8 ASC immunopositivity is found in the centre of Aβ deposits of APP/PS1 mice and patients with AD.
a, Identical samples from mouse fibre or core preparations as analysed in Fig. 2 were probed only with the secondary antibody used for ASC detection. b, Identical samples from human fibre or core preparations as analysed in Fig. 2 were probed only with the secondary antibody used for ASC detection. c, Recombinant ASC and synthetic Aβ1–42 were sequentially diluted and immunoprobed using ASC (AL177) or Aβ (6E10) antibodies. For further decription of the methods see ref. 31. d, Immunostaining of Aβ (6E10) and ASC (AL177) in sections derived from APP/PS1 mice with and without primary and/or secondary antibodies. e, Control section from Asc−/− animals stained for Aβ or ASC. f, g, Immunoprecipitation of ASC followed by immunoblot detection of ASC (f) or immunoblot detection of Aβ (g) in brain samples from controls without dementia (Con) and patients with AD. A further control shows the same detection of in vitro co-incubation of Aβ1–42, ASC and Aβ1–42 + ASC. h, Immunostaining of Aβ (green) and ASC (red) in sections derived from brains from patients with AD and age-matched controls without dementia (Con) and omission of both primary antibodies as a negative control. i, Immunoprecipitation experiment showing immunoprecipitation of ASC followed by western blot detection of ASC in brain samples from patients suffering from vascular dementia (VD), fronto-temporal dementia (FTD), corticobasal degeneration (CBD) and AD. j, Immunoprecipitation of ASC followed by western blot detection of Aβ in the same brain samples as i. ASC-bound Aβ was only detected in patients with AD. Experiments were performed independently three (a–c, f, g, i, j) or five (d, e, h) times. Scale bars, 15 μm (d, h) and 20 μm (e).
Extended Data Figure 9 Aβ levels and spatial navigation memory in APP/PS1;Asc−/− mice at 8 and 12 months of age.
a, ELISA quantification from SDS and FA fractions for Aβ1–38, Aβ1–40 and Aβ1–42 from 8-month-old APP/PS1 and APP/PS1;Asc−/− mice. n = 5 biologically independent animals. Two-tailed Student’s t-test, SDS: Aβ1–38 **P = 0.0016, Aβ1–40 **P = 0.0025, Aβ1–42 ***P = 0.0008; FA: Aβ1–38 **P = 0.0021, Aβ1–40 **P = 0.0040, Aβ1–42 *P = 0.0106. b, Spatial memory was assessed in the Morris water maze. Distance travelled to platform by wild-type, Asc−/−, APP/PS1 and APP/PS1;Asc−/− mice. Quantification was performed by integrating the distance travelled (AUC). n = 12 wild-type, n = 19 Asc−/−, n = 14 APP/PS1 and n = 21 APP/PS1;Asc−/− mice. One-way ANOVA, Tukey test, APP/PS1 versus APP/PS1;Asc−/− mice ***P = 0.0005; other: ***P < 0.0001. c, ELISA quantification from SDS and FA fractions for Aβ1–38, Aβ1–40 and Aβ1–42 from 12-month-old APP/PS1 and APP/PS1;Asc−/− mice. n = 5 mice. Two-tailed Student’s t-test, SDS: Aβ1–38 ***P < 0.0001, Aβ1–40 **P = 0.0015, Aβ1–42 ***P = 0.0002; FA: Aβ1–38 ***P = 0.0009, Aβ1–40 **P = 0.0084, Aβ1–42 **P = 0.0010. d, Hippocampal sections from wild-type, Asc−/−, APP/PS1 and APP/PS1;Asc−/− mice at 12 months of age and quantification of total area and the number of Aβ deposits. Scale bar, 500 μm. n = 6 mice. Two-tailed Student’s t-test, ***P < 0.0001. e, f, Spatial memory was assessed by Morris water maze testing. e, Time needed to reach the platform (latency) in wild-type (WT), Asc−/−, APP/PS1 and APP/PS1;Asc−/− mice and integrated time travelled. n = 11 wild-type, n = 11 Asc−/−, n = 17 APP/PS1 and n = 15 APP/PS1;Asc−/− mice. One-way ANOVA, Tukey test, wild-type versus APP/PS1 mice ***P < 0.0001; Asc−/− versus APP/PS1 mice ***P = 0.0003; APP/PS1 versus APP/PS1;Asc−/− mice **P = 0.0022. f, Distance travelled to platform (distance to platform) in wild-type, Asc−/−, APP/PS1 and APP/PS1;Asc−/− mice and integrated distance travelled. n = 11 wild-type, n = 11 Asc−/−, n = 17 APP/PS1 and n = 15 APP/PS1;Asc−/− mice. One-way ANOVA, Tukey test, APP/PS1 versus APP/PS1;Asc−/− mice ***P = 0.0004; other: ***P < 0.0001. g, h, At day 9, 24 h after the last training session, a spatial probe trial was conducted, where the platform was removed and the time mice spent in each quadrant was recorded. g, Q1, platform location on day 1–8. The values for the time spent in all other quadrants were averaged (o.a.). n = 12 wild-type, n = 19 Asc−/−, n = 14 APP/PS1 and n = 21 APP/PS1;Asc−/− mice. One-way ANOVA, Tukey test, Asc−/− mice Q1 versus all other quadrants *P = 0.0329. h, Representative runs of a single mouse from g are depicted. Experiments were performed independently twice (d). Data are mean ± s.e.m. (a–g).
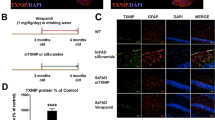
Extended Data Figure 10 Age-dependent modulation of cortical Aβ levels by ASC in APP/PS1 mice and analysis of CASP-1 cleavage, NEP and IDE.
a, Immunohistochemistry of cortical sections from wild-type (WT), Asc−/−, APP/PS1 and APP/PS1;Asc−/− mice at 3, 8 and 12 months of age using antibody 6E10. Scale bar, 500 μm. b, Quantification of Aβ deposition for total Aβ covered area (total area) and number of Aβ deposits in the respective cortical sections of APP/PS1 and APP/PS1;Asc−/− mice at 8 (n = 3 mice, two-tailed Student’s t-test, number of Aβ deposits ***P = 0.0009; total area ***P < 0.0001) and 12 months of age (n = 6 mice, two-tailed Student’s t-test, number of Aβ deposits ***P = 0.0003; total area ***P = 0.0002). Data are mean ± s.e.m. c–f, Analysis of experiment 1–4 for CASP-1, NEP and IDE levels in animals undergoing the respective experimental protocol (see also Extended Data Fig. 11b–d). Detection of β-actin levels served as a loading control. Positive controls represent wild-type mouse brain lysate spiked with CASP-1, NEP and IDE. R = right hemisphere lysate; L = left hemisphere lysate. The genetic background of the injected animals are indicated: EXPI: APP/PS1 mice; EXPII: APP/PS1 and APP/PS1;Asc−/− mice; EXPIII: APP/PS1 mice; EXP IV: APP/PS1 mice and respective controls as well as the injected material or brain lysate. Experiments were performed independently two (a) or three (c–f) times.
Extended Data Figure 11 Microglial Aβ phagocytosis in 8-month-old APP/PS1 and APP/PS1;Asc−/− mice and experimental schematics of Aβ in vivo seeding experiments
a, Representative scatter plots of mice analysed for microglial amyloid content after intraperitoneal injection of methoxy-X04 (Mx04), isolation of microglia at 8 months of age and quantification of amyloid content revealing no differences between groups. n = 11 APP/PS1, n = 10 APP/PS1;Asc−/− mice. Data are mean ± s.e.m. Two-tailed Student’s t-test. b, Design of in vivo experiments 1–4 (EXP-I–IV) showing the genetic background of host mice and injected materials. c, Time schedule of experiment 1 (EXP-I). d, Time schedule of experiments 2–4 (EXP-II–IV). e, Brain lysates were generated as described in refs 23, 32. Schematic of the preparation of the injection material from mouse forebrain. Aliquots of brain homogenates from APP/PS1 and APP/PS1;Asc−/− mice were analysed for Aβ content by immunoblot using antibody 82E1 and anti-actin antibody to normalize for protein loading. f, Site of bi-hippocampal injection and sections analysed with an equal distance of 120 μm to each other. Experiments were performed independently twice (a, e).
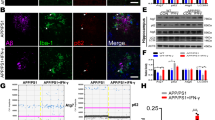
Extended Data Figure 12 ASC specks cause rostro-caudal spreading of Aβ pathology in APP/PS1 mice without affecting microglial phagocytosis.
a, b, Representative micrographs of injected hippocampi (a) and Aβ immunostained area (total area) and number of Aβ-immunopositive deposits (b). Scale bar, 500 μm. n = 8 biologically independent samples (APP/PS1 mice injected with control solution (con.sol.) or ASC specks, n = 4 biologically independent samples (non-injected (non-inj.) APP/PS1 mice). One-way ANOVA, Tukey test, total area of ASC speck versus control solution ***P < 0.0001, ASC specks versus not injected ***P = 0.0006; number of Aβ deposits ASC speck versus control solution ***P = 0.0003, ASC specks versus not injected ***P < 0.0001. c, Immunoblots for APP, α and β C-terminal fragments (α-CTF, β-CTF) and Aβ from injected hemispheres. Brain lysates from non-injected (non-inj.) six-month old APP/PS1 animals or wild-type mice were used as controls. d, Quantification of Aβ monomers. n = 5 biologically independent samples. One-way ANOVA, Tukey test, ASC speck versus control solution ***P < 0.0001, ASC specks versus not injected ***P = 0.0005. e–h, Determination of the rostro-caudal ASC-speck-induced spreading of Aβ pathology. Number of Aβ+ deposits displayed for each section for EXP-I (e), EXP-II (f), EXP-III (g) and EXP-IV (h). EXP-I: n = 7 biologically independent samples; EXP-II n = 3 biologically independent samples; EXP-III: n = 3 biologically independent samples; EXP-IV: n = 5 biologically independent samples. One-tailed Student’s t-test, (levels from −4 to +4) EXP-I: −2 **P = 0.0028, 1 *P = 0.0194, 2 **P = 0.061, 4 ***P = 0.0007; EXP-II: −3 *P = 0.0175, −2 *P = 0.0216, 1 **P = 0.0090, 2 *P = 0.0312; EXP-III: −4 *P = 0.0181, −2 *P = 0.0194, 2 *P = 0.0195, 3 **P = 0.0072; EXP-IV: −4 ***P = 0.0008, −3 **P = 0.0037, −2 *P = 0.0414, −1 *P = 0.0144, 1 ***P < 0.0001, 2 **P = 0.0088, 3 **P = 0.0012. i, Representative scatter plots of samples from mice analysed for microglial amyloid content after intraperitoneal injection of methoxy-X04 (Mx04) and isolation of microglia at one month after injection. Wild-type (WT) mice isolation of microglial cell population with immunostaining for Cd11b and CD45 (top) and after intraperitoneal administration of methoxy-X04 (bottom). APP/PS1 mice receiving intrahippocampal injections with control solvent (top) and ASC specks (bottom) immunostained for CD11b, CD45 and methoxy-X04. Quantification of phagocytosis reveals no differences between groups. n = 3 mice. Two-tailed student’s t-test. Experiments were performed independently two times (a), five times (c) and once (i). Data are mean ± s.e.m. (b, d–i).
Extended Data Figure 13 Lack of IDE and phagocytosis modulation in vivo seeding experiments.
Representative scatter plots of animals analysed for microglial amyloid content after intraperitoneal injection of methoxy-X04 (Mx04) and isolation of microglia one month after injection. a, Analysis of microglial cell population (top) from wild-type mice (WT) before and after intraperitoneal administration of Mx04 (bottom). APP/PS1 or APP/PS1;Asc−/− mice (host animals: red) injected with either APP/PS1 or wild-type mouse brain homogenate (injection material: green). b, Quantification of amyloid content revealed no differences between groups. n = 3 biologically independent samples. One-way ANOVA, Tukey test). c, Enzymatic IDE activity was analysed from mouse brain homogenates derived from experiments I–IV using the FRET substrate (5-FAM/QXL520) and given as relative fluorescence units (RFU) per mg brain tissue. EXP-I: n = 7 biologically independent samples (APP/PS1 mice injected with control solution (con.sol.) or ASC specks), n = 4 biologically independent samples non-injected (non-inj.) APP/PS1 mice; EXP-II: n = 4 biologically independent samples; EXP-III: n = 5 biologically independent samples; EXP-IV: n = 6 biologically independent samples. One-way ANOVA, Tukey test. Experiments were performed once (a). Data are mean ± s.e.m. (b, c).
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-3, the uncropped gels. (PDF 3738 kb)
Source data
Rights and permissions
About this article
Cite this article
Venegas, C., Kumar, S., Franklin, B. et al. Microglia-derived ASC specks cross-seed amyloid-β in Alzheimer’s disease. Nature 552, 355–361 (2017). https://doi.org/10.1038/nature25158
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature25158
This article is cited by
-
Nanotechnology for microglial targeting and inhibition of neuroinflammation underlying Alzheimer’s pathology
Translational Neurodegeneration (2024)
-
Exercise mimetics: a novel strategy to combat neuroinflammation and Alzheimer’s disease
Journal of Neuroinflammation (2024)
-
Drugging the NLRP3 inflammasome: from signalling mechanisms to therapeutic targets
Nature Reviews Drug Discovery (2024)
-
Inflammasomes in neurological disorders — mechanisms and therapeutic potential
Nature Reviews Neurology (2024)
-
Neuropathogenesis-on-chips for neurodegenerative diseases
Nature Communications (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.