Abstract
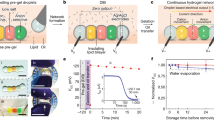
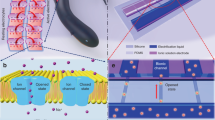
Progress towards the integration of technology into living organisms requires electrical power sources that are biocompatible, mechanically flexible, and able to harness the chemical energy available inside biological systems. Conventional batteries were not designed with these criteria in mind. The electric organ of the knifefish Electrophorus electricus (commonly known as the electric eel) is, however, an example of an electrical power source that operates within biological constraints while featuring power characteristics that include peak potential differences of 600 volts and currents of 1 ampere1,2. Here we introduce an electric-eel-inspired power concept that uses gradients of ions between miniature polyacrylamide hydrogel compartments bounded by a repeating sequence of cation- and anion-selective hydrogel membranes. The system uses a scalable stacking or folding geometry that generates 110 volts at open circuit or 27 milliwatts per square metre per gel cell upon simultaneous, self-registered mechanical contact activation of thousands of gel compartments in series while circumventing power dissipation before contact. Unlike typical batteries, these systems are soft, flexible, transparent, and potentially biocompatible. These characteristics suggest that artificial electric organs could be used to power next-generation implant materials such as pacemakers, implantable sensors, or prosthetic devices in hybrids of living and non-living systems3,4,5,6.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Bennett, M. V. L. in Fish Physiology Vol. 5 (eds Hoar, W. S. & Randall, D. J. ) 347–491 (Academic Press, 1971)
Gotter, A. L., Kaetzel, M. A. & Dedman, J. R. Electrophorus electricus as a model system for the study of membrane excitability. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 119, 225–241 (1998)
Xu, J., Sigworth, F. J. & LaVan, D. A. Synthetic protocells to mimic and test cell function. Adv. Mater. 22, 120–127 (2010)
Sun, H., Fu, X., Xie, S., Jiang, Y. & Peng, H. Electrochemical capacitors with high output voltages that mimic electric eels. Adv. Mater. 28, 2070–2076 (2016)
Kim, D.-H. et al. Epidermal electronics. Science 333, 838–843 (2011)
Whitesides, G. M. Assumptions: taking chemistry in new directions. Angew. Chem. Int. Ed. 43, 3632–3641 (2004)
Gallant, J. R. et al. Nonhuman genetics. Genomic basis for the convergent evolution of electric organs. Science 344, 1522–1525 (2014)
Catania, K. C. Leaping eels electrify threats, supporting Humboldt’s account of a battle with horses. Proc. Natl Acad. Sci. USA 113, 6979–6984 (2016)
Xu, J. & Lavan, D. A. Designing artificial cells to harness the biological ion concentration gradient. Nat. Nanotechnol. 3, 666–670 (2008)
Brown, M. V. The electric discharge of the electric eel. Electr. Eng. 69, 145–147 (1950)
Nachmansohn, D., Cox, R. T., Coates, C. W. & Machado, A. L. Action potential and enzyme activity in the electric organ of Electrophorus electricus (Linnaeus): I. Choline esterase and respiration. J. Neurophysiol. 5, 499–515 (1942)
Keynes, R. D. & Martins-Ferreira, H. Membrane potentials in the electroplates of the electric eel. J. Physiol. 119, 315–351 (1953)
Cox, R. T., Coates, C. W. & Brown, M. V. Electrical characteristics of electric tissue. Ann. NY Acad. Sci. 47, 487–500 (1946)
Pattle, R. E. Production of electric power by mixing fresh and salt water in the hydroelectric pile. Nature 174, 660–660 (1954)
Zeng, S., Li, B., Su, X., Qin, J. & Lin, B. Microvalve-actuated precise control of individual droplets in microfluidic devices. Lab Chip 9, 1340–1343 (2009)
Bardin, D. et al. High-speed, clinical-scale microfluidic generation of stable phase-change droplets for gas embolotherapy. Lab Chip 11, 3990–3998 (2011)
Young, C., Rozario, K., Serra, C., Poole-Warren, L. & Martens, P. Poly(vinyl alcohol)-heparin biosynthetic microspheres produced by microfluidics and ultraviolet photopolymerisation. Biomicrofluidics 7, 44109 (2013)
Gumuscu, B. et al. Desalination by electrodialysis using a stack of patterned ion-selective hydrogels on a microfluidic device. Adv. Funct. Mater. 26, 8685–8693 (2016)
Miura, K. Method of Packaging and Deployment of Large Membranes in Space. Report No. 618 (Institute of Space and Astronautical Science, 1985)
Shenkel, S. & Sigworth, F. J. Patch recordings from the electrocytes of Electrophorus electricus. Na currents and PNa/PK variability. J. Gen. Physiol. 97, 1013–1041 (1991)
Ide, T., Takeuchi, Y. & Yanagida, T. Development of an experimental apparatus for simultaneous observation of optical and electrical signals from single ion channels. Single Mol. 3, 33–42 (2002)
Feng, J. et al. Single-layer MoS2 nanopores as nanopower generators. Nature 536, 197–200 (2016)
Lingley, A. R. et al. A single-pixel wireless contact lens display. J. Micromech. Microeng. 21, 125014 (2011)
Mansouri, K., Medeiros, F. A., Tafreshi, A. & Weinreb, R. N. Continuous 24-hour monitoring of intraocular pressure patterns with a contact lens sensor: safety, tolerability, and reproducibility in patients with glaucoma. Arch. Ophthalmol. 130, 1534–1539 (2012)
Lai, Y.-C. et al. Electric eel-skin-inspired mechanically durable and super-stretchable nanogenerator for deformable power source and fully autonomous conformable electronic-skin applications. Adv. Mater. 28, 10024–10032 (2016)
Ilievski, F., Mazzeo, A. D., Shepherd, R. F., Chen, X. & Whitesides, G. M. Soft robotics for chemists. Angew. Chem. Int. Ed. 50, 1890–1895 (2011)
Chandrakasan, A. P., Verma, N. & Daly, D. C. Ultralow-power electronics for biomedical applications. Annu. Rev. Biomed. Eng. 10, 247–274 (2008)
Enger, C. C. & Simeone, F. A. Biologically energized cardiac pacemaker: in vivo experience with dogs. Nature 218, 180–181 (1968)
Chin, S. Y. et al. Additive manufacturing of hydrogel-based materials for next-generation implantable medical devices. Sci. Robot. 2, eaah6451 (2017)
Cox, R. T., Rosenblith, W. A., Cutler, J. A., Mathews, R. S. & Coates, C. W. A comparison of some electrical and anatomical characteristics of the electric eel, Electrophorus electricus. Zoologica 25, 553–562 (1940)
Schoffeniels, E. Ion movements studied with single isolated electroplax. Ann. NY Acad. Sci. 81, 285–306 (1959)
Altamirano, M. & Coates, C. W. Effect of potassium on electroplax of Electrophorus electricus. J. Cell. Comp. Physiol. 49, 69–101 (1957)
Nakamura, Y., Nakajima, S. & Grundfest, H. Analysis of spike electrogenesis and depolarizing K inactivation in electroplaques of Electrophorus electricus, L. J. Gen. Physiol. 49, 321–349 (1965)
Acknowledgements
We are grateful to B. Rothen-Rutishauser and A. Petri-Fink at the Adolphe Merkle Institute for the use of their 3DDiscovery printer. F. Bircher’s iPrint institute at the Haute École d’Ingénierie et d’Architecture Fribourg, particularly F. Bourguet and M. Soutrenon, donated time towards adapting a printer for our use and helped us to understand the intricacies of microvalve printing systems. Laser cutting was performed at Fablab Fribourg. U. Steiner’s group, in particular P. Sutton and M. Fischer, provided instrumentation and advice related to impedance measurements. Research reported in this publication was supported by the Air Force Office of Scientific Research (grant FA9550-12-1-0435 to M.M., J.Y., D.S. and M.S.) and the National Institute of General Medical Sciences of the National Institutes of Health under award T32GM008353, which funds the Cellular Biotechnology Training Program (T.B.H.S.). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
T.B.H.S., A.G., J.Y. and M.M. conceived the project and designed the experiments. T.B.H.S. and A.G. performed all data collection. A.L. and M.S. provided the idea of Miura-ori folding. G.V. helped to define the parameters of the fluidic implementation. T.B.H.S. and D.S. conducted analysis of literature electrical datasets of Electrophorus. T.B.H.S., A.G. and M.M. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer Information Nature thanks C. Bettinger, P. Calvert and A. Stokes for their contribution to the peer review of this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Figure 1 Charged monomers used in charge-selective ‘membrane’ gels.
a, 3-Sulfopropyl acrylate, a component of the cation-selective gel. b, (3-Acrylamidopropyl)trimethylammonium, a component of the anion-selective gel.
Extended Data Figure 2 Self-discharge of artificial electric organ over time after contact between all gels, with and without exposure to ambient air.
Curves were fit with a single exponential decay function (dotted curves); the half-time for each was 40 min. The artificial electric organ was assembled as described in Supplementary Information 3. Supplementary Video 1 shows a fluidic implementation of the artificial electric organ which positions gels into contact sequentially rather than simultaneously. Large-scale implementations of similar sequential positioning schemes may be prone to power loss from gradient depletion unless positioning could be accomplished rapidly.
Extended Data Figure 3 The artificial electric organ can be recharged.
Experimental details in Supplementary Information 3. a, Current versus time recordings of ten discharges of a single tetrameric gel cell at short circuit following recharging. Initial discharge shown in black; subsequent discharges in the following order: red, blue, magenta, green, navy, purple, plum, wine, olive. b, Bar graph of normalized integrals of discharge curves.
Extended Data Figure 4 Geometrical arrangement of the printed 45° Miura-ori gel cells.
Dotted lines of a single colour indicate gels forming a series. Different colours indicate parallel sequences. This fold geometry is scalable both in series for higher voltage output and in parallel for higher current. In contrast to the 80° Miura-ori fold, all gels are located on the same side of the substrate, facilitating fabrication by printing or other methods. (Supplementary Information 5).
Extended Data Figure 5 Internal resistance and power density of tetrameric gel cells as a function of thickness of low-salinity gel.
Internal resistance, black squares; power density, red circles. The thicknesses of all other gels were held constant at 1 mm.
Extended Data Figure 6 Equivalent circuit of an artificial electric organ connected to a load resistance.
The elements within the dotted line represent the contribution of a single tetrameric gel cell; these can be added in series or in parallel. The impedance of the voltmeter that was used exceeded 10 GΩ; current through this pathway was assumed to be negligible.
Supplementary information
Supplementary Information
This file contains seven discussion sections and two supplementary tables. The contents are theoretical background, characterization, fabrication methods, and calculations related to the artificial electric organ presented in this work. (PDF 593 kb)
Fluidic artificial organ implementation
This video shows fluidic artificial organ implementation. (MP4 11160 kb)
Printer depositing gels for serpentine implementation.
This video shows printer depositing gels for serpentine implementation. (MP4 19167 kb)
Miura-ori folding of a gel-bearing substrate
This video shows Miura-ori folding of a gel-bearing substrate. (MP4 2015 kb)
Rights and permissions
About this article
Cite this article
Schroeder, T., Guha, A., Lamoureux, A. et al. An electric-eel-inspired soft power source from stacked hydrogels. Nature 552, 214–218 (2017). https://doi.org/10.1038/nature24670
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature24670
This article is cited by
-
Reconfiguring hydrogel assemblies using a photocontrolled metallopolymer adhesive for multiple customized functions
Nature Chemistry (2024)
-
A high-current hydrogel generator with engineered mechanoionic asymmetry
Nature Communications (2024)
-
Nature-inspired interfacial engineering for energy harvesting
Nature Reviews Electrical Engineering (2024)
-
Polyelectrolyte elastomer-based ionotronic sensors with multi-mode sensing capabilities via multi-material 3D printing
Nature Communications (2023)
-
A microscale soft ionic power source modulates neuronal network activity
Nature (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.