Abstract
Heat is fundamental to power generation and many industrial processes, and is most useful at high temperatures because it can be converted more efficiently to other types of energy. However, efficient transportation, storage and conversion of heat at extreme temperatures (more than about 1,300 kelvin) is impractical for many applications. Liquid metals can be very effective media for transferring heat at high temperatures, but liquid-metal pumping has been limited by the corrosion of metal infrastructures. Here we demonstrate a ceramic, mechanical pump that can be used to continuously circulate liquid tin at temperatures of around 1,473–1,673 kelvin. Our approach to liquid-metal pumping is enabled by the use of ceramics for the mechanical and sealing components, but owing to the brittle nature of ceramics their use requires careful engineering. Our set-up enables effective heat transfer using a liquid at previously unattainable temperatures, and could be used for thermal storage and transport, electric power production, and chemical or materials processing.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Lorenzin, N. & Abánades, A. A review on the application of liquid metals as heat transfer fluid in concentrated solar power technologies. Int. J. Hydrogen Energy 41, 6990–6995 (2016)
Wilk, G. Liquid Metal Based High Temperature Concentrated Solar Power: Cost Considerations. MSc thesis, Georgia Institute of Technology (2016)
Nunes, V. M. B., Queirós, C. S., Lourenço, M. J. V., Santos, F. J. V. & Nieto de Castro, C. A. Molten salts as engineering fluids. Appl. Energy 183, 603–611 (2016)
Fritsch, A. et al. Conceptual study of central receiver systems with liquid metals as efficient heat transfer fluids. Energy Procedia 69, 644–653 (2015)
Elkin, B., Finkelstein, L., Dyer, T. & Raade, J. Molten oxide glass materials for thermal energy storage. Energy Procedia 49, 772–779 (2014)
Cárdenas, B., León, N., Pye, J. & García, H. D. Design and modelling of a high temperature solar thermal energy storage unit based on molten soda lime silica glass. Solar Energy 126, 32–43 (2016)
Elliott, A. J. & Pollock, T. M. Thermal analysis of the Bridgman and liquid-metal-cooled directional solidification investment casting processes. Metall. Mater. Trans. A 38, 871–882 (2007)
Plevan, M. et al. Thermal cracking of methane in a liquid metal bubble column reactor: experiments and kinetic analysis. Int. J. Hydrogen Energy 40, 8020–8033 (2015)
Fazio, C. & Balbaud, F. in Structural Materials for Generation IV Nuclear Reactors (ed. Yvon, P. ) 23–74 (Elsevier, 2017)
Legkikh, A. Y., Askhadullin, R. S. & Sadovnichiy, R. P. Ensuring the corrosion resistance of steels in heavy liquid metal coolants. Nuclear Energy Technol. 2, 136–141 (2016)
Emmerich, T. & Schroer, C. Corrosion in austenitic steels and nickel-based alloys caused by liquid tin at high temperature. Corros. Sci. 120, 171–183 (2017)
Danzer, R. On the relationship between ceramic strength and the requirements for mechanical design. J. Eur. Ceram. Soc. 34, 3435–3460 (2014)
Henry, A. & Prasher, R. The prospect of high temperature solid state energy conversion to reduce the cost of concentrated solar power. Energy Environ. Sci. 7, 1819–1828 (2014)
Opeka, M. M., Talmy, I. G., Wuchina, E. J., Zaykoski, J. A. & Causey, S. J. Mechanical, thermal, and oxidation properties of refractory hafnium and zirconium compounds. J. Eur. Ceram. Soc. 19, 2405–2414 (1999)
Opeka, M. M., Talmy, I. G. & Zaykoski, J. A. Oxidation-based materials selection for 2000°C + hypersonic aerosurfaces: theoretical considerations and historical experience. J. Mater. Sci. 39, 5887–5904 (2004)
Fahrenholtz, W. G. & Hilmas, G. E. Ultra-high temperature ceramics: materials for extreme environments. Scr. Mater. 129, 94–99 (2017)
Frazer, D., Stergar, E., Cionea, C. & Hosemann, P. Liquid metal as a heat transport fluid for thermal solar power applications. Energy Procedia 49, 627–636 (2014)
Pacio, J. & Wetzel, T. Assessment of liquid metal technology status and research paths for their use as efficient heat transfer fluids in solar central receiver systems. Sol. Energy 93, 11–22 (2013)
Vignarooban, K., Xu, X., Arvay, A., Hsu, K. & Kannan, A. M. Heat transfer fluids for concentrating solar power systems – a review. Appl. Energy 146, 383–396 (2015)
Weeks, J. R. Lead, bismuth, tin and their alloys as nuclear coolants. Nucl. Eng. Des. 15, 363–372 (1971)
Bruckner, A. P. & Mattick, A. T. High effectiveness liquid droplet/gas heat exchanger for space power applications. Acta Astronaut. 11, 519–526 (1984)
Welty, J. R. in Direct-Contact Heat Transfer (eds Kreith, F. & Boehm, R. F. ) 197–201 (Springer, 1988)
Gur, I., Sawyer, K. & Prasher, R. Searching for a better thermal battery. Science 335, 1454–1455 (2012)
Kapranos, P., Carney, C., Pola, A. & Jolly, M. in Comprehensive Materials Processing Vol. 5 (eds Hashmi, S. et al. 39–67 (Elsevier, 2014)
Thekdi, A. C. & Nimbalkar, S. U.Industrial Waste Heat Recovery: Potential Applications, Available Technologies and Crosscutting R&D Opportunities. Report No. ORNL/TM-2014/622 (Oak Ridge National Laboratory, 2015)
Seyf, H. R. & Henry, A. Thermophotovoltaics: a potential pathway to high efficiency concentrated solar power. Energy Environ. Sci. 9, 2654–2665 (2016)
Yang, W. et al. Thermodynamics analysis of carbothermal-chlorination reduction in aluminum production. Appl. Therm. Eng. 111, 876–883 (2017)
Dong, X., Huang, X., Liu, L., He, L. & Li, P. A liquid aluminum alloy electromagnetic transport process for high pressure die casting. J. Mater. Process. Technol. 234, 217–227 (2016)
Kim, H. R. & Lee, Y. B. A design and characteristic experiment of the small annular linear induction electromagnetic pump. Ann. Nucl. Energy 38, 1046–1052 (2011)
Bodbodak, S. & Moshfeghifar, M. in Eco-Friendly Technology for Postharvest Product Quality (ed. Siddiqui, M. W. ) 39–76 (Academic Press, 2016)
Thompson, A. K. Fruit and Vegetable Storage 21–36 (Elsevier, 2016)
Zhao, H. & Woods, R. in Advances in Brazing (ed. Sekulic, D. P. ) 280–322 (Woodhead Publishing, 2013)
Oden, L. L. & Gokcen, N. A. Sn-C and Al-Sn-C phase diagrams and thermodynamic properties of C in the alloys: 1550 °C to 2300 °C. Metall. Trans. B 24, 53–58 (1993)
Pietzka, M. A. & Schuster, J. C. Phase equilibria of the quaternary system Ti/Al/Sn/N at 900 °C. J. Alloys Compd. 247, 198–201 (1997)
Beebe, R. S. Predicitive Maintenance of Pumps Using Condition Monitoring 155–161 (Elsevier, 2004)
Forsthoffer, W. E. Forsthoffer’s Best Practice Handbook for Rotating Machinery 25–91 (Butterworth-Heinemann, 2011)
Hill, R. F. Boron nitride seal and method of use. US patent 5,678,832 (1997)
Nesbitt, B. (ed.) Handbook of Pumps and Pumping 1–54 (Elsevier, 2006)
Milak, P. C., Minatto, F. D., De Noni, A. Jr & Montedo, O. R. K. Wear performance of alumina-based ceramics — a review of the influence of microstructure on erosive wear. Ceramica 61, 88–103 (2015)
Hutchings, I . & Shipway, P. Friction and Wear of Engineering Materials 1–5 (Butterworth-Heinemann, 2017)
DeAngelis, A. Analysis and Design of a High Temperature Liquid Metal Solar Thermal Receiver MSc thesis, Georgia Institute of Technology (2016)
Dowling, A. W., Zheng, T. & Zavala, V. M. Economic assessment of concentrated solar power technologies: a review. Renew. Sustain. Energy Rev. 72, 1019–1032 (2017)
SunShot Vision Study. Report No. DOE/GO-102012–3037 (US Department of Energy, 2012)
On the Path to SunShot. Report No. DOE/EE 1412 (US Department of Energy, 2016)
Yuan, C., Jarrett, C., Chueh, W., Kawajiri, Y. & Henry, A. A new solar fuels reactor concept based on a liquid metal heat transfer fluid: reactor design and efficiency estimation. Sol. Energy 122, 547–561 (2015)
Sioshansi, R., Denholm, P., Jenkin, T. & Weiss, J. Estimating the value of electricity storage in PJM: arbitrage and some welfare effects. Energy Econ. 31, 269–277 (2009)
Cascella, F. & Teyssedou, A. Modeling a direct contact heat exchanger used in a supercritical water loop. Appl. Therm. Eng. 79, 132–139 (2015)
Wilcock, R. C., Young, J. B. & Horlock, J. H. The effect of turbine blade cooling on the cycle efficiency of gas turbine power cycles. J. Eng. Gas Turbines Power 127, 109–120 (2005)
Brundidge, C. L., Miller, J. D. & Pollock, T. M. Development of dendritic structure in the liquid-metal-cooled, directional-solidification process. Metall. Mater. Trans. A 42, 2723–2732 (2011)
Venkatraman, M. & Neumann, J. P. The Cr−Sn (chromium–tin) system. J. Phase Equilibria 9, 159–162 (1988)
Predel, B. Sn–W (Tin–Tungsten) http://doi.org/10.1007/10551312_2771 (Springer, 1998)
Massalski, T. B., Okamoto, H., Subramanian, P. R. & Kacprzak, L. (eds) Binary Alloy Phase Diagrams 2nd edn, 723–806 (ASM International, 1990)
House, J. E. & House, K. A. Descriptive Inorganic Chemistry 3rd edn, 177–196 (Academic Press, 2016)
Lima, R. S., Dionísio, P. H., Moro, J. T., Schreiner, W. H. & Achete, C. Thermal evolution and stability of a tin nitride obtained by reactive sputtering. Hyperfine Interact. 83, 315–319 (1994)
Casoli, P., Vacca, A. & Berta, G. L. Optimization of relevant design parameters of external gear pumps. Proc. JFPS Int. Symp. Fluid Power 2008, 277–282 (2008)
Amani, A., Spitas, C. & Spitas, V. Generalised non-dimensional multi-parametric involute spur gear design model considering manufacturability and geometrical compatibility. Mechanism Mach. Theory 109, 250–277 (2017)
Amy, C. Liquid Metal Pumps for Enabling Heat Transfer at Extreme Temperatures MSc thesis, Georgia Institute of Technology (2017)
He, X., Gong, Q., Guo, Y. & Liu, J. Microstructure and properties of AlN-BN composites prepared by sparking plasma sintering method. J. Alloys Compd. 675, 168–173 (2016)
Mucchi, E., Agazzi, A., D’Elia, G. & Dalpiaz, G. On the wear and lubrication regime in variable displacement vane pumps. Wear 306, 36–46 (2013)
Peterson, M. B. & Winer, W. O. (eds) Wear Control Handbook 35–80 (American Society of Mechanical Engineers, 1980)
Kato, K. Tribology of ceramics. Wear 136, 117–133 (1990)
Acknowledgements
We acknowledge funding support from the Advanced Research Projects Agency – Energy (ARPA-E) (DE-AR0000339). We also acknowledge the support of Y. Zhang, B. Capps, A. Robinson and M. Faniel.
Author information
Authors and Affiliations
Contributions
C.A., D.B. and M.B. performed the experiments, and A.H., D.E., F.D., G.W., C.K., J.H., H.W., B.G. and A.C. provided assistance. C.A. analysed the data and performed the simulations, and M.B. provided review and assistance. A.H. and D.E. supervised the project. K.H.S., C.J., C.Y., D.E., W.C.C. and Y.K. performed modelling and materials testing. C.A. drafted the majority of the manuscript, and A.H. provided chief contributions. K.H.S., D.B. and M.B. also edited extensively. All authors wrote and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer Information Nature thanks K. Lambrinou and R. Stieglitz for their contribution to the peer review of this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Figure 1 Oxygen content over time in preparation for the experiment.
After 17 h of nitrogen purging, the oxygen level decreased sufficiently for tin gettering to take over as the dominant oxygen reduction mechanism.
Extended Data Figure 2 Chamber view including seals and the visual flow meter port.
Seals were achieved by first applying room-temperature vulcanization (RTV) silicone (blue) and then applying vacuum grease. The viewport is the glowing orange slotted hole shown in the insulation. The camera position is shown as a reference to the image in Fig. 4.
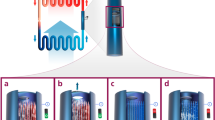
Extended Data Figure 3 Design and calibration of the visual flow meter.
a, Measured flow rate versus mass in the flow meter (mass uncertainty is ±5 g). The grey boxes identify the flow regimes, with the smaller box corresponding to transition from one- to two-hole flow, and the larger box corresponding to transition from two- to three-hole flow. The theoretical curve is determined using Bernoulli’s equation, whereby the flow rate is proportional to the square-root of mass. b–d, Illustrations of the three possible flow regimes that can be detected by the visual flow meter.
Extended Data Figure 4 Experimental set-up of the calibration of the visual flow meter.
The weight-based flow meter was positioned above the visual flow meter and the pump speed was adjusted until the desired flow (for example, through only two outlets or through three outlets) was achieved. Under these conditions, flow was monitored from both flow meters so that the quantitative information from the weight-based flow meter could be related to the qualitative information from the visual flow meter.
Extended Data Figure 5 Internal features of the gear pump, including wear surfaces.
a, View of the internal geometry of the pump system. b, View of the wear surfaces of the pump gear. Although wear occurred on most dynamic surfaces, the most extensive wear occurred on the gear shafts. Wear allows the gear tips to contact the pump body, which results in performance-reducing wear on the tips of the gear teeth.
Supplementary information
Supplementary Table 1
This file contains raw pump temperature data from 72-hour experiment. From a b type thermocouple attached to the pump inlet. This file contains data relating to Fig. 2. (XLSX 6056 kb)
Supplementary Table 2
This file contains raw data from oxygen sensor. The first column is in seconds and the second column is in ppm. This file contains data relating to Extended Data Fig. 2. (XLSX 2399 kb)
Supplementary Table 3
This file contains data from the calibration of the flow meter. This file contains data relating to Extended Data Fig. 3. (XLSX 144 kb)
Pumping liquid metal at 1,500 K
This video shows the testing of an all ceramic liquid metal pump at 1,200 °C, including clips before and after. The pump is shown disassembled, then the rotation of the gears is demonstrated. Next, the pump system is shown, followed by the liquid metal flow that was observed for 72 hours (at ~10,000 X), with the cool down included. The disassembly process is also shown, including the gears as removed, with visible wear. (MP4 24471 kb)
Rights and permissions
About this article
Cite this article
Amy, C., Budenstein, D., Bagepalli, M. et al. Pumping liquid metal at high temperatures up to 1,673 kelvin. Nature 550, 199–203 (2017). https://doi.org/10.1038/nature24054
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature24054
This article is cited by
-
Liquid-based high-temperature receiver technologies for next-generation concentrating solar power: A review of challenges and potential solutions
Frontiers in Energy (2023)
-
Thermophotovoltaic efficiency of 40%
Nature (2022)
-
Liquid metal hydraulics paradigm: Transmission medium and actuation of bimodal signals
Science China Technological Sciences (2022)
-
Improvement in mechanical properties in AlN-h-BN composites with high thermal conductivity
Journal of Advanced Ceramics (2021)
-
Uncovering the evolution of tin use in the United States and its implications
Frontiers of Environmental Science & Engineering (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.