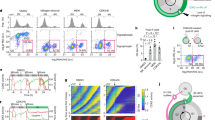
Abstract
Regulation of cell proliferation is necessary for immune responses, tissue repair, and upkeep of organ function to maintain human health1. When proliferating cells complete mitosis, a fraction of newly born daughter cells immediately enter the next cell cycle, while the remaining cells in the same population exit to a transient or persistent quiescent state2. Whether this choice between two cell-cycle pathways is due to natural variability in mitogen signalling or other underlying causes is unknown. Here we show that human cells make this fundamental cell-cycle entry or exit decision based on competing memories of variable mitogen and stress signals. Rather than erasing their signalling history at cell-cycle checkpoints before mitosis, mother cells transmit DNA damage-induced p53 protein and mitogen-induced cyclin D1 (CCND1) mRNA to newly born daughter cells. After mitosis, the transferred CCND1 mRNA and p53 protein induce variable expression of cyclin D1 and the CDK inhibitor p21 that almost exclusively determines cell-cycle commitment in daughter cells. We find that stoichiometric inhibition of cyclin D1–CDK4 activity by p21 controls the retinoblastoma (Rb) and E2F transcription program in an ultrasensitive manner. Thus, daughter cells control the proliferation–quiescence decision by converting the memories of variable mitogen and stress signals into a competition between cyclin D1 and p21 expression. We propose a cell-cycle control principle based on natural variation, memory and competition that maximizes the health of growing cell populations.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hsu, Y.-C., Li, L. & Fuchs, E. Transit-amplifying cells orchestrate stem cell activity and tissue regeneration. Cell 157, 935–949 (2014)
Spencer, S. L. et al. The proliferation-quiescence decision is controlled by a bifurcation in CDK2 activity at mitotic exit. Cell 155, 369–383 (2013)
Hitomi, M. & Stacey, D. W. Cyclin D1 production in cycling cells depends on Ras in a cell-cycle-specific manner. Curr. Biol. 9, 1075–1084 (1999)
Weinberg, R. A. The retinoblastoma protein and cell cycle control. Cell 81, 323–330 (1995)
Iaquinta, P. J. & Lees, J. A. Life and death decisions by the E2F transcription factors. Curr. Opin. Cell Biol. 19, 649–657 (2007)
Komatsu, N. et al. Development of an optimized backbone of FRET biosensors for kinases and GTPases. Mol. Biol. Cell 22, 4647–4656 (2011)
Aoki, K. et al. Stochastic ERK activation induced by noise and cell-to-cell propagation regulates cell density-dependent proliferation. Mol. Cell 52, 529–540 (2013)
Deng, C., Zhang, P., Harper, J. W., Elledge, S. J. & Leder, P. Mice lacking p21CIP1/WAF1 undergo normal development, but are defective in G1 checkpoint control. Cell 82, 675–684 (1995)
Brugarolas, J . et al. Radiation-induced cell cycle arrest compromised by p21 deficiency. Nature 377, 552–557 (1995)
Branzei, D. & Foiani, M. The DNA damage response during DNA replication. Curr. Opin. Cell Biol. 17, 568–575 (2005)
Stewart-Ornstein, J. & Lahav, G. Dynamics of CDKN1A in single cells defined by an endogenous fluorescent tagging toolkit. Cell Reports 14, 1800–1811 (2016)
Grillo, M. et al. Validation of cyclin D1/CDK4 as an anticancer drug target in MCF-7 breast cancer cells: effect of regulated overexpression of cyclin D1 and siRNA-mediated inhibition of endogenous cyclin D1 and CDK4 expression. Breast Cancer Res. Treat. 95, 185–194 (2006)
Zhou, B. B. & Elledge, S. J. The DNA damage response: putting checkpoints in perspective. Nature 408, 433–439 (2000)
Lukas, C. et al. 53BP1 nuclear bodies form around DNA lesions generated by mitotic transmission of chromosomes under replication stress. Nat. Cell Biol. 13, 243–253 (2011)
Barr, A. R. et al. DNA damage during S-phase mediates the proliferation-quiescence decision in the subsequent G1 via p21 expression. Nat. Commun. 8, 14728 (2017)
Arora, M., Moser, J., Phadke, H., Basha, A. A. & Spencer, S. L. Endogenous replication stress in mother cells leads to quiescence of daughter cells. Cell Reports 19, 1351–1364 (2017)
Maki, C. G. & Howley, P. M. Ubiquitination of p53 and p21 is differentially affected by ionizing and UV radiation. Mol. Cell. Biol. 17, 355–363 (1997)
Iwamoto, M., Björklund, T., Lundberg, C., Kirik, D. & Wandless, T. J. A general chemical method to regulate protein stability in the mammalian central nervous system. Chem. Biol. 17, 981–988 (2010)
LaBaer, J . et al. New functional activities for the p21 family of CDK inhibitors. Genes Dev. 11, 847–862 (1997)
Alt, J. R., Gladden, A. B. & Diehl, J. A. p21Cip1 Promotes cyclin D1 nuclear accumulation via direct inhibition of nuclear export. J. Biol. Chem. 277, 8517–23 (2002)
Chen, J. Y., Lin, J. R., Tsai, F. C. & Meyer, T. Dosage of Dyrk1a shifts cells within a p21-cyclin D1 signaling map to control the decision to enter the cell cycle. Mol. Cell 52, 87–100 (2013)
Sugimoto, M. et al. Activation of cyclin D1-kinase in murine fibroblasts lacking both p21Cip1 and p27Kip1. Oncogene 21, 8067–74 (2002)
Ferrell, J. E., Jr & Ha, S. H. Ultrasensitivity part II: multisite phosphorylation, stoichiometric inhibitors, and positive feedback. Trends Biochem. Sci. 39, 556–569 (2014)
Hanahan, D. & Weinberg, R. A. The hallmarks of cancer. Cell 100, 57–70 (2000)
Sakaue-Sawano, A. et al. Visualizing spatiotemporal dynamics of multicellular cell-cycle progression. Cell 132, 487–498 (2008)
Yusa, K., Rad, R., Takeda, J. & Bradley, A. Generation of transgene-free induced pluripotent mouse stem cells by the piggyBac transposon. Nat. Methods 6, 363–369 (2009)
Acknowledgements
We thank K. Aoki and M. Matsuda for EKAR sensors, J. Stewart-Ornstein and G. Lahav for the p21-and p53-tagged MCF7 cell line, J. Ferrell, K. Cimprich, S. Collins, A. Hayer, S. Cappell, L. Pack, C. Liu, Y. Fan, L. Daigh, A. Jaimovich and S. Spencer for discussions, and the Stanford Shared FACS Facility for cell sorting. This work was supported by the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2013R1A6A3A03025832) and NIGMS R01 grants (GM11837, GM063702 and PGM107615).
Author information
Authors and Affiliations
Contributions
H.Y. designed and carried out all experiments and data analysis. M.C. helped with the experimental set-up and idea for the cyclin D1, p21 and Rb staining experiments. M.C. and T.K. contributed to the image analysis. H.Y. and T.M. conceived the project and wrote the manuscript. All authors discussed the results and the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer Information Nature thanks R. Medema, J. Purvis and A. Raj for their contribution to the peer review of this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Figure 1 ERK activity in G2 and G0/G1 regulates CDK2inc and CDK2delay paths, respectively.
Single-cell CDK2 activity traces. Cells were selected if mitogens were transiently withdrawn for a 5 h period (marked in grey) ending 1–3 h before mitosis for G2 (left) or for a 5 h period starting 0–2 h after mitosis for G1 (right).
Extended Data Figure 2 Characterization of ERK sensor and classification of CDK2 paths.
a, Representation of ERK activity time courses at different concentrations of mitogens. b, Examples of single-cell time courses of ERK activity. Automatic detection of indicated features of ERK activity. c, Averaged ERK features shown in b. MCF10A cells expressing ERK sensor were monitored for more than 18 h. Data are mean ± s.e.m. (n = 4 biological replicates). d, e, Correlation between ERK activity, integrated over 5 h, and the fraction of nuclear ERK phosphorylation (d) or the levels of nuclear cyclin D1 protein (e). f, Examples of CDK2 and ERK single-cell traces classified by CDK2 paths. g, Single-cell traces of CDK2 and ERK activity in response to inhibition of MEK (PD0325901; 100 nM) showing that a peak of ERK activation during mitosis was not suppressed by MEK inhibition.
Extended Data Figure 3 ERK activity in G2 partially predicts CDK2 paths in daughter cells.
a, Single-cell traces of CDK2 and ERK activities. Mitogens were withdrawn 0–2 h after mitosis. b, Box plot of mean ERK activity during the G2 or G0/G1 phases based on CDK2 classification. The higher ERK activity in CDK2delay cells during G0/G1 compared to CDK2low cells suggests that ERK activity in G0/G1 regulates the delayed entry into the cell cycle. Yellow lines indicate the median, boxes denote the 25th and 75th percentiles, lines denote the total range for each population (n = 2,896 cells). c, Examples of ERK single-cell traces classified by CDK2 paths in daughter cells. d, Bar graph of differences in integrated ERK activity during the G0/G1 phases based on CDK2 classification. Corresponding fold changes in cyclin D1 were calculated based on Extended Data Fig. 2e. Data are mean ± s.d. (n = 3 biological replicates). e, Analysis of the fraction of daughter pairs from the same and different mothers that have the same CDK2 path. Data are mean ± s.d. (n = 24 wells (each well; n > 100 cells)). f, g, Odds ratio analysis showing how well the percentile of ERK activity in G2 for varying mitogen availability (f) and temporal proximity to mitosis (g) predicts cell-cycle entry in daughter cells. Strong prediction values are shown in blue, and random chance is marked as a purple dashed line. Data are mean ± s.d. (n = 3 (f) and n = 2 (g) biological replicates).
Extended Data Figure 4 p53 signalling in mother cells controls the CDK2inc path in daughter cells through p21.
a, Single-cell CDK2 activity traces aligned to the time of mitosis in MCF7 cells. b, c, CDK2 activity traces aligned to the time of mitosis after siRNA knockdown of p53 or control siRNA (b) or in wild-type and p53-knockout (TP53−/−) cell lines (c). d, e, CDK2 activity traces of cells exposed to 1 h application of p53 activator (10 μM nutlin-3, 10 μM tenovin-6) 2–3 h before mitosis (marked in grey) in a wild-type cell line (d) or in a p21-knockout (CDKN1A−/−) cell line (e). f, Cumulative distribution functions of S/G2/M duration (time between geminin rising point and mitosis) as a function of the strength of a 20-min NCS pulse. MCF10A cells expressing the Fucci (geminin) reporter were pre-imaged for 13 h, then treated with the indicated concentration of NCS for 20 min and imaged for a further 48 h. Cells were selected when NCS was applied during S/G2 phase (control, n = 645 cells; 10 ng ml−1 NCS, 786 cells; 50 ng ml−1 NCS, 1,314 cells; 200 ng ml−1 NCS, 471 cells). g, Examples of CDK2 activity traces in response to a 20-min NCS pulse (200 ng ml−1) or a control pulse applied 9–11 h before mitosis in Fig. 2d.
Extended Data Figure 5 DNA damage after mitosis does not regulate CDK2inc path selection.
a, An example of cell phase gating using Hoechst and 5-ethynyl-20-deoxyuridine (EdU) staining. b, c, Box plot of nuclear 53BP1 (b) and γH2AX (c) puncta area in different cell-cycle phases with and without addition of a 20-min NCS (200 ng ml−1) pulse 8 h before fixation. Boxes reflect the 25th and 75th percentiles, whiskers denote the total range (53BP1; n > 2,800, γH2AX; n > 3,000 cells for each condition). d, Representative images of γH2AX and 53BP1 staining 8 h after a pulse of NCS (200 ng ml−1) for 20 min. Scale bar, 20 μm. e, Classification of CDK2inc and CDK2low cells after mitosis. f, Plot of nuclear γH2AX puncta area as a function of time relative to mitosis in unperturbed cells. Data are mean ± s.e.m. (n = 4 biological replicates). g, Histogram of nuclear Rb (Ser807/S811) intensity. h, Comparison of the E2F1 mRNA abundance by nuclear Rb (Ser807/S811) status (E2F1 is an E2F target). Data are mean ± s.d. (n = 10 wells (each well; n > 500 cells)). i, After selecting cells based on time since mitosis, G0/G1 cells were gated using Hoechst and EdU staining (top), and then cells in G0/G1 were further classified into hypo- or hyper-Rb population (bottom). j, Bar graph of nuclear γH2AX and 53BP1 puncta area. Data are mean ± s.e.m. (control; n = 4, NCS; n = 3 biological replicates). P values were calculated by unpaired two-tailed t-tests.
Extended Data Figure 6 Inhibition of the DNA-damage sensing ATM, ATR and DNA-PK kinases in daughter cells does not regulate CDK2inc path selection.
a, CDK2 activity traces of cells exposed to inhibitors of ATM (5 μM KU-60019), ATR (2 μM AZ-20), and DNA-PK (1 μM NU-7441) (marked in grey), starting 5 h after a control pulse or a 20-min NCS pulse (marked in purple). Cells undergoing mitosis within 1 h before drug treatment were selected for analysis. b, Averaged p21 intensity traces in response to a 20-min NCS pulse (200 ng ml−1) in MCF7 cells. Cells were selected for which NCS was applied during a 4-h time window after mitosis. Data are mean ± 95% confidence intervals (n = 79 cells).
Extended Data Figure 7 CCND1 mRNA transduces mitogen signalling from mother to daughter cells.
a, Control and additional experiments to Fig. 3d. CDK2 activity traces of cells exposed to a control pulse or a 20-min NCS pulse (10 ng ml−1, marked in purple), followed by a 5-h incubation in different concentrations of mitogens (marked in grey). After the 5-h incubation, cells were replenished with full growth media. Cells undergoing mitosis within 1 h of replacement with full growth media were selected. b, Individual traces of CDK2 activity showing the time window when mitogens were withdrawn or MEK inhibited (100 nM PD0325901) for 5 h until cells were fixed (marked in grey). Cells that were fixed during the first 2 h after mitosis were selected. c, Percentage of hyper-Rb (Ser807/S811) in response to mitogen withdrawal and MEK inhibition. Data are mean ± s.d. (n = 2 biological replicates). d, Representative images of mRNA and protein levels of cyclin D1 (CCND1) and p21 (CDKN1A) after a 5-h period of mitogen withdrawal or MEK inhibition. Scale bar, 50 μm. e, mRNA and nuclear protein levels of p21 in response to mitogen withdrawal and MEK inhibition. Data are mean ± s.d. (n = 2 biological replicates). f, Expression level of nuclear cyclin D1 protein, normalized by initial level (time 0 h), in response to the translation inhibitor, cycloheximide (10 μg ml−1) (n = 2 biological replicates). g, Cyclin D1 protein (left) and mRNA (CCND1; right) levels, normalized by initial level (time 0 h), after mitogen withdrawal. The half-life of CCND1 mRNA was measured with or without the transcription inhibitor actinomycin D (ActD; 5 μg ml−1) (n = 2 biological replicates).
Extended Data Figure 8 p53 protein transduces stress signalling from mother to daughter cells.
a, Individual traces of CDK2 activity showing the time window when a 20-min NCS pulse (200 ng ml−1) (marked in grey) was applied to mother cells. Cells that were fixed during the first 2 h after mitosis were selected. b, Percentage of hyper-Rb (Ser807/S811). Data are mean ± s.d. (n = 4 biological replicates). c, Representative images of cyclin D1 (CCND1) mRNA and protein levels, and p21 (CDKN1A) mRNA and protein levels after a 20-min pulse of NCS. Scale bar, 50 μm. d, mRNA and nuclear protein levels of cyclin D1 in G1 phase after exposure of mother cells to a 20-min pulse of NCS (200 ng ml−1). Data are mean ± s.d. (n = 2 biological replicates). e–g, Expression levels of nuclear p21 (CDKN1A) mRNA (e) and protein (f), and p53 protein (g), normalized by initial level (time 0 h). Cells were treated with either cycloheximide (10 μg ml−1) or ActD (5 μg ml−1), within 1 h of the application of the 20-min pulse of the indicated concentration of NCS (n = 2 biological replicates).
Extended Data Figure 9 Cyclin D and p21 are opposing regulators of CDK2 path selection.
a, Single-cell traces of CDK2 activity after siRNA knockdown of cyclins D1, D2 and D3 (combined), siRNA knockdown of p21, or control siRNA. b, c, Chemical-induced rapid expression of cyclin D1 or p21 controls the CDK2 paths in daughter cells. b, Schematic of chemical-induced DHFR-mCherry-cyclin D1 or DHFR-mCherry-p21 constructs. c, CDK2 activity traces aligned to the time of mitosis. Only cells that were treated with DMSO, 0.05 μM TMP or 5 μM TMP 1–5 h before mitosis (marked in yellow) were selected for plotting. Note that TMP was not washed out (marked in grey).
Extended Data Figure 10 Stoichiometric p21-mediated inhibition of cyclin D1–CDK4 is ultrasensitive.
a, Histograms of the calibrated cyclin D1/p21 ratio in CDK2inc and CDK2low cells during a 2–4 h time window after mitosis (CDK2inc; n = 2,162, CDK2low; n = 932 cells). b–d, Histogram of Rb (Ser807/811) (b), histogram of nuclear cyclin D1 and p21 protein (c), and nuclear cyclin D1–p21 map colour-coded by percentage of hyper-Rb (Ser807/811) (d, top) and relative density (d, bottom) after 5 h incubation of the indicated mitogen concentration. e, Percentage of hyper-Rb (Ser807/S811) as a function of the nuclear cyclin D1/p21 ratio at three different fixed concentrations of p21. Intermediate and high p21 levels are twofold and fivefold relative to low p21 level, respectively. The data show a clear increase in ultrasensitivity for higher absolute p21 levels.
Supplementary information
Examples of ERK and Cdk2 activities classified by three different Cdk2 paths.
Top: time-lapse microscopy of DHB-mCherry and processed ERK images. Bottom: traces of Cdk2 activity (green) and ERK activity (Red). Scale bar is 10 µm. (MOV 4117 kb)
Rights and permissions
About this article
Cite this article
Yang, H., Chung, M., Kudo, T. et al. Competing memories of mitogen and p53 signalling control cell-cycle entry. Nature 549, 404–408 (2017). https://doi.org/10.1038/nature23880
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature23880
This article is cited by
-
Cyclers’ kinases in cell division: from molecules to cancer therapy
Cell Death & Differentiation (2023)
-
Non-canonical pathway for Rb inactivation and external signaling coordinate cell-cycle entry without CDK4/6 activity
Nature Communications (2023)
-
Cell cycle gene regulation dynamics revealed by RNA velocity and deep-learning
Nature Communications (2022)
-
Ammonia promotes the proliferation of bone marrow-derived mesenchymal stem cells by regulating the Akt/mTOR/S6k pathway
Bone Research (2022)
-
Relating individual cell division events to single-cell ERK and Akt activity time courses
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.