Abstract
Inflammatory gene expression following genotoxic cancer therapy is well documented, yet the events underlying its induction remain poorly understood. Inflammatory cytokines modify the tumour microenvironment by recruiting immune cells and are critical for both local and systemic (abscopal) tumour responses to radiotherapy1. A poorly understood feature of these responses is the delayed onset (days), in contrast to the acute DNA-damage responses that occur in minutes to hours. Such dichotomous kinetics implicate additional rate-limiting steps that are essential for DNA-damage-induced inflammation. Here we show that cell cycle progression through mitosis following double-stranded DNA breaks leads to the formation of micronuclei, which precede activation of inflammatory signalling and are a repository for the pattern-recognition receptor cyclic GMP–AMP synthase (cGAS). Inhibiting progression through mitosis or loss of pattern recognition by stimulator of interferon genes (STING)–cGAS impaired interferon signalling. Moreover, STING loss prevented the regression of abscopal tumours in the context of ionizing radiation and immune checkpoint blockade in vivo. These findings implicate temporal modulation of the cell cycle as an important consideration in the context of therapeutic strategies that combine genotoxic agents with immune checkpoint blockade.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Barker, H. E., Paget, J. T. E., Khan, A. A. & Harrington, K. J. The tumour microenvironment after radiotherapy: mechanisms of resistance and recurrence. Nat. Rev. Cancer 15, 409–425 (2015)
Jonathan, E. C., Bernhard, E. J. & McKenna, W. G. How does radiation kill cells? Curr. Opin. Chem. Biol. 3, 77–83 (1999)
Rodier, F. et al. Persistent DNA damage signalling triggers senescence-associated inflammatory cytokine secretion. Nat. Cell Biol. 11, 973–979 (2009)
Di Maggio, F. M. et al. Portrait of inflammatory response to ionizing radiation treatment. J. Inflamm. 12, http://dx.doi.org/10.1186/s12950-015-0058-3 (2015)
Crasta, K. et al. DNA breaks and chromosome pulverization from errors in mitosis. Nature 482, 53–58 (2012)
Terradas, M., Martín, M., Tusell, L. & Genescà, A. DNA lesions sequestered in micronuclei induce a local defective-damage response. DNA Repair 8, 1225–1234 (2009)
Terradas, M., Martín, M., Tusell, L. & Genescà, A. Genetic activities in micronuclei: is the DNA entrapped in micronuclei lost for the cell? Mutat. Res. 705, 60–67 (2010)
Ishikawa, H., Ma, Z. & Barber, G. N. STING regulates intracellular DNA-mediated, type I interferon-dependent innate immunity. Nature 461, 788–792 (2009)
Sun, L., Wu, J., Du, F., Chen, X. & Chen, Z. J. Cyclic GMP–AMP synthase is a cytosolic DNA sensor that activates the type I interferon pathway. Science 339, 786–791 (2013)
Erdal, E., Haider, S., Rehwinkel, J., Harris, A. L. & McHugh, P. J. A prosurvival DNA damage-induced cytoplasmic interferon response is mediated by end resection factors and is limited by Trex1. Genes Dev. 31, 353–369 (2017)
Lan, Y. Y., Londoño, D., Bouley, R., Rooney, M. S. & Hacohen, N. Dnase2a deficiency uncovers lysosomal clearance of damaged nuclear DNA via autophagy. Cell Reports 9, 180–192 (2014)
Ferguson, B. J., Mansur, D. S., Peters, N. E., Ren, H. & Smith, G. L. DNA-PK is a DNA sensor for IRF-3-dependent innate immunity. Life Sci. 1, e00047 (2012)
Hu, B. et al. The DNA-sensing AIM2 inflammasome controls radiation-induced cell death and tissue injury. Science 354, 765–768 (2016)
Karpova, A. Y., Trost, M., Murray, J. M., Cantley, L. C. & Howley, P. M. Interferon regulatory factor-3 is an in vivo target of DNA-PK. Proc. Natl Acad. Sci. USA 99, 2818–2823 (2002)
Woo, S.-R. et al. STING-dependent cytosolic DNA sensing mediates innate immune recognition of immunogenic tumors. Immunity 41, 830–842 (2014)
Deng, L. et al. STING-dependent cytosolic DNA sensing promotes radiation-induced type I interferon-dependent antitumor immunity in immunogenic tumors. Immunity 41, 843–852 (2014)
Andersen, J., VanScoy, S., Cheng, T.-F., Gomez, D. & Reich, N. C. IRF-3-dependent and augmented target genes during viral infection. Genes Immun. 9, 168–175 (2008)
Grandvaux, N. et al. Transcriptional profiling of interferon regulatory factor 3 target genes: direct involvement in the regulation of interferon-stimulated genes. J. Virol. 76, 5532–5539 (2002)
Hatch, E. M., Fischer, A. H., Deerinck, T. J. & Hetzer, M. W. Catastrophic nuclear envelope collapse in cancer cell micronuclei. Cell 154, 47–60 (2013)
Irianto, J. et al. DNA damage follows repair factor depletion and portends genome variation in cancer cells after pore migration. Curr. Biol. 27, 210–223 (2017)
Härtlova, A. et al. DNA damage primes the type I interferon system via the cytosolic DNA sensor STING to promote anti-microbial innate immunity. Immunity 42, 332–343 (2015)
Schaue, D. & McBride, W. H. Opportunities and challenges of radiotherapy for treating cancer. Nat. Rev. Clin. Oncol. 12, 527–540 (2015)
Postow, M. A. et al. Immunologic correlates of the abscopal effect in a patient with melanoma. N. Engl. J. Med. 366, 925–931 (2012)
Twyman-Saint Victor, C. et al. Radiation and dual checkpoint blockade activate non-redundant immune mechanisms in cancer. Nature 520, 373–377 (2015)
Benci, J. L. et al. Tumor interferon signaling regulates a multigenic resistance program to immune checkpoint blockade. Cell 167, 1540–1554 (2016)
Rodriguez-Ruiz, M. E. et al. Abscopal effects of radiotherapy are enhanced by combined immunostimulatory mAbs and are dependent on CD8 T cells and crosspriming. Cancer Res. 76, 5994–6005 (2016)
Syljuåsen, R. G., Jensen, S., Bartek, J. & Lukas, J. Adaptation to the ionizing radiation-induced G2 checkpoint occurs in human cells and depends on checkpoint kinase 1 and Polo-like kinase 1 kinases. Cancer Res. 66, 10253–10257 (2006)
Deckbar, D., Jeggo, P. A. & Löbrich, M. Understanding the limitations of radiation-induced cell cycle checkpoints. Crit. Rev. Biochem. Mol. Biol. 46, 271–283 (2011)
Harding, S. M., Boiarsky, J. A. & Greenberg, R. A. ATM dependent silencing links nucleolar chromatin reorganization to DNA damage recognition. Cell Reports 13, 251–259 (2015)
Sanjana, N. E., Shalem, O. & Zhang, F. Improved vectors and genome-wide libraries for CRISPR screening. Nat. Methods 11, 783–784 (2014)
Cho, N. W., Dilley, R. L., Lampson, M. A. & Greenberg, R. A. Interchromosomal homology searches drive directional ALT telomere movement and synapsis. Cell 159, 108–121 (2014)
Coschi, C. H. et al. Mitotic chromosome condensation mediated by the retinoblastoma protein is tumor-suppressive. Genes Dev. 24, 1351–1363 (2010)
Harding, S. M. & Bristow, R. G. Discordance between phosphorylation and recruitment of 53BP1 in response to DNA double-strand breaks. Cell Cycle 11, 1432–1444 (2012)
Diner, E. J. et al. The innate immune DNA sensor cGAS produces a noncanonical cyclic dinucleotide that activates human STING. Cell Reports 3, 1355–1361 (2013)
Acknowledgements
We thank members of the Greenberg laboratory for critical discussion and A. Jackson (University of Edinburgh, Scotland) for sharing unpublished results. We are also grateful to R. Vance (UC Berkeley) for providing the IFNβ-promoter-driven plasmid that we subsequently modified to drive eGFP expression. This work was supported by NIH grants CA17494, GM101149, and CA138835 (to R.A.G.), PSOC U54-CA193417 to (D.E.D. and R.A.G.), and by funds from the Basser Center for BRCA.
Author information
Authors and Affiliations
Contributions
S.M.H. and R.A.G. designed the study and wrote the manuscript. S.M.H. performed most of the experiments. J.L.B. designed and performed in vivo experiments described in Fig. 4 and Extended Data Fig. 6. J.I. designed and performed experiments described in Extended Data Fig. 4d, e. A.J.M. and D.E.D. provided guidance for the design of experiments and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer Information Nature thanks N. Gekara, K.-P. Hopfner and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
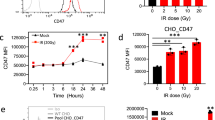
Extended Data Figure 1 Non-homologous end-joining inhibition impedes DSB-induced inflammatory signalling.
a, Total STAT1 protein levels are increased in a time-dependent manner after ionizing radiation (representative of three biological replicates). b, U2OS cells were monitored for STAT1 activation during recovery from I-PpoI nuclease (representative of two biological replicates). c, Gene expression changes were monitored for indicated genes during recovery from I-PpoI damage in MCF10A cells (see Fig. 1d). Error bars represent s.e.m. of 2–3 biological replicates. d, Prostate epithelial cells (PREC) were analysed for STAT1 phosphorylation at 6 days following 10 Gy (n = 2 biological replicates). e, STAT1 phosphorylation following IFNβ1 treatment is not DNA-PK or CDK1 dependent (n = 2 biological replicates). f, pSTAT1 activation measured by western blot is unchanged by ATMi. DNA-PKi and ATMi block phosphorylation of DNA-PKcs S2056 and pKAP1 S824, respectively, as measured by western blot.
Extended Data Figure 2 Prevention of mitotic entry and cell cycle progression impairs ionizing-radiation-induced STAT1 activation.
a, Cell cycle profiles of MCF10A cells were monitored by flow cytometry of propidium-iodide-stained cells treated as indicated (n = 3 biological replicates). Error bars represent s.e.m. b, U2OS cells were monitored for STAT1 activation in the presence of the indicated inhibitors (n = 2 biological replicates). c, H3 (Ser10) phosphorylation in MCF10A cells was measured by flow cytometry and expressed as a percentage of total single cells. Error bars represent s.e.m. of at least two biological replicates. Western blots show loss of STAT1 activation in treatments corresponding to the flow cytometry data. d, Example dot plots of pH3 (H3P) flow cytometry showing the gating strategy using a sample with and without the conjugated pH3 antibody. An example propidium-iodide-staining gating strategy is also shown.
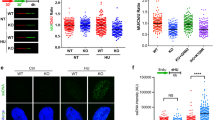
Extended Data Figure 3 Loss of cGAS–STING signalling in micronuclei impedes ionizing-radiation-induced inflammatory gene activation with minimal effect on senescence.
a, RT–qPCR of CCL5 and ISG56 at 6 days following 20 Gy in parental MCF10A cells untreated or treated with CDK1i (RO-3306) or derivatives harbouring deletion of cGAS or STING. Error bars represent s.e.m. of three biological replicates. b, Knockouts for two separate CRISPR–Cas9 sgRNA for cGAS or STING cause similar reductions in STAT1 signalling (representative of two biological replicates). c, Senesence-associated β-galactosidase staining at 6 days following indicated treatments. Error bars represent s.e.m. of at least two biological replicates. d, Representative immunofluorescent staining of cGAS shows loss of staining in cGAS-knockout cells. Scale bar, 10 μm. e, Immunofluorescent staining shows co-staining of lamin B2 in cGAS positive micronuclei. Scale bar, 10 μm.
Extended Data Figure 4 cGAS dynamically relocalizes to micronuclei following mitosis, during migration and after Aurora B inhibition.
a, Cells in Fig. 2e were analysed for the fraction of cGAS-positive micronuclei following release from CDK1i. Error bars represent s.e.m. of three biological replicates. b, mCherry–cGAS-expressing cells were monitored by immunofluorescence following 10 Gy. Arrowhead indicates mCherry–cGAS-positive micronucleus (representative of three independent experiments). c, Histogram represents the fraction of daughter cells with cGAS-positive micronuclei or that underwent apoptosis during live-cell imaging. All non-apoptotic daughter cells were micronucleated after division. Error bars are s.e.m. of three biological replicates (n = 99 total daughters). d, Schematic of nuclear migration transwell system. Blue, DAPI; green, lamin A; red, lamin B. cGAS is shown in grayscale. e, Quantification of aberrant nuclei (nuclear blebs and micronuclei) that are cGAS-positive in the transwell migration assay. f, Immunofluorescent staining in an untreated and two representative cells after six-day treatment with Aurora B kinase inhibition (AuroraBi). Scale bar is 10 μm and similar patterns were observed in two independent experiments. g, Representative (n = 3) western blot of STAT1 activation in non-irradiated cells treated with Aurora B inhibitor for 3 or 6 days.
Extended Data Figure 5 cGAS–STING-driven inflammatory signalling occurs in micronucleated cells.
a, Representative images of pIFNβ–GFP reporter cells treated as indicated. Scale bar, 10 μm. Quantification is as described in the Methods and error bars represent s.e.m. of two biological replicates. b, Mean nuclear intensity of pIRF3 staining was quantified. P values are based on pooled data from three independent experiments and calculated by one-way ANOVA. c, Representative (n = 2) western blot of STAT1 activation following transfection of herring testis DNA (HT-DNA) in indicated CRISPR–Cas9 knockout MCF10A cell lines shows STAT1 activation in DNA-PKcs- and all NHEJ-deficient cells, but not in cGAS STING knockouts.
Extended Data Figure 6 STING knockout in B16-F10 impairs ionizing-radiation-induced inflammatory gene expression in vitro and the abscopal response after in vivo irradiation of contralateral tumour.
a, b, Radiotherapy- (a) and cGAMP-induced (b) gene induction is absent in STING knockout B16 cells. c, Injection of B16 parental or STING knockout cells (with or without irradiation) without combination anti-CTLA4 treatment is insufficient to induce an abscopal effect. d, Overall survival of mice when B16 parental or STING knockout were injected after indicated treatment. All mice received anti-CTLA4 antibody. P value calculated by log-rank test. e, B16 tumours are injected into opposite flanks of mice and treated as indicated. The index tumour is irradiated with 20 Gy and both the index and abscopal tumours are measured starting on day 11 after injection. f, Irradiation of the index tumour leads to an abscopal response that is dependent on STING (left panel). This response is not seen in unirradiated mice (right panel).
Supplementary information
Supplementary Figure
This file contains source data of western blot scans.
Supplementary Tables
This file contains supplementary tables 1-3 regarding data on Target sequences for CRISPR gRNAs, antibodies and corresponding dilutions used and primer sequences for qPCR.
Video 1: Representative video of mitotic cell becoming micronucleated shown in Fig 3e.
H2B-GFP (green) and mCherry-cGAS (red) were imaged as described in the text. The inset timestamp is in hh:mm and rendered at 1 frame per second.
Video 2: Representative video of post-mitotic apoptotic cell as quantified in Fig 3e.
H2B-GFP (green) and mCherry-cGAS (red) were imaged as described in the text. The inset timestamp is in hh:mm and rendered at 1 frame per second.
Rights and permissions
About this article
Cite this article
Harding, S., Benci, J., Irianto, J. et al. Mitotic progression following DNA damage enables pattern recognition within micronuclei. Nature 548, 466–470 (2017). https://doi.org/10.1038/nature23470
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature23470
This article is cited by
-
Radiomics signature for dynamic changes of tumor-infiltrating CD8+ T cells and macrophages in cervical cancer during chemoradiotherapy
Cancer Imaging (2024)
-
Harnessing innate immune pathways for therapeutic advancement in cancer
Signal Transduction and Targeted Therapy (2024)
-
Scrambling the genome in cancer: causes and consequences of complex chromosome rearrangements
Nature Reviews Genetics (2024)
-
The two sides of chromosomal instability: drivers and brakes in cancer
Signal Transduction and Targeted Therapy (2024)
-
Activating STING/TBK1 suppresses tumor growth via degrading HPV16/18 E7 oncoproteins in cervical cancer
Cell Death & Differentiation (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.