Abstract
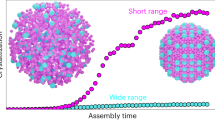
Crystallization of colloidal nanocrystals into superlattices represents a practical bottom-up process with which to create ordered metamaterials with emergent functionalities1,2,3. With precise control over the size, shape and composition of individual nanocrystals4,5,6, various single- and multi-component nanocrystal superlattices have been produced, the lattice structures and chemical compositions of which can be accurately engineered7,8,9. Nanocrystal superlattices are typically prepared by carefully controlling the assembly process through solvent evaporation or destabilization2,10,11,12,13,14,15 or through DNA-guided crystallization16,17,18. Slow solvent evaporation or cooling of nanocrystal solutions (over hours or days) is the key element for successful crystallization processes10,18. Here we report the rapid growth (seconds) of micrometre-sized, face-centred-cubic, three-dimensional nanocrystal superlattices during colloidal synthesis at high temperatures (more than 230 degrees Celsius). Using in situ small-angle X-ray scattering, we observe continuous growth of individual nanocrystals within the lattices, which results in simultaneous lattice expansion and fine nanocrystal size control due to the superlattice templates. Thermodynamic models demonstrate that balanced attractive and repulsive interparticle interactions dictated by the ligand coverage on nanocrystal surfaces and nanocrystal core size are responsible for the crystallization process. The interparticle interactions can also be controlled to form different superlattice structures, such as hexagonal close-packed lattices. The rational assembly of various nanocrystal systems into novel materials is thus facilitated for both fundamental research and for practical applications in the fields of magnetics19, electronics3 and catalysis20.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Boles, M. A., Engel, M. & Talapin, D. V. Self-assembly of colloidal nanocrystals: from intricate structures to functional materials. Chem. Rev. 116, 11220–11289 (2016)
Dong, A. G., Chen, J., Vora, P. M., Kikkawa, J. M. & Murray, C. B. Binary nanocrystal superlattice membranes self-assembled at the liquid–air interface. Nature 466, 474–477 (2010)
Cargnello, M. et al. Substitutional doping in nanocrystal superlattices. Nature 524, 450–453 (2015)
Park, J. et al. One-nanometer-scale size-controlled synthesis of monodisperse magnetic iron oxide nanoparticles. Angew. Chem. Int. Ed. 44, 2872–2877 (2005)
Ye, X. C. et al. Competition of shape and interaction patchiness for self-assembling nanoplates. Nat. Chem. 5, 466–473 (2013)
Sun, S. H., Murray, C. B., Weller, D., Folks, L. & Moser, A. Monodisperse FePt nanoparticles and ferromagnetic FePt nanocrystal superlattices. Science 287, 1989–1992 (2000)
Murray, C. B., Kagan, C. R. & Bawendi, M. G. Self-organization of CdSe nanocrystallites into three-dimensional quantum-dot superlattices. Science 270, 1335–1338 (1995)
Shevchenko, E. V., Talapin, D. V., Kotov, N. A., O’Brien, S. & Murray, C. B. Structural diversity in binary nanoparticle superlattices. Nature 439, 55–59 (2006)
Tian, Y. et al. Lattice engineering through nanoparticle-DNA frameworks. Nat. Mater. 15, 654–661 (2016)
Shevchenko, E. V., Talapin, D. V., Murray, C. B. & O’Brien, S. Structural characterization of self-assembled multifunctional binary nanoparticle superlattices. J. Am. Chem. Soc. 128, 3620–3637 (2006)
Jiang, Z. et al. Subnanometre ligand-shell asymmetry leads to Janus-like nanoparticle membranes. Nat. Mater. 14, 912–917 (2015)
Geuchies, J. J. et al. In situ study of the formation mechanism of two-dimensional superlattices from PbSe nanocrystals. Nat. Mater. 15, 1248–1254 (2016)
Weidman, M. C., Smilgies, D. M. & Tisdale, W. A. Kinetics of the self-assembly of nanocrystal superlattices measured by real-time in situ X-ray scattering. Nat. Mater. 15, 775–781 (2016)
Rupich, S. M., Shevchenko, E. V., Bodnarchuk, M. I., Lee, B. & Talapin, D. V. Size-dependent multiple twinning in nanocrystal superlattices. J. Am. Chem. Soc. 132, 289–296 (2010)
de Nijs, B. et al. Entropy-driven formation of large icosahedral colloidal clusters by spherical confinement. Nat. Mater. 14, 56–60 (2015)
Nykypanchuk, D., Maye, M. M., van der Lelie, D. & Gang, O. DNA-guided crystallization of colloidal nanoparticles. Nature 451, 549–552 (2008)
Park, S. Y. et al. DNA-programmable nanoparticle crystallization. Nature 451, 553–556 (2008)
Auyeung, E. et al. DNA-mediated nanoparticle crystallization into Wulff polyhedra. Nature 505, 73–77 (2014)
Chen, J. et al. Bistable magnetoresistance switching in exchange-coupled CoFe2O4-Fe3O4 binary nanocrystal superlattices by self-assembly and thermal annealing. ACS Nano 7, 1478–1486 (2013)
Zhao, H. et al. Reversible trapping and reaction acceleration within dynamically self-assembling nanoflasks. Nat. Nanotechnol. 11, 82–88 (2016)
LaMer, V. K. & Dinegar, R. H. Theory, production and mechanism of formation of monodispersed hydrosols. J. Am. Chem. Soc. 72, 4847–4854 (1950)
Erdemir, D., Lee, A. Y. & Myerson, A. S. Nucleation of crystals from solution: classical and two-step models. Acc. Chem. Res. 42, 621–629 (2009)
Xia, Y. N., Xiong, Y. J., Lim, B. & Skrabalak, S. E. Shape-controlled synthesis of metal nanocrystals: simple chemistry meets complex physics? Angew. Chem. Int. Ed. 48, 60–103 (2009)
Jones, A. G. & Mullin, J. W. Programmed cooling crystallization of potassium sulfate solutions. Chem. Eng. Sci. 29, 105–118 (1974)
He, G. W. et al. Direct growth of g-glycine from neutral aqueous solutions by slow, evaporation-driven crystallization. Cryst. Growth Des. 6, 1746–1749 (2006)
Smith, D. K., Goodfellow, B., Smilgies, D. M. & Korgel, B. A. Self-assembled simple hexagonal AB2 binary nanocrystal superlattices: SEM, GISAXS, and defects. J. Am. Chem. Soc. 131, 3281–3290 (2009)
Murray, C. B., Kagan, C. R. & Bawendi, M. G. Synthesis and characterization of monodisperse nanocrystals and close-packed nanocrystal assemblies. Annu. Rev. Mater. Sci. 30, 545–610 (2000)
Kim, S. W. et al. Synthesis of monodisperse palladium nanoparticles. Nano Lett. 3, 1289–1291 (2003)
Cargnello, M. et al. Control of metal nanocrystal size reveals metal-support interface role for ceria catalysts. Science 341, 771–773 (2013)
Yang, Y. et al. Entropic ligands for nanocrystals: from unexpected solution properties to outstanding processability. Nano Lett. 16, 2133–2138 (2016)
Cargnello, M., Doan-Nguyen, V. V. T. & Murray, C. B. Engineering uniform nanocrystals: mechanism of formation and self-assembly into bimetallic nanocrystal superlattices. AIChE J. 62, 392–398 (2015)
Peng, S., Wang, C., Xie, J. & Sun, S. H. Synthesis and stabilization of monodisperse Fe nanoparticles. J. Am. Chem. Soc. 128, 10676–10677 (2006)
Ganguly, S. & Brock, S. L. Toward nanostructured thermoelectrics: synthesis and characterization of lead telluride gels and aerogels. J. Mater. Chem. 21, 8800–8806 (2011)
Ilavsky, J. & Jemian, P. R. Irena: tool suite for modeling and analysis of small-angle scattering. J. Appl. Cryst. 42, 347–353 (2009)
Israelachvili, J. N. Intermolecular and Surface Forces 3rd edn, Ch. 13 (Academic Press, 2011)
Visser, J. On Hamaker constants: a comparison between Hamaker constants and Lifshitz–Van der Waals constants. Adv. Colloid Interf. Sci. 3, 331–363 (1972)
Glicksman, M. Plasmas in solids. Solid State Phys. 26, 275–427 (1971)
Bishop, K. J. M., Wilmer, C. E., Soh, S. & Grzybowski, B. A. Nanoscale forces and their uses in self-assembly. Small 5, 1600–1630 (2009)
Kim, J. U. & Matsen, M. W. Interaction between polymer-grafted particles. Macromolecules 41, 4435–4443 (2008)
Acknowledgements
This work was supported by the US Department of Energy (DOE), Laboratory Directed Research and Development programme at SLAC National Accelerator Laboratory, under contract DE-AC02-76SF00515. In situ SAXS experiments were performed at the Beamline 1-5 at the Stanford Synchrotron Radiation Lightsource (SSRL) of the SLAC National Accelerator Laboratory, and use of the SSRL was supported by the DOE Office of Science, Office of Basic Energy Sciences under contract number DE-AC02-76SF00515. I.S.M. was supported by the Department of Defense through the National Defense Science & Engineering Graduate Fellowship Program, and by the Fannie and John Hertz Foundation through a Hertz Foundation Fellowship. M.C. acknowledges support from the School of Engineering at Stanford University and from a Terman Faculty Fellowship. The electron microscopy characterization of nanocrystals and superlattices was performed at the Stanford Nano Shared Facilities (SNSF) at Stanford University. The work at the Center for Nanoscale Materials of Argonne National Laboratory was supported by the DOE Office of Science under contract number DE-AC02-06CH11357. L.W. thanks T. J. Dunn from SSRL for his assistance during the SAXS measurements. M.C. thanks N. Abbott (University of Wisconsin), D. Talapin (University of Chicago) and C. B. Murray (University of Pennsylvania) for discussions.
Author information
Authors and Affiliations
Contributions
L.W., M.C. and C.J.T. conceived and designed the experiments. L.W. carried out the in situ experiments and analysed the SAXS data. L.W., J.J.W. and I.S.M. performed the ex situ characterizations of nanocrystals and superlattices. B.T.D. contributed to data analysis. J.Q. analysed interparticle potentials. M.C. and C.J.T. supervised the entire project. L.W. and M.C. wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer Information Nature thanks E. Kumacheva, D. Vanmaekelbergh and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Figure 1 In situ SAXS characterization of Pd nanocrystals during the synthesis.
a, A photograph of the in situ reaction setup at Beamline 1-5 of SSRL at SLAC National Accelerator Laboratory. The inset shows a photograph of the custom-made flask reactor. To obtain optimized signal-to-noise ratio for SAXS measurements, the thickness of the two flat glass windows in the reactor is set to be 0.5 mm and the distance between two windows is 4 mm. b, One-dimensional SAXS data of 1-octadecene solvent in the flask reactor at different temperatures used for background subtraction. c, One-dimensional SAXS patterns of the in situ synthesized Pd nanocrystals before superlattice formation. The increase of scattering at low q is due to the formation of nanocrystals. We detect the onset of nanocrystal formation at about 203 °C and this reaction point is set as time 0 (0 s). d, Time-resolved one-dimensional SAXS patterns of the Pd superlattices. With increased reaction time, the diffraction peaks shift to lower q, suggesting the lattice expansion caused by the increased size of individual Pd nanocrystals. In the meantime, peak intensities increase as the size of nanocrystals increase. All the patterns shown here are not background-subtracted. e, Reaction temperature versus time during the in situ experiment. All experiments discussed in this work show similar reaction temperature profiles. (a.u., arbitrary units.)
Extended Data Figure 2 Quantitative analysis of the SAXS data.
a, One-dimensional background-subtracted SAXS patterns of the in situ synthesized Pd nanocrystals before the formation of superlattices. Coloured plots are experimental results and black lines represent the corresponding fits. The SAXS intensity and fits are scaled for clarity. b, Number-weighted size distribution of the Pd nanocrystals from quantitative analysis. c, Quantitative comparison of the form factor from dispersed nanocrystals in solution at 66 s and 77 s. At 77 s, clear scattering peaks appear at q = 0.102 Å−1, 0.117 Å−1, 0.166 Å−1, 0.196 Å−1, 0.204 Å−1, which are indexed to the (111), (200), (220), (311), (222) diffraction planes of f.c.c. structure. The concentration of dispersed nanocrystals decreases by about 90% within 11 s owing to their assembly into f.c.c. superlattices. d, Representative structure factor S(q) of the formed superlattices at 88 s. The S(q) value is calculated by dividing the I(q) value by the form factor of the nanocrystals. The peak positions correspond well to a f.c.c. lattice. e, Change of the lattice parameter with reaction time. f, Change of the (111) peak intensity of the f.c.c. superlattices as the volume (size) of an individual Pd nanocrystal increases during the post-assembly growth. The increased diffraction intensity is due to an increase in the number of scattered electrons by the growing nanocrystals. g, Change of lattice parameter of the final superlattices with temperatures during the cooling process. The lattice parameter is almost constant during a wide range of temperatures, indicating that the temperature has negligible effect on the f.c.c. lattice constant.
Extended Data Figure 3 Size characterization of the final Pd nanocrystals synthesized from different acid ligands.
a, Hexanoic acid; b, oleic acid; c, 2-hexyldecanoic acid. (i), TEM images of the final Pd nanocrystals. The final nanocrystals are re-dispersed in hexane after oleylamine treatment. (ii), Two-dimensional SAXS patterns of the final Pd nanocrystals dispersed in hexane. (iii), Background-subtracted one-dimensional SAXS data of the final Pd nanocrystals (black plots) and their corresponding fits (red lines). (iv), Size distribution of the Pd nanocrystals obtained from TEM characterization (histogram and red lines) and SAXS fits (blue lines, normalized to the same maximum of the red line). The sizes from TEM are 8.0 ± 0.3 nm (a), 6.7 ± 0.2 nm (b), and 6.0 ± 0.3 nm (c). The sizes from SAXS analysis are 8.1 ± 0.4 nm (a), 6.8 ± 0.4 nm (b), and 6.1 ± 0.3 nm (c), which are in good agreement with TEM measurements.
Extended Data Figure 4 Characterization of ex situ templated growth using superlattices of 9.6-nm Pd nanocrystals.
a, TEM image of the templated superlattices. b, TEM image of the dispersed Pd nanocrystals after treatment with oleylamine. The histogram (inset) shows the Pd nanocrystals to be 9.6 ± 0.4 nm. c, d, SEM images of different domains of the templated superlattices showing different morphologies and facets. e, SEM image of a representative superlattice after the templated growth. f, TEM images of the disperse Pd nanocrystals after the templated growth. The inset histogram shows the nanocrystal size to be 11.9 ± 0.5 nm.
Extended Data Figure 5 SAXS characterization of the Pd nanocrystals and their superlattices synthesized using oleic acid as the surfactant.
a, Representative two-dimensional SAXS patterns at different reaction time showing the transition from diffuse scattering to well defined scattering rings. b, Background-subtracted one-dimensional SAXS patterns of the in situ synthesized Pd nanocrystals before the formation of superlattices. The coloured plots are experimental results and the black lines are their corresponding fits. c, One-dimensional SAXS patterns of the Pd nanocrystal superlattices during the in situ experiment. The peak positions show characteristics of f.c.c. structure with the ratios qn/q1 = 1,  ,
,  ,
,  and 2. The shifts of these peaks to lower q indicate lattice expansion by the growth of individual nanocrystals. d, Number-weighted size distribution of the Pd nanocrystals from quantitative analysis of the real-time SAXS data shown in b. e, Kinetics of the growth of Pd nanocrystals and their crystallization into superlattices over reaction time. Error bars result from the fits to the SAXS data.
and 2. The shifts of these peaks to lower q indicate lattice expansion by the growth of individual nanocrystals. d, Number-weighted size distribution of the Pd nanocrystals from quantitative analysis of the real-time SAXS data shown in b. e, Kinetics of the growth of Pd nanocrystals and their crystallization into superlattices over reaction time. Error bars result from the fits to the SAXS data.
Extended Data Figure 6 SAXS characterization of the Pd nanocrystals and their superlattices synthesized using 2-hexyldecanoic acid as the surfactant.
a, Representative two-dimensional SAXS patterns at different reaction times. b, Background-subtracted one-dimensional SAXS patterns of the in situ synthesized Pd nanocrystals before the formation of superlattices showing experimental results (coloured plots) and corresponding fits of nanocrystal form factor (black lines). c, One-dimensional SAXS patterns of the Pd nanocrystal superlattices during the in situ experiment with continuous shifts of the f.c.c. diffraction peaks. d, Number-weighted size distribution of the Pd nanocrystals from quantitative analysis of the SAXS data in b. e, Kinetics of the growth of Pd nanocrystals and their crystallization into superlattices over reaction time. The branched acid-capped nanocrystal superlattices are less stable at 280 °C and show an equilibrium state of about 30% nanocrystal in isolated form. Error bars result from the fits to the SAXS data. f, Comparison of the SAXS patterns of the final superlattices in solution at 280 °C and 50 °C. After cooling down to 50 °C, the dispersed nanocrystals recrystallized, as shown by the disappearance of scattering at low q from the form factor of the nanocrystals. Two additional peaks at 0.090 Å−1 and 0.101 Å−1 appear, owing to the formation of h.c.p. lattices. g, TEM image of small domains of the superlattices obtained at room temperature. In addition to large three-dimensional f.c.c. superlattices (see Fig. 2c and d), some small h.c.p. lattices are obtained from the recrystallization of the dispersed nanocrystals during the cooling process.
Extended Data Figure 7 Surface characterization of the final Pd nanocrystals synthesized using oleic acid as the surfactant.
a, FT-IR spectra of the as-synthesized Pd nanocrystals showing the absence of C=O stretching mode at about 1,707 cm−1 and the appearance of OCO stretching vibration at about 1,377 cm−1, indicating oleic acid bonding on the surface. b, Enlarged FT-IR spectra of a in wavenumbers 2,600–3,100 cm−1. The CH-stretching vibration of the alkyl group in oleic-acid-capped Pd nanocrystals are shifted to lower wavenumbers owing to the loss of conformational freedom by bonding on Pd nanocrystal surface. c, FT-IR spectra of the oleic acid ligands on Pd nanocrystals after different washing cycles. After the second wash, the alkyl group intensity drops substantially, indicating that most of the oleic acid is stripped off by the washing step owing to the weak bonding between oleic acid and the Pd nanocrystals. d, FT-IR of the Pd nanocrystals after oleylamine treatment. Even after four washing cycles, the oleylamine-capped nanocrystals are still well dispersed and the FT-IR spectrum shows strong CH-stretching vibrations.
Extended Data Figure 8 Characterization of the Pd nanocrystals synthesized with a 1:10 molar ratio of oleic acid:TOP and their superlattices.
a, One-dimensional SAXS patterns of the Pd nanocrystals in solution during the synthesis. All nanocrystals are well dispersed in the solution during the entire synthesis, showing only the form factor of dispersed nanocrystals in the SAXS data. b, Representative one-dimensional background-subtracted SAXS patterns at different reaction time (coloured plots) and their corresponding fits (black lines). c, Evolution of nanocrystal size over reaction time obtained from SAXS fits. Error bars result from the fits to the SAXS data. d, TEM image of the final Pd nanocrystals. e, Size distribution of the as-synthesized nanocrystals from TEM characterization (histogram and red line, 6.0 ± 0.3 nm) and SAXS measurement (blue line, 6.2 ± 0.4 nm). f, SAXS patterns of the obtained Pd nanocrystals after synthesis at 100 °C with and without adding extra oleic acid ligands. Adding extra oleic acid caused the assembly of Pd nanocrystals. g, Energy-dispersive X-ray spectroscopy spectra of the Pd nanocrystals synthesized before and after adding extra oleic acid in the nanocrystal solution. The P:Pd atomic ratio decreases from 0.19 to 0.12 after adding extra oleic acid, indicating less coverage of TOP on the nanocrystal surface. h, Schematic of the dissolution-crystallization of nanocrystal superlattices upon thermal cycling. i, One-dimensional SAXS data showing the dissolution of the superlattices during the heating process. j, One-dimensional SAXS data showing the recrystallization of the Pd nanocrystals into superlattices during the cooling process. k, Plot of the fraction of unassembled nanocrystals as a function of temperature during heating (red squares) and cooling (blue dots) cycles.
Extended Data Figure 9 Characterization of Fe nanoparticles and their superlattices formed during synthesis at 180 °C.
a, Representative background-subtracted one-dimensional SAXS patterns (coloured plots) of the Fe nanoparticles in solution during the synthesis before the formation of superlattices and their corresponding SAXS fits (black lines). b, Representative background-subtracted one-dimensional SAXS patterns of Fe nanoparticles in solution after the formation of f.c.c. superlattices. The diffraction peaks shift to smaller q, confirming postgrowth of Fe nanoparticles inside the superlattices. c, A typical structure factor of the superlattices at 880 s. The structure factor corresponds well with a f.c.c. lattice. d, Kinetics of the growth of Fe nanoparticles and their crystallization into f.c.c. superlattices over reaction time. Error bars result from the fits to the SAXS data. e, TEM image of a representative f.c.c. superlattices of Fe nanoparticles. The inset shows the final dispersed Fe nanoparticles. f, SEM image of a representative f.c.c. superlattice. g, Interaction potential between two Fe nanoparticles as a function of interparticle separation. Here T = 453 K is the reaction temperature. The solid plots are the sum of van der Waals attraction and steric repulsive, while the dotted plot is the sum of van der Waals attraction, steric repulsion and magnetic dipole–dipole interaction. Here d represents the diameter of the Fe nanocrystals.
Extended Data Figure 10 Characterization of ex situ synthesized PbTe nanocrystals and their superlattices.
a, SEM image of a representative superlattice of cubic PbTe nanocrystals formed in 1-octadecene at 160 °C. Inset shows a photograph of the superlattice precipitates in 1-octadecene after the synthesis without any purification. b, TEM image of a representative superlattice loaded directly on a Cu TEM grid. c, TEM image of the PbTe nanocrystals from their hexane dispersion after purification. The inset shows a photograph of PbTe nanocrystals dispersed in hexane. d, Size histogram of PbTe nanocrystals obtained from c, showing the PbTe nanocrystal size to be 14.8 ± 2.1 nm.
Rights and permissions
About this article
Cite this article
Wu, L., Willis, J., McKay, I. et al. High-temperature crystallization of nanocrystals into three-dimensional superlattices. Nature 548, 197–201 (2017). https://doi.org/10.1038/nature23308
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature23308
This article is cited by
-
Real-time monitoring of CdTe quantum dots growth in aqueous solution
Scientific Reports (2024)
-
Crystallization of binary nanocrystal superlattices and the relevance of short-range attraction
Nature Synthesis (2023)
-
Nanocluster superstructures assembled via surface ligand switching at high temperature
Nature Synthesis (2023)
-
Surface-ligand-induced crystallographic disorder–order transition in oriented attachment for the tuneable assembly of mesocrystals
Nature Communications (2022)
-
Observation of ordered organic capping ligands on semiconducting quantum dots via powder X-ray diffraction
Nature Communications (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.