Abstract
Retracing the evolutionary history of arthropods has been one of the greatest challenges in biology1. During the past decade, phylogenetic analyses of morphological and molecular data2,3,4 have coalesced towards the conclusion that Mandibulata, the most diverse and abundant group of animals, is a distinct clade from Chelicerata, in that its members possess post-oral head appendages specialized for food processing, notably the mandible2,5. The origin of the mandibulate body plan, however, which encompasses myriapods, crustaceans and hexapods, has remained poorly documented1,6. Here we show that Tokummia katalepsis gen. et sp. nov., a large bivalved arthropod from the 508 million-year-old Marble Canyon fossil deposit (Burgess Shale, British Columbia), has unequivocal mandibulate synapomorphies, including mandibles and maxillipeds, as well as characters typically found in crustaceans, such as enditic, subdivided basipods and ring-shaped trunk segments. Tokummia and its closest relative, Branchiocaris (in Protocarididae, emended), also have an anteriormost structure housing a probable bilobed organ, which could support the appendicular origin of the labrum7. Protocaridids are retrieved with Canadaspis and Odaraia (in Hymenocarina, emended) as part of an expanded mandibulate clade, refuting the idea that these problematic bivalved taxa, as well as other related forms, are representatives of the basalmost euarthropods8,9. Hymenocarines now illustrate that the subdivision of the basipod and the presence of proximal endites are likely to have been ancestral conditions critical for the evolution of coxal and pre-coxal features in mandibulates10,11. The presence of crustaceomorph traits in the Cambrian larvae of various clades basal to Mandibulata is reinterpreted as evidence for the existence of distinct ontogenetic niches among stem arthropods. Larvae would therefore have constituted an important source of morphological novelty during the Cambrian period, and, through heterochronic processes, may have contributed to the rapid acquisition of crown-group characters and thus to greater evolutionary rates during the early radiation of euarthropods12.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Edgecombe, G. D. & Legg, D. A. Origins and early evolution of arthropods. Palaeontology 57, 457–468 (2014)
Edgecombe, G. D., Wilson, G. D. F., Colgan, D. J., Gray, M. R. & Cassis, G. Arthropod cladistics: combined analysis of histone H3 and U2 snRNA sequences and morphology. Cladistics 16, 155–203 (2000)
Regier, J. C. et al. Arthropod relationships revealed by phylogenomic analysis of nuclear protein-coding sequences. Nature 463, 1079–1083 (2010)
Rota-Stabelli, O . et al. A congruent solution to arthropod phylogeny: phylogenomics, microRNAs and morphology support monophyletic Mandibulata. Proc. R. Soc. B 278, 298–306 (2011)
Scholtz, G., Mittmann, B. & Gerberding, M. The pattern of Distal-less expression in the mouthparts of crustaceans, myriapods and insects: new evidence for a gnathobasic mandible and the common origin of Mandibulata. Int. J. Dev. Biol. 42, 801–810 (1998)
Vaccari, N. E., Edgecombe, G. D. & Escudero, C. Cambrian origins and affinities of an enigmatic fossil group of arthropods. Nature 430, 554–557 (2004)
Haas, M. S., Brown, S. J. & Beeman, R. W. Homeotic evidence for the appendicular origin of the labrum in Tribolium castaneum. Dev. Genes Evol. 211, 96–102 (2001)
Budd, G. E. A palaeontological solution to the arthropod head problem. Nature 417, 271–275 (2002)
Legg, D. A ., Sutton, M. D ., Edgecombe, G. D . & Caron, J. B. Cambrian bivalved arthropod reveals origin of arthrodization. Proc. R. Soc. B 279, 4699–4704 (2012)
Walossek, D. & Müller, K. J. in Arthropod Relationships (eds Fortey, R. R. & Thomas, R. ) 139–153 (Chapman & Hall, 1998)
Boxshall, G. A. The evolution of arthropod limbs. Biol. Rev. Camb. Philos. Soc. 79, 253–300 (2004)
Lee, M. S. Y., Soubrier, J. & Edgecombe, G. D. Rates of phenotypic and genomic evolution during the Cambrian explosion. Curr. Biol. 23, 1889–1895 (2013)
Walcott. C. D. Middle Cambrian Branchiopoda, Malacostraca, Trilobita and Merostomata. Smithson. Misc. Collect. 57, 145–228 (1912)
Resser, C. E. New Lower and Middle Cambrian Crustacea. Proc. US Natl Mus. 76, 1–18 (1929)
Briggs, D. E. G. The Arthropod Branchiocaris n. gen., Middle Cambrian, Burgess Shale, British Columbia. Geol. Surv. Can. Bull. 264, 1–29 (1976)
Briggs, D. E. G. The morphology, mode of life, and affinities of Canadaspis perfecta (Crustacea: Phyllocarida), Middle Cambrian, Burgess Shale, British Columbia. Phil. Trans. R. Soc. Lond. B 281, 439–487 (1978)
Briggs, D. E. G. The arthropod Odaraia alata Walcott, Middle Cambrian, Burgess Shale, British Columbia. Phil. Trans. R. Soc. Lond. B 291, 541–582 (1981)
Ortega-Hernández, J. Homology of head sclerites in Burgess Shale euarthropods. Curr. Biol. 25, 1625–1631 (2015)
Ortega-Hernández, J. Making sense of ‘lower’ and ‘upper’ stem-group Euarthropoda, with comments on the strict use of the name Arthropoda von Siebold, 1848. Biol. Rev. Camb. Philos. Soc. 91, 255–273 (2016)
Legg, D. A. An acercostracan marrellomorph (Euarthropoda) from the Lower Ordovician of Morocco. Sci. Nat. 103, 21 (2016)
Caron, J.-B., Gaines, R. R., Aria, C., Mángano, M. G. & Streng, M. A new phyllopod bed-like assemblage from the Burgess Shale of the Canadian Rockies. Nat. Commun. 5, 3210 (2014)
Dunlop, J. A. The epistomo-labral plate and lateral lips in solifuges, pseudoscorpions and mites. Ekologia (Bratisl.) 19, 67–78 (2000)
Ungerer, P. & Wolff, C. External morphology of limb development in the amphipod Orchestia cavimana (Crustacea, Malacostraca, Peracarida). Zoomorphology 124, 89–99 (2005)
Liu, Y., Maas, A. & Waloszek, D. Early development of the anterior body region of the grey widow spider Latrodectus geometricus Koch, 1841 (Theridiidae, Araneae). Arthropod Struct. Dev. 38, 401–416 (2009)
Hughes, C. L. & Kaufman, T. C. Hox genes and the evolution of the arthropod body plan. Evol. Dev. 4, 459–499 (2002)
Aria, C., Caron, J.-B. & Gaines, R. A large new leanchoiliid from the Burgess Shale and the influence of inapplicable states on stem arthropod phylogeny. Palaeontology 58, 629–660 (2015)
Yang, J., Ortega-Hernández, J., Butterfield, N. J. & Zhang, X. G. Specialized appendages in fuxianhuiids and the head organization of early euarthropods. Nature 494, 468–471 (2013)
Strausfeld, N. J. et al. Arthropod eyes: the early Cambrian fossil record and divergent evolution of visual systems. Arthropod Struct. Dev. 45, 152–172 (2016)
Aria, C. & Caron, J.-B. Cephalic and limb anatomy of a new isoxyid from the Burgess Shale and the role of “stem bivalved arthropods” in the disparity of the frontalmost appendage. PLoS One 10, e0124979 (2015)
Nakazawa, T. Ontogenetic niche shifts matter in community ecology: a review and future perspectives. Popul. Ecol. 57, 347–354 (2015)
Swofford, D. L. PAUP*. Phylogenetic Analysis using Parsimony (*and Other Methods) Version 4.0a147 (Sinauer Associates, 2002)
Acknowledgements
We thank D. Erwin and B. Lieberman for access to collections at the Smithsonian Institution and at the University of Kansas Natural History Museum, respectively, M. S. Lee for help with the phylogenetic reconstruction, S. Scharf for editorial comments, D. Dufault for the technical drawings of Tokummia and L. Fields for the animations. We thank T. Keith from Parks Canada for providing field and logistical assistance. C.A.’s research was supported by fellowships from the University of Toronto (Department of Ecology and Evolutionary Biology) and J.-B.C.’s Natural Sciences and Engineering Research Council Discovery Grant (341944). This is Royal Ontario Museum Burgess Shale project number 69.
Author information
Authors and Affiliations
Contributions
C.A. wrote the initial drafts of the manuscript, Supplementary Information (including the taxonomic description) and character list, compiled morphological data and ran the phylogenetic analyses. J.-B.C. organized the expeditions at Marble Canyon, and prepared and photographed the specimens. Both authors contributed to the collection, observation and interpretation of fossils, as well as the conception and realisation of figures and reconstructions, and the writing of the final version of this manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer Information Nature thanks J. Vannier and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
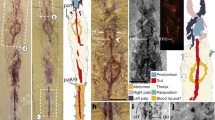
Extended Data Figure 1 Tokummia katalepsis, holotype ROM 63823.
a, b, Part, full specimen before (a) and after (b) preparation. Boxes in b are magnified in c/d and e. c, Close-up of frontal area showing left maxilliped curved under the body and the following two pairs of thoracic limbs, asterisks mark the remains of exopods. d, Magnified inset of b as in c, low-angle lighting. e, Close-up of posterior thoracic region, showing stout endopods and terminal claws. The specimen was photographed dry using direct light (d) or cross-polarized light (a, b, c, e). an, antennule; pn, podomere of maxilliped n; tn, thoracic appendage n. Scale bars, 10 mm (a) and 2 mm (c, e).
Extended Data Figure 2 Tokummia katalepsis, paratype ROM 63824.
a, Part, full specimen after preparation. Large and small boxes indicate regions magnified in b and c, respectively. b, Close-up of anterior region (large inset in a), the arrow indicates the start of post-thoracic tagma with broad exopods. c, Close-up of the fourth and fifth thoracic appendages (small inset in a), arrows point to the paired spines of the ante-penultimate podomeres. The specimen was photographed dry using cross-polarized light. an, antennule; mxp, maxilliped; pn, endopod podomere n; tn, thoracic appendage n. Scale bars, 10 mm (a), 5 mm (b) and 2 mm (c).
Extended Data Figure 3 Tokummia katalepsis, paratype ROM 63825.
a, Part, full specimen (before preparation; see Fig. 1e for prepared specimen). Top left and bottom right boxed regions are magnified in b and c, respectively. b, Close-up of the right maxilliped chela (small inset in a), the arrow indicates lateral carina corresponding to the margin of the sharp edge of dactyl. c, Close-up of the posterior region (large inset in a), showing tergites and sternites, as well as bases and exopods of appendages. The bar marks a region of stacked exopods, arrows indicate preservation of imbricated tergites. Scale bars, 5 mm (a) and 2 mm (b, c).
Extended Data Figure 4 Tokummia katalepsis, paratype ROM 63014.
a, b, Part, full specimen, before (a) and after (b) preparation. c, Close-up of maxillipeds and anteriormost region. an, antennule; dyl, dactyl; mus, manus; pex, pollex; pn, podomere n (maxilliped). Scale bars, 10 mm (a), 5 mm (b) and 2 mm (c).
Extended Data Figure 5 Tokummia katalepsis.
a, Paratype ROM 63736 (part), molt (see close-ups in Fig. 2d, f, g). b, Paratype ROM 63827, close-up of anterior region, showing tergite of ocular somite, antennules and first post-maxilliped thoracic appendages. c–e, Paratype ROM 63081, molts of maxilliped chelae. c, Pair of chelae, boxed regions are magnified in d, e. d, e, Insets in c, magnification of the articles of the chelae (pollex and dactyl). Arrows in d indicate teeth on the dactyl. f, g, Paratype ROM 63826. f, Close-up of maxillipeds and front showing anterior carapacal processes and putative eye. Boxed region is magnified in g. g, Close-up of left maxilliped chela. Arrows indicate teeth on the dactyl, the asterisk marks breakage at the base of the pollex, showing that the dactyl was likely articulating with the pollex at its base. All specimens were photographed dry using cross-polarized light, except ROM 63827, which was photographed using low-angle lighting after coating in ammonium chloride. acp, anterior carapacal processes; dyl, dactyl; e?, putative eye; lan, left antennule; pex, pollex; ran, right antennule; tn, thoracic appendage n; tos, tergite of ocular somite. Scale bars, 10 mm (a), 5 mm (b, c, f) and 2 mm (d, e, g).
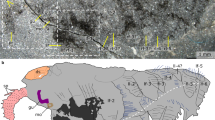
Extended Data Figure 6 Branchiocaris pretiosa Resser, ROM 60751.
a, Part, full specimen, arrow points to margin of posterior tergal plate. Boxed areas on the left and right are magnified in b and c, respectively. b, Close-up of the anterior region (left inset in a). c, Close-up of the tailpiece (right inset in a). Asterisk indicates the location of the anus. d, Close-up of labral complex (part). e, Interpretative drawing of d, solid lines are contours of body features, dashed lines are cracks or preparation marks within sediment or body features, and striated (grey) lines are likely contours of body features. f, Close up of labral complex (counterpart). g, Interpretative drawing of f, lines as in e. Specimens were photographed wet using cross-polarized light (a, b, d, f) or direct light (c). bd, body; db, doublure; lan, left antennule; lman, left mandible; lp, labral protrusion; lcv, left carapacal valve; ran, right antennule; rman, right mandible; rcv, right carapacal valve; tos, tergite of ocular somite. Scale bars, 10 mm (a–c) and 2.5 mm (d, f).
Extended Data Figure 7 Branchiocaris pretiosa Resser.
a–d, USNM 189030. a, Counterpart, dorsal view. Boxes indicate regions shown in b and c. b, Interpretative drawing of a (large inset), showing the relative position of labral complex, antennules and mandibles. Asterisks mark the contiguity between attachment of antennules and anterior margin of mandibles, where the post-antennular segment would be expected (hence thought to be the intercalary segment of early ontogenetic stages). Plain lines are contours of body features, dashed lines are cracks or preparation marks within sediment or body features, and striated (grey) lines are likely contours of body features. Note that in this specimen, the tergite of the ocular somite (preserved here in positive) bears a median carina. c, Close-up of the labral complex in a (small inset). Arrows indicate the inner margins of sclerite, arrowheads indicate the inner margins of the lobes of protrusion and asterisks mark posterior parts of the left and right lobes. d, Part of a. e, USNM 80483, close-up of mid-section of body, showing sternites, tergites and appendages with broad exopods, asterisks mark individual sternites, arrows indicate proximal lobe of exopods. All specimens were photographed using cross-polarized light in dry (a, d, e) or wet (c) conditions. it, intestinal tract; lan, left antennule; lbc, labral complex; lman, left mandible; lp, labral protrusion; ran, right antennule; rman, right mandible; rmxp, right maxilliped; tos, tergite of ocular somite. Scale bars, 2 mm (a, d, e) and 1 mm (c).
Extended Data Figure 8 Branchiocaris cf. pretiosa Resser.
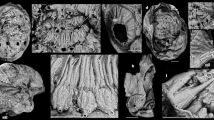
a–d, KUMIP 204795. e, f, KUMIP 204797. g–i, KUMIP 204790. a, Part, full specimen. Boxed region is magnified in c. b, Counterpart, low-angle lighting. c, Close-up of anteriormost region, arrows indicate the margins of the rounded ocular tergite. Note the underlying material, which is probably equivalent to the labral protrusion described in this paper. d, Anterior area on counterpart. e, Part, full specimen with large and distinctly preserved mandibles. Boxed region is magnified in f. f, Close-up of anterior axial region, showing mandibles, maxillule, maxilla and thoracic limbs. g, Part, full specimen, breakage of carapace reveals mandibles. Boxed region is magnified in h. h, close-up of mandibles. i, Close-up of gnathal edge of left mandible (inset in h) showing a multitude of fine striations. All specimens were photographed using cross-polarized light in dry (a) or wet (b–d) conditions. db, doublure (of ocular tergite); it, intestinal tract; lman, left mandible; lma, left maxilla; lmu, left maxillule; lp, labral protrusion; m, mouth; pm, pars molaris; rman, right mandible; tos, tergite of ocular somite. Scale bars, 10 mm (a, e), 1 mm (b, c), 5 mm (d, f–h) and 0.5 mm (i).
Extended Data Figure 9 Canadaspis perfecta Walcott and Odaraia alata Walcott.
a–f, Canadaspis perfecta Walcott USNM 207290 (a–d) and USNM 213860 (e, f). a, Part, full specimen, boxed area is magnified in b–d. b, Close-up of the frontalmost region, showing eyes, inter-ocular lobes and rostral ocular tergite. Asterisks mark inter-ocular lobes. c, d, Same region as b. c, Wet specimen, plain lighting. d, Wet specimen, cross-polarized lighting. e, Part, full specimen, photographed using cross-polarized light in dry conditions, boxed region is shown in f. f, Close-up of the anteriormost region, showing eyes, antennules, inter-ocular lobes and rounded ocular tergite. Asterisks mark inter-ocular lobes. g, h, Odaraia alata Walcott, ROM 63838. Possible molt, preserved in ventral aspect. g, Part, full specimen with inset magnified in h. h, Close-up of limbs, showing seven-segmented endopods, basipods and thin, ‘vertebra-like’ sternites. ba, basipod; ex, endopod podomere number x; en, endopod; st, sternite, tos, tergite of ocular somite. Scale bars, 10 mm (g), 5 mm (a, e), 2 mm (h) and 1 mm (b–d, f).
Extended Data Figure 10 Detailed phylogenetic results.
a, b, Majority-rule consensus cladogram (cut at 50%) of the 500 most parsimonious trees of length 888 obtained by tree bisection and reconnection and under backbone constraint based on molecular results from a dataset of 90 taxa and 212 characters. a, The bottom diagram represents the main external antero-posterior metameric features of corresponding clades on the left of the tree; among terminals. Body plans shown in blue are larval. T. katalepsis is shown in green and Hymenocarina in bold purple. Lorraine crosses mark extinct taxa. c–d, Complementary topologies using the same dataset as in Fig. 4 and a, b (tree bisection and reconnection with 1,000 replicates). c, Strict consensus tree of a, b. d, Majority-rule consensus of 840 most parsimonious trees of length 763, excluding larval taxa. e, Majority-rule consensus of 170 most parsimonious trees of length 849, without backbone constraint. In b, d, e, numbers above branches are frequencies of occurrence. Characters are unweighted and unordered.
Supplementary information
Supplementary Information
This file contains a Supplementary Discussion, Character list and Supplementary references. (PDF 775 kb)
Supplementary Data
This file contains the morphological matrix in Excel format. (XLS 284 kb)
Supplementary Data
This file contains PAUP* data and command lines (Nexus file). (TXT 39 kb)
Three-dimensional reconstruction of Tokummia katalepsis.
Rotating animation by Lars Fields. (AVI 16367 kb)
Three-dimensional reconstruction of Tokummia katalepsis.
Walk cycle animation by Lars Fields. (AVI 7520 kb)
Rights and permissions
About this article
Cite this article
Aria, C., Caron, JB. Burgess Shale fossils illustrate the origin of the mandibulate body plan. Nature 545, 89–92 (2017). https://doi.org/10.1038/nature22080
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature22080
This article is cited by
-
A network analysis of early arthropod evolution and the potential of the primitive
Scientific Reports (2024)
-
New fossil assemblages from the Early Ordovician Fezouata Biota
Scientific Reports (2022)
-
Functional importance of the mandibular skeleto-muscular system in the bivalved arthropod Heterocypris incongruens (Crustacea, Ostracoda, Cyprididae)
The Science of Nature (2022)
-
Fossils from South China redefine the ancestral euarthropod body plan
BMC Evolutionary Biology (2020)
-
An early Cambrian euarthropod with radiodont-like raptorial appendages
Nature (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.