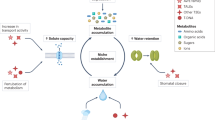
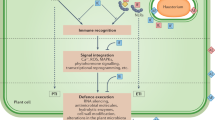
Abstract
Plants encounter a myriad of microorganisms, particularly at the root–soil interface, that can invade with detrimental or beneficial outcomes. Prevalent beneficial associations between plants and microorganisms include those that promote plant growth by facilitating the acquisition of limiting nutrients such as nitrogen and phosphorus. But while promoting such symbiotic relationships, plants must restrict the formation of pathogenic associations. Achieving this balance requires the perception of potential invading microorganisms through the signals that they produce, followed by the activation of either symbiotic responses that promote microbial colonization or immune responses that limit it.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Müller, D. B., Vogel, C., Bai, Y. & Vorholt, J. A. The plant microbiota: systems-level insights and perspectives. Annu. Rev. Genet. 50, 211–234 (2016).
Cook, D. E., Mesarich, C. H. & Thomma, B. P. Understanding plant immunity as a surveillance system to detect invasion. Annu. Rev. Phytopathol. 53, 541–563 (2015).
Boller, T. & Felix, G. A renaissance of elicitors: perception of microbe-associated molecular patterns and danger signals by pattern-recognition receptors. Annu. Rev. Plant Biol. 60, 379–406 (2009).
Niehl, A., Wyrsch, I., Boller, T. & Heinlein, M. Double-stranded RNAs induce a pattern-triggered immune signaling pathway in plants. New Phytol. 211, 1008–1019 (2016).
Manosalva, P. et al. Conserved nematode signalling molecules elicit plant defenses and pathogen resistance. Nature Commun. 6, 7795 (2015).
Prince, D. C., Drurey, C., Zipfel, C. & Hogenhout, S. A. The leucine-rich repeat receptor-like kinase BRASSINOSTEROID INSENSITIVE 1-ASSOCIATED KINASE 1 and the cytochrome P450 PHYTOALEXIN DEFICIENT 3 contribute to innate immunity to aphids in Arabidopsis . Plant Physiol. 164, 2207–2219 (2014).
Hegenauer, V. et al. Detection of the plant parasite Cuscuta reflexa by a tomato cell surface receptor. Science 353, 478–481 (2016). This paper shows that the perception of parasitic plants utilizes PAMP recognition in a way that is similar to the perception of microbes.
Oldroyd, G. E. Speak, friend, and enter: signalling systems that promote beneficial symbiotic associations in plants. Nature Rev. Microbiol. 11, 252–263 (2013).
Limpens, E., van Zeijl, A. & Geurts, R. Lipochitooligosaccharides modulate plant host immunity to enable endosymbioses. Annu. Rev. Phytopathol. 53, 311–334 (2015). A comprehensive review on the perception of symbiotic chitin oligosaccharides and LCOs by legumes.
Couto, D. & Zipfel, C. Regulation of pattern recognition receptor signalling in plants. Nature Rev. Immunol. 16, 537–552 (2016). A comprehensive review on the mechanisms of plant immune perception and signalling.
Fliegmann, J. & Felix, G. Immunity: flagellin seen from all sides. Nature Plants 2, 16136 (2016).
Furukawa, T., Inagaki, H., Takai, R., Hirai, H. & Che, F. S. Two distinct EF-Tu epitopes induce immune responses in rice and Arabidopsis . Mol. Plant Microbe Interact. 27, 113–124 (2014).
Lopez-Gomez, M., Sandal, N., Stougaard, J. & Boller, T. Interplay of flg22-induced defence responses and nodulation in Lotus japonicus . J. Exp. Bot. 63, 393–401 (2012).
Bressendorff, S. et al. An innate immunity pathway in the moss Physcomitrella patens . Plant Cell 28, 1328–1342 (2016). This study reveals that the perception of chitin is evolutionarily conserved in mosses.
Dénarié, J., Debellé, F. & Promé, J.-C. Rhizobium lipo-chitooligosaccharide Nodulation factors: signaling molecules mediating recognition and morphogenesis. Annu. Rev. Biochem. 65, 503–535 (1996).
Lerouge, P. et al. Symbiotic host-specificity of Rhizobium meliloti is determined by a sulphated and acylated glucosamine oligosaccharide signal. Nature 344, 781–784 (1990).
Maillet, F. et al. Fungal lipochitooligosaccharide symbiotic signals in arbuscular mycorrhiza. Nature 469, 58–63 (2011).
Genre, A. et al. Short-chain chitin oligomers from arbuscular mycorrhizal fungi trigger nuclear Ca2+ spiking in Medicago truncatula roots and their production is enhanced by strigolactone. New Phytol. 198, 190–202 (2013).
Oldroyd, G., Mitra, R. M., Wais, R. & Long, S. Evidence for structurally specific negative feedback in the Nod factor signal transduction pathway. Plant J. 28, 191–199 (2001).
Sun, J. et al. Activation of symbiosis signaling by arbuscular mycorrhizal fungi in legumes and rice. Plant Cell 27, 823–838 (2015).
Walker, S. A., Viprey, V. & Downie, J. A. Dissection of nodulation signaling using pea mutants defective for calcium spiking induced by Nod factors and chitin oligomers. Proc. Natl Acad. Sci. USA 97, 13413–13418 (2000).
Gust, A. A., Willmann, R., Desaki, Y., Grabherr, H. M. & Nurnberger, T. Plant LysM proteins: modules mediating symbiosis and immunity. Trends Plant Sci. 17, 495–502 (2012).
Shinya, T., Nakagawa, T., Kaku, H. & Shibuya, N. Chitin-mediated plant-fungal interactions: catching, hiding and handshaking. Curr. Opin. Plant Biol. 26, 64–71 (2015).
Shinya, T. et al. Functional characterization of CEBiP and CERK1 homologs in Arabidopsis and rice reveals the presence of different chitin receptor systems in plants. Plant Cell Physiol. 53, 1696–1706 (2012).
Faulkner, C. et al. LYM2-dependent chitin perception limits molecular flux via plasmodesmata. Proc. Natl Acad. Sci. USA 110, 9166–9170 (2013).
Narusaka, Y. et al. Presence of LYM2 dependent but CERK1 independent disease resistance in Arabidopsis . Plant Signal. Behav. 8, (2013).
Willmann, R. et al. Arabidopsis lysin-motif proteins LYM1 LYM3 CERK1 mediate bacterial peptidoglycan sensing and immunity to bacterial infection. Proc. Natl Acad. Sci. USA 108, 19824–19829 (2011).
Liu, T. et al. Chitin-induced dimerization activates a plant immune receptor. Science 336, 1160–1164 (2012).
Kouzai, Y. et al. Targeted gene disruption of OsCERK1 reveals its indispensable role in chitin perception and involvement in the peptidoglycan response and immunity in rice. Mol. Plant Microbe Interact. 27, 975–982 (2014).
Ao, Y. et al. OsCERK1 and OsRLCK176 play important roles in peptidoglycan and chitin signaling in rice innate immunity. Plant J. 80, 1072–1084 (2014).
Czaja, L. F. et al. Transcriptional responses toward diffusible signals from symbiotic microbes reveal MtNFP- and MtDMI3-dependent reprogramming of host gene expression by arbuscular mycorrhizal fungal lipochitooligosaccharides. Plant Physiol. 159, 1671–1685 (2012).
Amor, B. B. et al. The NFP locus of Medicago truncatula controls an early step of Nod factor signal transduction upstream of a rapid calclium flux and root hair deformation. Plant J. 34, 495–506 (2003).
Radutoiu, S. et al. Plant recognition of symbiotic bacteria requires two LysM receptor-like kinases. Nature 425, 585–592 (2003).
Zhang, X. et al. The receptor kinase CERK1 has dual functions in symbiosis and immunity signalling. Plant J. 81, 258–267 (2015).
Op den Camp, R. et al. LysM-type mycorrhizal receptor recruited for Rhizobium symbiosis in nonlegume Parasponia . Science 331, 909–912 (2011).
Buendia, L., Wang, T., Girardin, A. & Lefebvre, B. The LysM receptor-like kinase SlLYK10 regulates the arbuscular mycorrhizal symbiosis in tomato. New Phytol. 210, 184–195 (2016). This paper demonstrates that LysM-receptor kinases are required for the association of plants with arbuscular mycorrhizal fungi.
Miyata, K. et al. Evaluation of the role of the LysM receptor-like kinase, OsNFR5/OsRLK2 for AM symbiosis in rice. Plant Cell Physiol. 57, 2283–2290 (2016).
Gaude, N., Bortfeld, S., Duensing, N., Lohse, M. & Krajinski, F. Arbuscule-containing and non-colonized cortical cells of mycorrhizal roots undergo extensive and specific reprogramming during arbuscular mycorrhizal development. Plant J. 69, 510–528 (2012).
Gomez, S. K. & Harrison, M. J. Laser microdissection and its application to analyze gene expression in arbuscular mycorrhizal symbiosis. Pest Manag. Sci. 65, 504–511 (2009).
Hogekamp, C. et al. Laser microdissection unravels cell-type-specific transcription in arbuscular mycorrhizal roots, including CAAT-box transcription factor gene expression correlating with fungal contact and spread. Plant Physiol. 157, 2023–2043 (2011).
Fliegmann, J. et al. Lipo-chitooligosaccharidic symbiotic signals are recognized by LysM receptor-like kinase LYR3 in the legume Medicago truncatula . ACS Chem. Biol. 8, 1900–1906 (2013).
Fliegmann, J. et al. LYR3, a high-affinity LCO-binding protein of Medicago truncatula, interacts with LYK3, a key symbiotic receptor. FEBS Lett. 590, 1477–1487 (2016).
Malkov, N. et al. Molecular basis of lipo-chitooligosaccharide recognition by the lysin motif receptor-like kinase LYR3 in legumes. Biochem. J. 473, 1369–1378 (2016).
De Mita, S., Streng, A., Bisseling, T. & Geurts, R. Evolution of a symbiotic receptor through gene duplications in the legume–Rhizobium mutualism. New Phytol. 201, 961–972 (2014).
Miyata, K. et al. The bifunctional plant receptor, OsCERK1, regulates both chitin-triggered immunity and arbuscular mycorrhizal symbiosis in rice. Plant Cell Physiol. 55, 1864–1872 (2014). One of several papers that show that CERK1 in rice has a dual role in immunity and symbiosis signalling.
Nakagawa, T. et al. From defense to symbiosis: limited alterations in the kinase domain of LysM receptor-like kinases are crucial for evolution of legume–Rhizobium symbiosis. Plant J. 65, 169–180 (2011).
Broghammer, A. et al. Legume receptors perceive the rhizobial lipochitin oligosaccharide signal molecules by direct binding. Proc. Natl Acad. Sci. USA 109, 13859–13864 (2012).
Madsen, E. B. et al. Autophosphorylation is essential for the in vivo function of the Lotus japonicus Nod factor receptor 1 and receptor-mediated signalling in cooperation with Nod factor receptor 5. Plant J. 65, 404–417 (2011).
Pietraszewska-Bogiel, A. et al. Interaction of Medicago truncatula lysin motif receptor-like kinases, NFP and LYK3, produced in Nicotiana benthamiana induces defence-like responses. PLoS ONE 8, e65055 (2013).
Ben, C. et al. Natural diversity in the model legume Medicago truncatula allows identifying distinct genetic mechanisms conferring partial resistance to Verticillium wilt. J. Exp. Bot. 64, 317–332 (2013).
Rey, T. et al. NFP, a LysM protein controlling Nod factor perception, also intervenes in Medicago truncatula resistance to pathogens. New Phytol. 198, 875–886 (2013).
Rey, T., Chatterjee, A., Buttay, M., Toulotte, J. & Schornack, S. Medicago truncatula symbiosis mutants affected in the interaction with a biotrophic root pathogen. New Phytol. 206, 497–500 (2015).
Wang, E. et al. A common signaling process that promotes mycorrhizal and oomycete colonization of plants. Curr. Biol. 22, 2242–2246 (2012).
Downie, J. A. The roles of extracellular proteins, polysaccharides and signals in the interactions of rhizobia with legume roots. FEMS Microbiol. Rev. 34, 150–170 (2010).
Gully, D. et al. A peptidoglycan-remodeling enzyme is critical for bacteroid differentiation in Bradyrhizobium spp. during legume symbiosis. Mol. Plant Microbe Interact. 29, 447–457 (2016).
Kawaharada, Y. et al. Receptor-mediated exopolysaccharide perception controls bacterial infection. Nature 523, 308–312 (2015). The first paper to describe the receptors in plants that are responsible for the recognition of exopolysaccharides from bacteria.
Cardenas, L. et al. Rhizobium Nod factors induce increases in intracellular free calcium and extracellular calcium influxes in bean root hairs. Plant J. 19, 347–352 (1999).
Shaw, S. L. & Long, S. R. Nod factor elicits two separable calcium responses in Medicago truncatula root hair cells. Plant Physiol. 131, 976–984 (2003).
Ma, X., Xu, G., He, P. & Shan, L. SERKing coreceptors for receptors. Trends Plant Sci. 21, 1017–1033 (2016).
Halter, T. et al. The leucine-rich repeat receptor kinase BIR2 is a negative regulator of BAK1 in plant immunity. Curr. Biol. 24, 134–143 (2014).
Yeh, Y. H. et al. The Arabidopsis malectin-like/LRR-RLK IOS1 is critical for BAK1-dependent and BAK1-independent pattern-triggered immunity. Plant Cell 28, 1701–1721 (2016).
Le, M. H., Cao, Y., Zhang, X. C. & Stacey, G. LIK1, a CERK1-interacting kinase, regulates plant immune responses in Arabidopsis . PLoS ONE 9, e102245 (2014).
Endre, G. et al. A receptor kinase gene regulating symbiotic nodule development. Nature 417, 962–966 (2002).
Stracke, S. et al. A plant receptor-like kinase required for both bacterial and fungal symbiosis. Nature 417, 959–962 (2002).
Antolín-Llovera, M., Ried, M. K. & Parniske, M. Cleavage of the SYMBIOSIS RECEPTOR-LIKE KINASE ectodomain promotes complex formation with Nod factor receptor 5. Curr. Biol. 24, 422–427 (2014).
Ried, M. K., Antolin-Llovera, M. & Parniske, M. Spontaneous symbiotic reprogramming of plant roots triggered by receptor-like kinases. eLife 3, e03891 (2014).
Chen, T. et al. A MAP kinase kinase interacts with SymRK and regulates nodule organogenesis in Lotus japonicus . Plant Cell 24, 823–838 (2012).
Venkateshwaran, M. et al. A role for the mevalonate pathway in early plant symbiotic signaling. Proc. Natl Acad. Sci. USA 112, 9781–9786 (2015).
Akula, M. K. et al. Control of the innate immune response by the mevalonate pathway. Nature Immunol. 17, 922–929 (2016).
Zhang, J. et al. Receptor-like cytoplasmic kinases integrate signaling from multiple plant immune receptors and are targeted by a Pseudomonas syringae effector. Cell Host Microbe 7, 290–301 (2010).
Lu, D. et al. A receptor-like cytoplasmic kinase, BIK1, associates with a flagellin receptor complex to initiate plant innate immunity. Proc. Natl Acad. Sci. USA 107, 496–501 (2010).
Shinya, T. et al. Selective regulation of the chitin-induced defense response by the Arabidopsis receptor-like cytoplasmic kinase PBL27. Plant J. 79, 56–66 (2014).
Yamaguchi, K. et al. A receptor-like cytoplasmic kinase targeted by a plant pathogen effector is directly phosphorylated by the chitin receptor and mediates rice immunity. Cell Host Microbe 13, 347–357 (2013).
Sreekanta, S. et al. The receptor-like cytoplasmic kinase PCRK1 contributes to pattern-triggered immunity against Pseudomonas syringae in Arabidopsis thaliana . New Phytol. 207, 78–90 (2015).
Kadota, Y. et al. Direct regulation of the NADPH Oxidase RBOHD by the PRR-associated kinase BIK1 during plant immunity. Mol. Cell 54, 43–55 (2014).
Li, L. et al. The FLS2-associated kinase BIK1 directly phosphorylates the NADPH oxidase RbohD to control plant immunity. Cell Host Microbe 15, 329–338 (2014).
Feng, F. et al. A Xanthomonas uridine 5′-monophosphate transferase inhibits plant immune kinases. Nature 485, 114–118 (2012).
Yamada, K. et al. The Arabidopsis CERK1-associated kinase PBL27 connects chitin perception to MAPK activation. EMBO J. 35, 2468–2483 (2016). A study that links an activated PAMP receptor complex to MAP kinase activation in response to chitin perception.
Andrio, E. et al. Hydrogen peroxide-regulated genes in the Medicago truncatula–Sinorhizobium meliloti symbiosis. New Phytol. 198, 179–189 (2013).
Charpentier, M. et al. Nuclear-localized cyclic nucleotide-gated channels mediate symbiotic calcium oscillations. Science 352, 1102–1105 (2016).
Charpentier, M. et al. Lotus japonicus CASTOR and POLLUX are ion channels essential for perinuclear calcium spiking in legume root endosymbiosis. Plant Cell 20, 3467–3479 (2008).
Capoen, W. et al. Nuclear membranes control symbiotic calcium signaling of legumes. Proc. Natl Acad. Sci. USA 108, 14348–14353 (2011).
Lévy, J. et al. A putative Ca2+ and calmodulin-dependent protein kinase required for bacterial and fungal symbioses. Science 303, 1361–1364 (2004).
Mitra, R. M. et al. A Ca2+/calmodulin-dependent protein kinase required for symbiotic nodule development: gene identification by transcript-based cloning. Proc. Natl Acad. Sci. USA 101, 4701–4705 (2004).
Miller, J. B. et al. Calcium/calmodulin-dependent protein kinase is negatively and positively regulated by calcium, providing a mechanism for decoding calcium responses during symbiosis signaling. Plant Cell 25, 5053–5066 (2013).
Takeda, N., Maekawa, T. & Hayashi, M. Nuclear-localized and deregulated calcium- and calmodulin-dependent protein kinase activates rhizobial and mycorrhizal responses in Lotus japonicus . Plant Cell 24, 810–822 (2012).
Gleason, C. et al. Nodulation independent of rhizobia induced by a calcium-activated kinase lacking autoinhibition. Nature 441, 1149–1152 (2006).
Tirichine, L. et al. Deregulation of a Ca2+/calmodulin-dependent kinase leads to spontaneous nodule development. Nature 441, 1153–1156 (2006).
Messinese, E. et al. A novel nuclear protein interacts with the symbiotic DMI3 calcium and calmodulin dependent protein kinase of Medicago truncatula . Mol. Plant Microbe Interact. 20, 912–921 (2007).
Yano, K. et al. CYCLOPS, a mediator of symbiotic intracellular accommodation. Proc. Natl Acad. Sci. USA 105, 20540–20545 (2008).
Singh, S., Katzer, K., Lambert, J., Cerri, M. & Parniske, M. CYCLOPS, a DNA-binding transcriptional activator, orchestrates symbiotic root nodule development. Cell Host Microbe 15, 139–152 (2014).
Fonouni-Farde, C. et al. DELLA-mediated gibberellin signalling regulates Nod factor signalling and rhizobial infection. Nature Commun. 7, 12636 (2016).
Jin, Y. et al. DELLA proteins are common components of symbiotic rhizobial and mycorrhizal signalling pathways. Nature Commun. 7, 12433 (2016).
Pimprikar, P. et al. A CCaMK–CYCLOPS–DELLA complex activates transcription of RAM1 to regulate arbuscule branching. Curr. Biol. 26, 987–998 (2016). A report that characterizes the calcium-decoding transcription-factor complex that regulates arbuscular mycorrhizal gene expression.
Floss, D. S., Levy, J. G., Levesque-Tremblay, V., Pumplin, N. & Harrison, M. J. DELLA proteins regulate arbuscule formation in arbuscular mycorrhizal symbiosis. Proc. Natl Acad. Sci. USA 110, E5025–E5034 (2013).
Maekawa, T. et al. Gibberellin controls the nodulation signaling pathway in Lotus japonicus . Plant J. 58, 183–194 (2009).
Navarro, L. et al. DELLAs control plant immune responses by modulating the balance of jasmonic acid and salicylic acid signaling. Curr. Biol. 18, 650–655 (2008).
Cárdenas, L., Martinez, A., Sanchez, F. & Quinto, C. Fast, transient and specific intracellular ROS changes in living root hair cells responding to Nod factors (NFs). Plant J. 56, 802–813 (2008).
Marino, D. et al. A Medicago truncatula NADPH oxidase is involved in symbiotic nodule functioning. New Phytol. 189, 580–592 (2011).
Montiel, J. et al. A Phaseolus vulgaris NADPH oxidase gene is required for root infection by rhizobia. Plant Cell Physiol. 53, 1751–1767 (2012).
Morieri, G. et al. Host-specific Nod-factors associated with Medicago truncatula nodule infection differentially induce calcium influx and calcium spiking in root hairs. New Phytol. 200, 656–662 (2013).
Garcia-Brugger, A. et al. Early signaling events induced by elicitors of plant defenses. Mol. Plant Microbe Interact. 19, 711–724 (2006).
Nars, A. et al. Aphanomyces euteiches cell wall fractions containing novel glucan-chitosaccharides induce defense genes and nuclear calcium oscillations in the plant host Medicago truncatula . PLoS ONE 8, e75039 (2013).
Thor, K. & Peiter, E. Cytosolic calcium signals elicited by the pathogen-associated molecular pattern flg22 in stomatal guard cells are of an oscillatory nature. New Phytol. 204, 873–881 (2014). Refs 104 and 105 use live single-cell imaging to reveal that PAMP perception induces cytosolic oscillatory calcium signals.
Keinath, N. F. et al. Live cell imaging with R-GECO1 sheds light on flg22- and chitin-induced transient [Ca2+]cyt patterns in Arabidopsis . Mol. Plant 8, 1188–1200 (2015).
Seybold, H. et al. Ca2+ signalling in plant immune response: from pattern recognition receptors to Ca2+ decoding mechanisms. New Phytol. 204, 782–790 (2014).
Yu, F., Tian, W. & Luan, S. From receptor-like kinases to calcium spikes: what are the missing links? Mol. Plant 7, 1501–1504 (2014).
Boudsocq, M. et al. Differential innate immune signalling via Ca2+ sensor protein kinases. Nature 464, 418–422 (2010).
Dubiella, U. et al. Calcium-dependent protein kinase/NADPH oxidase activation circuit is required for rapid defense signal propagation. Proc. Natl Acad. Sci. USA 110, 8744–8749 (2013).
Ranf, S., Eschen-Lippold, L., Pecher, P., Lee, J. & Scheel, D. Interplay between calcium signalling and early signalling elements during defence responses to microbe- or damage-associated molecular patterns. Plant J. 68, 100–113 (2011).
Evans, M. J., Choi, W. G., Gilroy, S. & Morris, R. J. A ROS-assisted calcium wave dependent on the AtRBOHD NADPH oxidase and TPC1 cation channel propagates the systemic response to salt stress. Plant Physiol. 171, 1771–1784 (2016).
Gilroy, S. et al. ROS, calcium, and electric signals: key mediators of rapid systemic signaling in plants. Plant Physiol. 171, 1606–1615 (2016).
Yang, S., Tang, F., Gao, M., Krishnan, H. B. & Zhu, H. R gene-controlled host specificity in the legume–rhizobia symbiosis. Proc. Natl Acad. Sci. USA 107, 18735–18740 (2010).
Deakin, W. J. & Broughton, W. J. Symbiotic use of pathogenic strategies: rhizobial protein secretion systems. Nature Rev. Microbiol. 7, 312–320 (2009).
Kloppholz, S., Kuhn, H. & Requena, N. A secreted fungal effector of Glomus intraradices promotes symbiotic biotrophy. Curr. Biol. 21, 1204–1209 (2011).
Marchetti, M. et al. Experimental evolution of a plant pathogen into a legume symbiont. PLoS Biol. 8, e1000280 (2010).
Okazaki, S., Kaneko, T., Sato, S. & Saeki, K. Hijacking of leguminous nodulation signaling by the rhizobial type III secretion system. Proc. Natl Acad. Sci. USA 110, 17131–17136 (2013). This paper presents exciting evidence to show that bacterial effectors can activate nodulation signalling without the need for Nod factors.
Giraud, E. et al. Legume symbioses: absence of Nod genes in photosynthetic bradyrhizobia. Science 316, 1307–1312 (2007).
Gourion, B., Berrabah, F., Ratet, P. & Stacey, G. Rhizobium–legume symbioses: the crucial role of plant immunity. Trends Plant Sci. 20, 186–194 (2015).
Liang, Y. et al. Nonlegumes respond to rhizobial Nod factors by suppressing the innate immune response. Science 341, 1384–1387 (2013). This paper shows that LCOs can suppress immunity signalling in a wide array of plant species.
Lebeis, S. L. et al. Salicylic acid modulates colonization of the root microbiome by specific bacterial taxa. Science 349, 860–864 (2015). This paper shows the importance of defence signalling for regulating bacterial associations in the plant microbiome.
Berrabah, F. et al. A nonRD receptor-like kinase prevents nodule early senescence and defense-like reactions during symbiosis. New Phytol. 203, 1305–1314 (2014).
Berrabah, F., Ratet, P. & Gourion, B. Multiple steps control immunity during the intracellular accommodation of rhizobia. J. Exp. Bot. 66, 1977–1985 (2015). This paper demonstrates that a suite of genes are required to regulate immunity in the nitrogen-fixing nodule.
Domonkos, A. et al. The identification of novel loci required for appropriate nodule development in Medicago truncatula . BMC Plant Biol. 13, 157 (2013).
Wang, C. et al. NODULES WITH ACTIVATED DEFENSE 1 is required for maintenance of rhizobial endosymbiosis in Medicago truncatula . New Phytol. 212, 176–191 (2016).
Bourcy, M. et al. Medicago truncatula DNF2 is a PI–PLC–XD-containing protein required for bacteroid persistence and prevention of nodule early senescence and defense-like reactions. New Phytol. 197, 1250–1261 (2013).
Zipfel, C. Plant pattern-recognition receptors. Trends Immunol. 35, 345–351 (2014).
Zuo, W. et al. A maize wall-associated kinase confers quantitative resistance to head smut. Nature Genet. 47, 151–157 (2015).
Liu, Y. et al. A gene cluster encoding lectin receptor kinases confers broad-spectrum and durable insect resistance in rice. Nature Biotechnol. 33, 301–305 (2015).
Hurni, S. et al. The maize disease resistance gene Htn1 against northern corn leaf blight encodes a wall-associated receptor-like kinase. Proc. Natl Acad. Sci. USA 112, 8780–8785 (2015).
Hind, S. R. et al. Tomato receptor FLAGELLIN-SENSING 3 binds flgII-28 and activates the plant immune system. Nature Plants 2, 16128 (2016).
Shiu, S. H. & Bleecker, A. B. Expansion of the receptor-like kinase/Pelle gene family and receptor-like proteins in Arabidopsis . Plant Physiol. 132, 530–543 (2003).
Fischer, I., Dievart, A., Droc, G., Dufayard, J. F. & Chantret, N. Evolutionary dynamics of the leucine-rich repeat receptor-like kinase (LRR-RLK) subfamily in angiosperms. Plant Physiol. 170, 1595–1610 (2016).
Lacombe, S. et al. Interfamily transfer of a plant pattern-recognition receptor confers broad-spectrum bacterial resistance. Nature Biotechnol. 28, 365–369 (2010).
Mendes, B. M. J. et al. Reduction in susceptibility to Xanthomonas axonopodis pv. citri in transgenic Citrus sinens is expressing the rice Xa21 gene. Plant Pathol. 59, 68–75 (2010).
Tripathi, J. N., Lorenzen, J., Bahar, O., Ronald, P. & Tripathi, L. Transgenic expression of the rice Xa21 pattern-recognition receptor in banana (Musa sp.) confers resistance to Xanthomonas campestris pv musacearum . Plant Biotechnol. J. (2014).
Schoonbeek, H.-J. et al. Arabidopsis EF-Tu receptor enhances bacterial disease resistance in transgenic wheat. New Phytol. 206, 606–613 (2015).
Schwessinger, B. et al. Transgenic expression of the dicotyledonous pattern recognition receptor EFR in rice leads to ligand-dependent activation of defense responses. PLoS Pathog. 11, e1004809 (2015).
Lu, F. et al. Enhancement of innate immune system in monocot rice by transferring the dicotyledonous elongation factor Tu receptor EFR. J. Integr. Plant Biol. 57, 641–652 (2015).
Du, J. et al. Elicitin recognition confers enhanced resistance to Phytophthora infestans in potato. Nature Plants 1, 15034 (2015).
Albert, I. et al. An RLP23–SOBIR1–BAK1 complex mediates NLP-triggered immunity. Nature Plants 1, 15140 (2015).
Hao, G., Pitino, M., Duan, Y. & Stover, E. Reduced susceptibility to Xanthomonas citri in transgenic citrus expressing the FLS2 receptor from Nicotiana benthamiana . Mol. Plant Microbe Interact. 29, 132–142 (2016).
Stokstad, E. The nitrogen fix. Science 353, 1225–1227 (2016).
Ivleva, N. B., Groat, J., Staub, J. M. & Stephens, M. Expression of active subunit of nitrogenase via integration into plant organelle genome. PLoS ONE 11, e0160951 (2016).
López-Torrejón, G. et al. Expression of a functional oxygen-labile nitrogenase component in the mitochondrial matrix of aerobically grown yeast. Nature Commun. 7, 11426 (2016).
Acknowledgements
Research on this topic in the authors' laboratories is supported by: the Gatsby Charitable Foundation (C.Z.); the European Research Council (PHOSPHinnATE; C.Z.); the 2Blades Foundation (C.Z.); the UK Biotechnology and Biological Sciences Research Council (BB/J004553/1; C.Z. and G.O.); and the Bill and Melinda Gates Foundation (G.O.). The authors thank members of their laboratories for continuous discussions and P. Kalo for providing the images presented in Fig. 4. We apologize to our colleagues whose work could not be cited owing to space limitations.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reprints and permissions information is available at www.nature.com/reprints.
Reviewer Information Nature thanks B. Thomma, J. Dangl and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Rights and permissions
About this article
Cite this article
Zipfel, C., Oldroyd, G. Plant signalling in symbiosis and immunity. Nature 543, 328–336 (2017). https://doi.org/10.1038/nature22009
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature22009
This article is cited by
-
Temporally-coordinated bivalent histone modifications of BCG1 enable fungal invasion and immune evasion
Nature Communications (2024)
-
Degradation of wild fruit forests created less diverse and diffuse bacterial communities decreased bacterial diversity, enhanced fungal pathogens and altered microbial assembly in the Tianshan Mountain, China
Plant and Soil (2024)
-
Root transcriptomic provides insights on molecular mechanisms involved in the tolerance to water deficit in Pisum sativum inoculated with Pseudomonas sp.
Planta (2024)
-
Genome-wide identification and expression analysis of the cyclic nucleotide-gated ion channel (CNGC) gene family in Saccharum spontaneum
BMC Genomics (2023)
-
Plant latent defense response against compatibility
The ISME Journal (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.