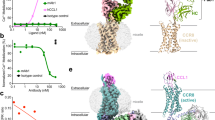
Abstract
Chemokines and their G-protein-coupled receptors play a diverse role in immune defence by controlling the migration, activation and survival of immune cells1. They are also involved in viral entry, tumour growth and metastasis and hence are important drug targets in a wide range of diseases2,3. Despite very significant efforts by the pharmaceutical industry to develop drugs, with over 50 small-molecule drugs directed at the family entering clinical development, only two compounds have reached the market: maraviroc (CCR5) for HIV infection and plerixafor (CXCR4) for stem-cell mobilization4. The high failure rate may in part be due to limited understanding of the mechanism of action of chemokine antagonists and an inability to optimize compounds in the absence of structural information5. CC chemokine receptor type 9 (CCR9) activation by CCL25 plays a key role in leukocyte recruitment to the gut and represents a therapeutic target in inflammatory bowel disease6. The selective CCR9 antagonist vercirnon progressed to phase 3 clinical trials in Crohn’s disease but efficacy was limited, with the need for very high doses to block receptor activation6. Here we report the crystal structure of the CCR9 receptor in complex with vercirnon at 2.8 Å resolution. Remarkably, vercirnon binds to the intracellular side of the receptor, exerting allosteric antagonism and preventing G-protein coupling. This binding site explains the need for relatively lipophilic ligands and describes another example of an allosteric site on G-protein-coupled receptors7 that can be targeted for drug design, not only at CCR9, but potentially extending to other chemokine receptors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Pease, J. E. Targeting chemokine receptors in allergic disease. Biochem. J. 434, 11–24 (2011)
Wilkin, T. J. & Gulick, R. M. CCR5 antagonism in HIV infection: current concepts and future opportunities. Annu. Rev. Med. 63, 81–93 (2012)
Vela, M., Aris, M., Llorente, M., Garcia-Sanz, J. A. & Kremer, L. Chemokine receptor-specific antibodies in cancer immunotherapy: achievements and challenges. Front. Immunol. 6, 12 (2015)
Solari, R., Pease, J. E. & Begg, M. “Chemokine receptors as therapeutic targets: why aren’t there more drugs?”. Eur. J. Pharmacol. 746, 363–367 (2015)
Pease, J. & Horuk, R. Chemokine receptor antagonists. J. Med. Chem. 55, 9363–9392 (2012)
Wendt, E. & Keshav, S. CCR9 antagonism: potential in the treatment of inflammatory bowel disease. Clin. Exp. Gastroenterol. 8, 119–130 (2015)
Changeux, J. P. & Christopoulos, A. Allosteric modulation as a unifying mechanism for receptor function and regulation. Cell 166, 1084–1102 (2016)
Serrano-Vega, M. J., Magnani, F., Shibata, Y. & Tate, C. G. Conformational thermostabilization of the β1-adrenergic receptor in a detergent-resistant form. Proc. Natl Acad. Sci. USA 105, 877–882 (2008)
Robertson, N. et al. The properties of thermostabilised G protein-coupled receptors (StaRs) and their use in drug discovery. Neuropharmacology 60, 36–44 (2011)
Walters, M. J. et al. Characterization of CCX282-B, an orally bioavailable antagonist of the CCR9 chemokine receptor, for treatment of inflammatory bowel disease. J. Pharmacol. Exp. Ther. 335, 61–69 (2010)
Tan, Q. et al. Structure of the CCR5 chemokine receptor-HIV entry inhibitor maraviroc complex. Science 341, 1387–1390 (2013)
Wu, B. et al. Structures of the CXCR4 chemokine GPCR with small-molecule and cyclic peptide antagonists. Science 330, 1066–1071 (2010)
Hollenstein, K. et al. Structure of class B GPCR corticotropin-releasing factor receptor 1. Nature 499, 438–443 (2013)
Jazayeri, A. et al. Extra-helical binding site of a glucagon receptor antagonist. Nature 533, 274–277 (2016)
Tchernychev, B. et al. Discovery of a CXCR4 agonist pepducin that mobilizes bone marrow hematopoietic cells. Proc. Natl Acad. Sci. USA 107, 22255–22259 (2010)
Andrews, G., Jones, C. & Wreggett, K. A. An intracellular allosteric site for a specific class of antagonists of the CC chemokine G protein-coupled receptors CCR4 and CCR5. Mol. Pharmacol. 73, 855–867 (2008)
Gonsiorek, W. et al. Pharmacological characterization of Sch527123, a potent allosteric CXCR1/CXCR2 antagonist. J. Pharmacol. Exp. Ther. 322, 477–485 (2007)
Salchow, K. et al. A common intracellular allosteric binding site for antagonists of the CXCR2 receptor. Br. J. Pharmacol. 159, 1429–1439 (2010)
Lazaar, A. L. et al. SB-656933, a novel CXCR2 selective antagonist, inhibits ex vivo neutrophil activation and ozone-induced airway inflammation in humans. Br. J. Clin. Pharmacol. 72, 282–293 (2011)
Nicholls, D. J. et al. Identification of a putative intracellular allosteric antagonist binding-site in the CXC chemokine receptors 1 and 2. Mol. Pharmacol. 74, 1193–1202 (2008)
de Kruijf, P. et al. Nonpeptidergic allosteric antagonists differentially bind to the CXCR2 chemokine receptor. J. Pharmacol. Exp. Ther. 329, 783–790 (2009)
Thelen, M. Dancing to the tune of chemokines. Nature Immunol. 2, 129–134 (2001)
Deupi, X. & Standfuss, J. Structural insights into agonist-induced activation of G-protein-coupled receptors. Curr. Opin. Struct. Biol. 21, 541–551 (2011)
Tehan, B. G., Bortolato, A., Blaney, F. E., Weir, M. P. & Mason, J. S. Unifying family A GPCR theories of activation. Pharmacol. Ther. 143, 51–60 (2014)
Rasmussen, S. G. et al. Crystal structure of the β2 adrenergic receptor-Gs protein complex. Nature 477, 549–555 (2011)
Kang, Y. et al. Crystal structure of rhodopsin bound to arrestin by femtosecond X-ray laser. Nature 523, 561–567 (2015)
Ungashe, S. et al. Aryl sulphonamides. US patent 2006/0111351A1 (2006)
Nugent, T. & Jones, D. T. Membrane protein orientation and refinement using a knowledge-based statistical potential. BMC Bioinformatics 14, 276–285 (2013)
Alva, V. et al. The MPI bioinformatics Toolkit as an integrative platform for advanced protein sequence and structure analysis. Nucleic Acids Res. 44 (Suppl. W1), W410–W415 (2016)
Kawate, T. & Gouaux, E. Fluorescence-detection size-exclusion chromatography for precrystallization screening of integral membrane proteins. Structure 14, 673–681 (2006)
Caffrey, M. & Cherezov, V. Crystallizing membrane proteins using lipidic mesophases. Nature Protocols 4, 706–731 (2009)
Kabsch, W. Integration, scaling, space-group assignment and post-refinement. Acta Crystallogr. D 66, 133–144 (2010)
Collaborative Computational Project, Number 4. The CCP4 suite: programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994)
Evans, P. R. & Murshudov, G. N. How good are my data and what is the resolution? Acta Crystallogr. D 69, 1204–1214 (2013)
McCoy, A. J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007)
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010)
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D 66, 213–221 (2010)
Murshudov, G. N. et al. REFMAC5 for the refinement of macromolecular crystal structures. Acta Crystallogr. D 67, 355–367 (2011)
Afonine, P. V. et al. Towards automated crystallographic structure refinement with phenix.refine. Acta Crystallogr. D 68, 352–367 (2012)
Martyna, G. J., Tobias, D. J. & Klein, M. L. Constant pressure molecular dynamics algorithms. J. Chem. Phys. 101, 4177–4189 (1994)
Diederichs, K. & Karplus, P. A. Better models by discarding data? Acta Crystallogr. D 69, 1215–1222 (2013)
Acknowledgements
We thank D. Axford, R. Owen and D. Sherrell at I24, Diamond Light Source, Oxford, UK, for technical support. We thank colleagues at Heptares Therapeutics for suggestions and comments, and G. Brown and S. Bucknell for assistance in radioligand preparation.
Author information
Authors and Affiliations
Contributions
J.K. and A.J. devised and performed the conformational thermostabilization and mutagenesis of the receptor, characterized expression constructs and performed radioligand binding analysis of mutants. Computational analysis of the structure and modelling was performed by F.D. and J.S.M. A.S.D. established the platform/protocols for LCP crystallization and solved the structure. J.C.E. supported expression and scouted purification of the final StaR. M.R. designed and characterized all constructs, collected and processed X-ray diffraction data and solved the structure. C.O. optimized purification, performed LCP crystallization, harvested crystals, collected and processed X-ray diffraction data, and solved and refined the structure. K.B. performed and analysed the pharmacology data. J.A.C. and M.C. identified and sourced the chemical compound(s) used in the study. Project management was performed by J.A.C., R.M.C. and F.H.M. The manuscript was prepared by A.S.D., C.O., F.D., M.C. and F.H.M. All authors contributed to the final editing and approval of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors are employees of Heptares Therapeutics and are shareholders of Sosei Group, the parent company of Heptares. Heptares is a drug discovery and development company working in the field of G-protein-coupled receptor structure-based drug design.
Extended data figures and tables
Extended Data Figure 1 CCR9 crystallization construct StaR(25-340) in schematic representation.
Thermostabilizing mutations (green) are Thr77Ala, Val79Ala, Met82Ala, Ser141Cys, Thr216Ala, Val255Ala, Asn294Ala, Thr304Ala. Further mutations to remove sites of post-translational modifications (light blue) are Cys337Ala and Thr34Glu. Residues forming the allosteric pocket are pink. Disordered residues in the structure are grey. The disulfide bonds between (Cys1193.25) and extracellular loop 2 and linking the N terminus (Cys38) with the top of TM7 (Cys2897.25) are denoted by dashed yellow lines.
Extended Data Figure 2 Comparison of wild-type and thermostabilized CCR9 in radioligand binding of [3H]vercirnon.
The thermal stability of wild-type CCR9 (filled circles) and CCR9-StaR(1-369) (open circles) analysed in decyl-maltoside are shown. Error bars are derived from standard deviations and calculated from duplicate temperature points (n = 2) within a single experiment. Data shown are representative of three independent experiments. CCR9-StaR(1-369) produced a mean Tm of 39.5 °C. The Tm of wild-type CCR9 was not determinable under these conditions; however, binding is observed and a Tm can be calculated in dodecyl-maltoside (data not shown).
Extended Data Figure 3 Pharmacology of WT CCR9 and CCR9-StaR.
Saturation binding experiments performed in membranes from HEK293 cells transiently expressing (a) human CCR9 or (b) CCR9-StaR(1-369). Non-specific binding was determined by addition of 1 μM cold vercirnon. Data shown as mean ± s.e.m. are representative of three independent experiments performed in duplicate. Data were fitted globally to a one-site saturation isotherm. Affinity and expression level (Bmax) values are given below the graphs for both WT CCR9 and CCR9-StaR(1-369). There was no difference in the affinity of [3H]vercirnon at WT CCR9 or CCR9-StaR(1-369) (unpaired, two-tailed t-test = 0.51). CCR9-StaR(1-369) showed significantly higher expression levels (Bmax) than WT CCR9 (unpaired, two-tailed t-test = 0.0007).
Extended Data Figure 4 Crystal packing in the CCR9-StaR(25-340) triclinic system.
a, Typical CCR9-StaR(25-340) non-fusion crystals grown in LCP complexed with vercirnon. b, The two copies of CCR9-StaR(25-340) in the triclinic asymmetric unit assemble in a parallel fashion with contacts mediated by TM4 – CCR9-StaR(25-340) shown in chainbow colouration (blue to red equals N to C terminus). c, View as in b rotated by 90° with the two copies of CCR9-StaR(25-340) now coloured yellow and cyan. d–f, Views of CCR9-StaR(25-340) packing in the triclinic crystal system along the a, b and c axes respectively, molecules coloured as in c.
Extended Data Figure 6 Electron density around the vercirnon binding site.
a, Cross-eye stereoscopic view of 2Fo − Fc density contoured at 1.6σ covering vercirnon and surrounding residues as viewed from intracellular space. Vercirnon in stick representation, with carbon, nitrogen, chlorine, sulfur and oxygen atoms coloured magenta, blue, green, yellow and red, respectively; CCR9 in stick representation with carbon, nitrogen, oxygen and sulfur atoms coloured white, blue, red and yellow respectively. b, View as in a rotated by 180°.
Extended Data Figure 7 Saturation binding analysis of mutants with [3H]vercirnon.
a–f, Saturation binding of [3H]vercirnon to homogenized cell lysates containing indicated mutant variants of CCR9. Data are representative of three independent experiments performed in duplicate ± s.d. Kd values (inset) are mean of three independent experiments with s.d. in parentheses. The datasets for L87F, Y317A and G321A could not be analysed unambiguously owing to near-complete loss of specific binding.
Extended Data Figure 8 Molecular dynamics analysis of the CCR9–vercirnon complex.
a, Stability of the CCR9–vercirnon complex during 100 ns molecular dynamics. Structural alignment of the wild-type CCR9–vercirnon complex at 0 (blue), 25 (green), 50 (yellow), 75 (orange) and 100 ns (red) molecular dynamics. Proteins are shown as ribbon with TM7 partly hidden for clarity; vercirnon is represented in sticks. b, Two-dimensional representation of the ligand–protein contacts. c, d, Induced-fit binding of vercirnon to CCR9. Superposition of the CCR9–vercirnon complex (cyan) and the pseudo-apo state of CCR9 receptor at 100 ns molecular dynamics (magenta). Vercirnon is shown as sticks with carbons coloured in grey. Arg323, Phe324 and Tyr317 are shown as sticks with carbons coloured in cyan for the crystallographic structure and stick (c) or spheres (d) with carbons coloured in magenta for the molecular dynamics output.
Supplementary information
Supplementary Figure
This file contains Supplementary Figure 1, preparation of [3H]vercirnon and vercirnon. (PDF 39 kb)
Rights and permissions
About this article
Cite this article
Oswald, C., Rappas, M., Kean, J. et al. Intracellular allosteric antagonism of the CCR9 receptor. Nature 540, 462–465 (2016). https://doi.org/10.1038/nature20606
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature20606
This article is cited by
-
Class B1 GPCR activation by an intracellular agonist
Nature (2023)
-
Allosteric modulation of dopamine D2L receptor in complex with Gi1 and Gi2 proteins: the effect of subtle structural and stereochemical ligand modifications
Pharmacological Reports (2022)
-
Activation pathway of a G protein-coupled receptor uncovers conformational intermediates as targets for allosteric drug design
Nature Communications (2021)
-
G protein-coupled receptors: structure- and function-based drug discovery
Signal Transduction and Targeted Therapy (2021)
-
Ligand-binding and -scavenging of the chemerin receptor GPR1
Cellular and Molecular Life Sciences (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.