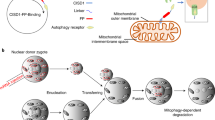
Abstract
Maternally inherited mitochondrial (mt)DNA mutations can cause fatal or severely debilitating syndromes in children1,2,3, with disease severity dependent on the specific gene mutation and the ratio of mutant to wild-type mtDNA (heteroplasmy) in each cell and tissue4. Pathogenic mtDNA mutations are relatively common, with an estimated 778 affected children born each year in the United States5. Mitochondrial replacement therapies or techniques (MRT) circumventing mother–to–child mtDNA disease transmission involve replacement of oocyte maternal mtDNA6,7,8. Here we report MRT outcomes in several families with common mtDNA syndromes. The mother’s oocytes were of normal quality and mutation levels correlated with those in existing children. Efficient replacement of oocyte mutant mtDNA was performed by spindle transfer8, resulting in embryos containing >99% donor mtDNA. Donor mtDNA was stably maintained in embryonic stem cells (ES cells) derived from most embryos. However, some ES cell lines demonstrated gradual loss of donor mtDNA and reversal to the maternal haplotype. In evaluating donor–to–maternal mtDNA interactions, it seems that compatibility relates to mtDNA replication efficiency rather than to mismatch or oxidative phosphorylation dysfunction. We identify a polymorphism within the conserved sequence box II region of the D-loop as a plausible cause of preferential replication of specific mtDNA haplotypes. In addition, some haplotypes confer proliferative and growth advantages to cells. Hence, we propose a matching paradigm for selecting compatible donor mtDNA for MRT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Archer, S. L. Mitochondrial dynamics–mitochondrial fission and fusion in human diseases. N. Engl. J. Med. 369, 2236–2251 (2013)
Koopman, W. J., Willems, P. H. & Smeitink, J. A. Monogenic mitochondrial disorders. N. Engl. J. Med. 366, 1132–1141 (2012)
Schon, E. A., DiMauro, S. & Hirano, M. Human mitochondrial DNA: roles of inherited and somatic mutations. Nat. Rev. Genet. 13, 878–890 (2012)
Wallace, D. C. & Chalkia, D. Mitochondrial DNA genetics and the heteroplasmy conundrum in evolution and disease. Cold Spring Harb. Perspect. Biol . 5, a021220 (2013)
Gorman, G. S. et al. Mitochondrial donation–how many women could benefit? N. Engl. J. Med. 372, 885–887 (2015)
Wolf, D. P., Mitalipov, N. & Mitalipov, S. Mitochondrial replacement therapy in reproductive medicine. Trends Mol. Med. 21, 68–76 (2015)
Craven, L. et al. Pronuclear transfer in human embryos to prevent transmission of mitochondrial DNA disease. Nature 465, 82–85 (2010)
Tachibana, M. et al. Mitochondrial gene replacement in primate offspring and embryonic stem cells. Nature 461, 367–372 (2009)
Wallace, D. C. A mitochondrial bioenergetic etiology of disease. J. Clin. Invest. 123, 1405–1412 (2013)
McClelland, K. MELAS Within a family: Mitocondrial DNA Heteroplamy and Clinical Varability. MSc thesis, California State University (2014)
Monnot, S. et al. Segregation of mtDNA throughout human embryofetal development: m.3243A>G as a model system. Hum. Mutat. 32, 116–125 (2011)
Smeets, H. J., Sallevelt, S. C., Dreesen, J. C., de Die-Smulders, C. E. & de Coo, I. F. Preventing the transmission of mitochondrial DNA disorders using prenatal or preimplantation genetic diagnosis. Ann. NY Acad. Sci . 1350, 29–36 (2015)
Kelsey, T. W., Wright, P., Nelson, S. M., Anderson, R. A. & Wallace, W. H. A validated model of serum anti-Müllerian hormone from conception to menopause. PLoS One 6, e22024 (2011)
Kelsey, T. W., Anderson, R. A., Wright, P., Nelson, S. M. & Wallace, W. H. Data-driven assessment of the human ovarian reserve. Mol. Hum. Reprod. 18, 79–87 (2012)
Brown, D. T., Samuels, D. C., Michael, E. M., Turnbull, D. M. & Chinnery, P. F. Random genetic drift determines the level of mutant mtDNA in human primary oocytes. Am. J. Hum. Genet. 68, 533–536 (2001)
Boucret, L. et al. Relationship between diminished ovarian reserve and mitochondrial biogenesis in cumulus cells. Hum. Reprod. 30, 1653–1664 (2015)
Woodson, J. D. & Chory, J. Coordination of gene expression between organellar and nuclear genomes. Nat. Rev. Genet. 9, 383–395 (2008)
Kearney, H. M., Thorland, E. C., Brown, K. K., Quintero-Rivera, F. & South, S. T. American College of Medical Genetics standards and guidelines for interpretation and reporting of postnatal constitutional copy number variants. Genet. Med. 13, 680–685 (2011)
Tachibana, M. et al. Towards germline gene therapy of inherited mitochondrial diseases. Nature 493, 627–631 (2013)
Paull, D. et al. Nuclear genome transfer in human oocytes eliminates mitochondrial DNA variants. Nature 493, 632–637 (2013)
Tachibana, M. et al. Human embryonic stem cells derived by somatic cell nuclear transfer. Cell 153, 1228–1238 (2013)
Ma, H. et al. Metabolic rescue in pluripotent cells from patients with mtDNA disease. Nature 524, 234–238 (2015)
Kang, E. et al. Age-related accumulation of somatic mitochondrial DNA mutations in adult-derived human iPSCs. Cell Stem Cell 18, 625–636 (2016)
Burgstaller, J. P., Johnston, I. G. & Poulton, J. Mitochondrial DNA disease and developmental implications for reproductive strategies. Mol. Hum. Reprod. 21, 11–22 (2015)
Agaronyan, K., Morozov, Y. I., Anikin, M. & Temiakov, D. Mitochondrial biology. Replication–transcription switch in human mitochondria. Science 347, 548–551 (2015)
Jemt, E. et al. Regulation of DNA replication at the end of the mitochondrial D-loop involves the helicase TWINKLE and a conserved sequence element. Nucleic Acids Res . 43, 9262–9275 (2015)
Yamada, M. et al. Genetic drift can compromise mitochondrial replacement by nuclear transfer in human oocytes. Cell Stem Cell 18, 749–754 (2016)
Hyslop, L. A. et al. Towards clinical application of pronuclear transfer to prevent mitochondrial DNA disease. Nature 534, 383–386 (2016)
Lee, H. S. et al. Rapid mitochondrial DNA segregation in primate preimplantation embryos precedes somatic and germline bottleneck. Cell Reports 1, 506–515 (2012)
Koehler, C. M. et al. Replacement of bovine mitochondrial DNA by a sequence variant within one generation. Genetics 129, 247–255 (1991)
Wu, J. et al. An alternative pluripotent state confers interspecies chimaeric competency. Nature 521, 316–321 (2015)
Lian, X. et al. Directed cardiomyocyte differentiation from human pluripotent stem cells by modulating Wnt/β-catenin signaling under fully defined conditions. Nat. Protoc. 8, 162–175 (2013)
Spinazzi, M., Casarin, A., Pertegato, V., Salviati, L. & Angelini, C. Assessment of mitochondrial respiratory chain enzymatic activities on tissues and cultured cells. Nat. Protoc. 7, 1235–1246 (2012)
Monnot, S. et al. Mutation dependance of the mitochondrial DNA copy number in the first stages of human embryogenesis. Hum. Mol. Genet. 22, 1867–1872 (2013)
Ma, H. et al. Abnormalities in human pluripotent cells due to reprogramming mechanisms. Nature 511, 177–183 (2014)
Acknowledgements
The authors acknowledge the OHSU Embryonic Stem Cell Research Oversight Committee and the Institutional Review Board for oversight and guidance. We thank all study participants for tissue donations and the Women’s Health Research Unit staff, University Fertility Consultants and the Reproductive Endocrinology and Infertility Division in the Department of Obstetrics and Gynecology, Oregon Health and Science University for support and procurement of human gametes. Studies were supported by the Leducq Foundation, OHSU institutional funds and Cincinnati Children’s Hospital Research Foundation. Work in the laboratory of J.C.I.B. was supported by the G. Harold and Leila Y. Mathers Charitable Foundation, the Leona M. and Harry B. Helmsley Charitable Trust and the Moxie Foundation. A.P.L. was partially supported by a fellowship from the Hewitt Foundation. P.M.R. was partially supported by a fellowship from Fundación Alfonso Martín Escudero.
Author information
Authors and Affiliations
Contributions
E.K., N.M.G., P.A. and S.M. conceived the study and designed the experiments. T.Hu., P.A. and A.K. evaluated clinical genetic results and coordinated recruitment of study participants. P.A. and J.J. performed skin biopsies and blood collections. P.A., D.B., D.L. and D.W. performed ovarian stimulations and oocyte retrievals. N.M.G., R.T.H. and S.M. conducted spindle transfer, intracytoplasmic sperm injection, embryo culture and establishment of ES cell lines. E.K., R.T.H., H.M., Y.Le., Y.Li, R.A., T.Ha. and N.M.G. cultured ES cells. E.K., C.V.D., Y.Le. and T.Ha. performed teratoma studies. E.K., C.V.D., Y.Li and D.J. performed mitochondrial complex assays. J.W., P.M.R. and A.P.L. performed in vitro differentiation and Seahorse assays. K.A. and D.T. performed mtDNA transcription experiments. E.K., C.V.D., R.T.H. and A.K. prepared mtDNA and performed MiSeq assays. E.K. and Y.Le. performed NGS data analysis and interpretation. E.K., C.V.D. validated mtDNA mutations by Sanger sequencing. H.M. and R.T.H. performed ARMS–qPCR. X.W., S.L. and T.Hu. performed CNV data analysis. R.K. and C.C. performed the comparative genome hybridization array. S.O. performed G-banding karyotypes. E.K., J.W., D.P.W., J.C.I.B., P.A. and S.M. analysed data and wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer Information Nature thanks E. Shoubridge and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Extended data figures and tables
Extended Data Figure 1 Families with mitochondrial disease.
a, Families with mitochondrial disease and women of reproductive age were recruited for MRT. All Leigh syndrome families had an existing, severely affected child. F, family; non-syn, non-synonymous; NA, not applicable. b, Family 5 was selected from an extensive MELAS syndrome pedigree. Clinical phenotypes of A3243G MELAS syndrome varied even with similar heteroplasmy levels. Asterisk indicates the first MELAS patient diagnosed clinically. Heteroplasmy in blood (upper panel) and urine (lower panel). Squares, males; circles, females. B, blood; U, urine. NT, not tested. Percentage values refer to heteroplasmy; y, years-old.
Extended Data Figure 2 Response to controlled ovarian stimulation and oocyte characteristics.
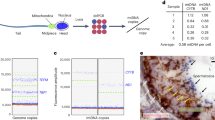
a, Age of oocyte donor(s) was similar between carrier and healthy groups. b, c, Ovarian reserve (b) and antral follicle count (c) were significantly lower in carriers than in healthy oocyte donors. d, e, Duration of controlled ovarian stimulation (d, COS) was longer in carriers and peak oestradiol (e, E2) on day of hCG trended to be lower in carriers. f, g, Total oocyte yield (f) and number of mature oocytes (g) were significantly lower in carriers than healthy oocyte donors. n = the number of controlled ovarian stimulation cycles. Data are represented as mean ± s.d. *P < 0.05. h, Baseline characteristics and cycle outcomes. Carriers had a lower anti-Müllerian hormone (AMH) levels. Lower peak oestradiol levels were measured in cED1 and cED2. Birth control method: combined oral contraceptive, medroxyprogesterone. i, Analysis of heteroplasmic mtDNA variants detected in MII oocytes. De novo indicates unique mutations found in individual oocytes; inherited indicates mutations shared with other oocytes, sibling children or egg donors. n = the number of mutations in individual oocytes. j, anti-Müllerian hormone levels, a measure of ovarian reserve, were not correlated with mtDNA copy number. n = the number of oocytes. NS, not significant. Source data files are available for a–g.
Extended Data Figure 3 Fertilization and development after spindle transfer.
a, Whole mtDNA sequencing was performed to identify haplotypes of all egg donors and spindle transfer was performed to match various haplotypes. Cyto, cytoplast; Karyo, karyoplast. b, Normal fertilization (2 pronuclei) and abnormal fertilization (1 or 3 pronuclei); see arrows. Scale bars, 50 μm. c, Carrier spindle transfer showed higher abnormal fertilization than controls. One zygote from the control spindle transfer group with three pronuclei developed to a blastocyst and displayed a 69, XXY karyotype. d, The blastulation rate was similar between vitrified cytoplasm with fresh spindles and vitrified spindles with fresh cytoplasm (P > 0.05; t-test). e, Control spindle transfer embryo development as a function of donor mtDNA matching distances. The numbers on the top of bars are percentage of embryo development. n = the number of embryos; NS, not significant (P > 0.05; t-test).
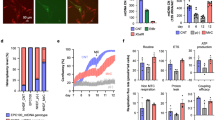
Extended Data Figure 4 Mitochondrial function in cells with donor mtDNA.
a, Energy metabolism  was measured and compared between neural progenitor cells (NPCs) derived from MRT and control embryos carrying either donor or maternal mtDNA. OCR, oxygen consumption rate (representing oxidative phosphorylation); ECAR, extracellular acidification rate (representing glycolysis). NPCs from MRT ES cells with 6 and 49 SNP differences displayed comparable oxygen consumption rates to NPCs harbouring maternal or donor mtDNA except maximal respiration in the 49 SNP group. The oxygen consumption rate was reduced in NPCs with 33 SNPs. The oxygen consumption rate data were normalized by live cell DNA content. *P < 0.05 versus host mtDNA, **P < 0.05 versus donor mtDNA, ***P < 0.05 versus spindle transfer (t-test for 6 SNPs and one-way ANOVA for 33 and 49 SNPs, n = 16 technical replicates). Data are shown as mean ± s.e.m. NS, not significant. b, Mitochondrial respiratory chain enzyme activity in differentiated cells from spindle transfer ES cell lines. Mitochondrial complex I and IV activities in differentiated cells from spindle transfer ES cell lines carrying donor mtDNA with 49 SNP differences were comparable to controls with either donor or maternal mtDNA (P > 0.05). One-way ANOVA, n = 4 technical replicates. Source data file is available for b.
was measured and compared between neural progenitor cells (NPCs) derived from MRT and control embryos carrying either donor or maternal mtDNA. OCR, oxygen consumption rate (representing oxidative phosphorylation); ECAR, extracellular acidification rate (representing glycolysis). NPCs from MRT ES cells with 6 and 49 SNP differences displayed comparable oxygen consumption rates to NPCs harbouring maternal or donor mtDNA except maximal respiration in the 49 SNP group. The oxygen consumption rate was reduced in NPCs with 33 SNPs. The oxygen consumption rate data were normalized by live cell DNA content. *P < 0.05 versus host mtDNA, **P < 0.05 versus donor mtDNA, ***P < 0.05 versus spindle transfer (t-test for 6 SNPs and one-way ANOVA for 33 and 49 SNPs, n = 16 technical replicates). Data are shown as mean ± s.e.m. NS, not significant. b, Mitochondrial respiratory chain enzyme activity in differentiated cells from spindle transfer ES cell lines. Mitochondrial complex I and IV activities in differentiated cells from spindle transfer ES cell lines carrying donor mtDNA with 49 SNP differences were comparable to controls with either donor or maternal mtDNA (P > 0.05). One-way ANOVA, n = 4 technical replicates. Source data file is available for b.
Extended Data Figure 5 Differentiation of ES cells derived from MRT embryos.
a, Summary of in vitro and in vivo differentiation of ES cells derived from spindle transfer or SCNT embryos carrying donor mtDNA. Similar to control IVF ES cells, all tested MRT ES cells produced teratoma tumours in vivo and formed neural progenitor cells (NPCs) and cardiomyocytes in vitro. NT, not tested. b, Histological analysis of differentiated tissues from MRT ES cells. The representative tissues were collected and used for mtDNA carry-over analysis and measurements of mitochondrial function. Scale bars, 1 mm (live image) and 50 μm (haematoxylin and eosin (H&E) and immunochemistry staining).
Extended Data Figure 6 Nuclear genome abnormalities associated with spindle transfer.
a, Aneuploidy rate in blastocysts determined by a comparative genome hybridization array was not significantly different in spindle transfer groups compared to controls. b, Karyotype abnormality rate in ES cells determined by G-banding analysis was also comparable among spindle transfer groups (P > 0.05). Normal, 46 XX or 46 XY. Number inside bars, the number of blastocysts or ES cell lines (abnormal/total). Simple χ2 tests.
Extended Data Figure 7 Maternal mtDNA heteroplasmy changes and mitochondrial function.
a, Maternal mtDNA (X2c haplotype) heteroplasmy increased during extended in vitro culture and reached homoplasmy in pooled cultures or in individual colony subcultures (n = 1). See also Supplementary Table 4. b, Respiratory chain complex 1 (COM I) and complex 4 (COM IV) enzyme activities were measured in fibroblasts carrying various human mtDNA haplotypes used in this study. No significant differences were noted (P > 0.05). One-way ANOVA, n = 4 technical replicates. Source data file is available for b.
Supplementary information
Supplementary Table 1
Sequence differences between egg donor haplotypes understudy by whole mtDNA sequencing (syn, synonymous; non-syn, nonsynonymous; frmshft, frameshift). (XLSX 35 kb)
Supplementary Table 2
mtDNA variants in oocytes and somatic tissues from mutant carriers and children and healthy oocyte donors (syn, synonymous; non-syn,non- synonymous; frmshft, frameshift). (XLSX 25 kb)
Supplementary Table 3
Maternal mtDNA analysis in ESCs derived from ST blastocysts (syn, synonymous; non-syn, non- synonymous; frmshft, frameshift). (XLSX 66 kb)
Supplementary Table 4
Analysis of maternal mtDNA heteroplasmy in ESCs derived from somatic cell nuclear transfer (SCNT) blastocysts (syn, synonymous; nonsyn, non- synonymous; frmshft, frameshift). (XLSX 47 kb)
Supplementary Table 5
Maternal mtDNA heteroplasmy changes in 18 ST-ES and 8NT-ES cell lines. Filtered reads were aligned to the human mitochondrial sequence reference NC_012920.1 followed by variant calling. Presented nucleotide positions indicate difference from the reference or between different human haplotypes. (XLSX 90 kb)
Rights and permissions
About this article
Cite this article
Kang, E., Wu, J., Gutierrez, N. et al. Mitochondrial replacement in human oocytes carrying pathogenic mitochondrial DNA mutations. Nature 540, 270–275 (2016). https://doi.org/10.1038/nature20592
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature20592
This article is cited by
-
Secondary structure of the human mitochondrial genome affects formation of deletions
BMC Biology (2023)
-
Nuclear genetic control of mtDNA copy number and heteroplasmy in humans
Nature (2023)
-
First UK children born using three-person IVF: what scientists want to know
Nature (2023)
-
Novel economical, accurate, sensitive, single-cell analytical method for mitochondrial DNA quantification in mtDNA mutation carriers
Journal of Assisted Reproduction and Genetics (2023)
-
Eight Strategies to Engineer Acceptance of Human Germline Modifications
Journal of Bioethical Inquiry (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.