Abstract
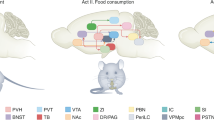
Atypical food intake is a primary cause of obesity and other eating and metabolic disorders. Insight into the neural control of feeding has previously focused mainly on signalling mechanisms associated with the hypothalamus1,2,3,4,5, the major centre in the brain that regulates body weight homeostasis6,7. However, roles of non-canonical central nervous system signalling mechanisms in regulating feeding behaviour have been largely uncharacterized. Acetylcholine has long been proposed to influence feeding8,9,10 owing in part to the functional similarity between acetylcholine and nicotine, a known appetite suppressant. Nicotine is an exogenous agonist for acetylcholine receptors, suggesting that endogenous cholinergic signalling may play a part in normal physiological regulation of feeding. However, it remains unclear how cholinergic neurons in the brain regulate food intake. Here we report that cholinergic neurons of the mouse basal forebrain potently influence food intake and body weight. Impairment of cholinergic signalling increases food intake and results in severe obesity, whereas enhanced cholinergic signalling decreases food consumption. We found that cholinergic circuits modulate appetite suppression on downstream targets in the hypothalamus. Together our data reveal the cholinergic basal forebrain as a major modulatory centre underlying feeding behaviour.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Aponte, Y., Atasoy, D. & Sternson, S. M. AGRP neurons are sufficient to orchestrate feeding behavior rapidly and without training. Nat. Neurosci. 14, 351–355 (2011)
Chen, Y., Lin, Y. C., Kuo, T. W. & Knight, Z. A. Sensory detection of food rapidly modulates arcuate feeding circuits. Cell 160, 829–841 (2015)
Luquet, S., Perez, F. A., Hnasko, T. S. & Palmiter, R. D. NPY/AgRP neurons are essential for feeding in adult mice but can be ablated in neonates. Science 310, 683–685 (2005)
Pinto, S. et al. Rapid rewiring of arcuate nucleus feeding circuits by leptin. Science 304, 110–115 (2004)
Garfield, A. S. et al. A neural basis for melanocortin-4 receptor-regulated appetite. Nat. Neurosci. 18, 863–871 (2015)
Morton, G. J., Cummings, D. E., Baskin, D. G., Barsh, G. S. & Schwartz, M. W. Central nervous system control of food intake and body weight. Nature 443, 289–295 (2006)
Elmquist, J. K., Elias, C. F. & Saper, C. B. From lesions to leptin: hypothalamic control of food intake and body weight. Neuron 22, 221–232 (1999)
Pistelli, F., Aquilini, F. & Carrozzi, L. Weight gain after smoking cessation. Monaldi Arch. Chest Dis. 71, 81–87 (2009)
Fulkerson, J. A. & French, S. A. Cigarette smoking for weight loss or control among adolescents: gender and racial/ethnic differences. J. Adolesc. Health 32, 306–313 (2003)
Voorhees, C. C., Schreiber, G. B., Schumann, B. C., Biro, F. & Crawford, P. B. Early predictors of daily smoking in young women: The National Heart, Lung, And Blood Institute growth and health study. Prev. Med. 34, 616–624 (2002)
Zaborszky, L., van den Pol, A. & Gyengesi, E. in The Mouse Nervous System (eds Watson C., Paxinos, G. & Puelles, L. ) 684–718 (2012)
Wang, D. et al. Whole-brain mapping of the direct inputs and axonal projections of POMC and AgRP neurons. Front. Neuroanat. 9, 40 (2015)
Sakurai, T. et al. Input of orexin/hypocretin neurons revealed by a genetically encoded tracer in mice. Neuron 46, 297–308 (2005)
Atasoy, D., Aponte, Y., Su, H. H. & Sternson, S. M. A FLEX switch targets Channelrhodopsin-2 to multiple cell types for imaging and long-range circuit mapping. J. Neurosci. 28, 7025–7030 (2008)
Saunders, A., Granger, A. J. & Sabatini, B. L. Corelease of acetylcholine and GABA from cholinergic forebrain neurons. eLife 4, e06412 (2015)
Ichikawa, T., Ajiki, K., Matsuura, J. & Misawa, H. Localization of two cholinergic markers, choline acetyltransferase and vesicular acetylcholine transporter in the central nervous system of the rat: in situ hybridization histochemistry and immunohistochemistry. J. Chem. Neuroanat. 13, 23–39 (1997)
Gropp, E. et al. Agouti-related peptide-expressing neurons are mandatory for feeding. Nat. Neurosci. 8, 1289–1291 (2005)
Mineur, Y. S. et al. Nicotine decreases food intake through activation of POMC neurons. Science 332, 1330–1332 (2011)
Soudais, C., Laplace-Builhe, C., Kissa, K. & Kremer, E. J. Preferential transduction of neurons by canine adenovirus vectors and their efficient retrograde transport in vivo. FASEB J. 15, 2283–2285 (2001)
Henry, F. E., Sugino, K., Tozer, A., Branco, T. & Sternson, S. M. Cell type-specific transcriptomics of hypothalamic energy-sensing neuron responses to weight-loss. eLife 4, e09800 (2015)
Ren, J. et al. Habenula “cholinergic” neurons co-release glutamate and acetylcholine and activate postsynaptic neurons via distinct transmission modes. Neuron 69, 445–452 (2011)
Saunders, A. et al. A direct GABAergic output from the basal ganglia to frontal cortex. Nature 521, 85–89 (2015)
Saper, C. B., Chou, T. C. & Elmquist, J. K. The need to feed: homeostatic and hedonic control of eating. Neuron 36, 199–211 (2002)
Liu, C., Lee, S. & Elmquist, J. K. Circuits controlling energy balance and mood: inherently intertwined or just complicated intersections? Cell Metab. 19, 902–909 (2014)
Wise, R. A. Brain reward circuitry: insights from unsensed incentives. Neuron 36, 229–240 (2002)
Picciotto, M. R., Higley, M. J. & Mineur, Y. S. Acetylcholine as a neuromodulator: cholinergic signaling shapes nervous system function and behavior. Neuron 76, 116–129 (2012)
Miwa, J. M., Freedman, R. & Lester, H. A. Neural systems governed by nicotinic acetylcholine receptors: emerging hypotheses. Neuron 70, 20–33 (2011)
Hangya, B., Ranade, S. P., Lorenc, M. & Kepecs, A. Central cholinergic neurons are rapidly recruited by reinforcement feedback. Cell 162, 1155–1168 (2015)
Hoebel, B. G., Avena, N. M. & Rada, P. Accumbens dopamine-acetylcholine balance in approach and avoidance. Curr. Opin. Pharmacol. 7, 617–627 (2007)
Website: © 2015 Allen Institute for Brain Science. Allen Mouse Brain Atlas [Internet]. Available from: http://mouse.brain-map.org
Acknowledgements
This study was supported by NIH grants 5F31NS089411 to A.M.H., R01NS078294 to B.R.A., R01DK109934 to B.R.A. and Q.T., P30DK079638 to the BCM Mouse Metabolic Core, and U54HD083092 to the BCM IDDRC. Support was also provided to B.R.A. from the Klarman Family Foundation, the Klingenstein-Simons Fellowship Award, the Brain and Behavior Research Foundation, the Charif Souki Fund, and the McNair Medical Institute. We thank H. Zoghbi, H. Bellen, M. Wang, and M. Krashes for input on this manuscript.
Author information
Authors and Affiliations
Contributions
B.R.A., A.M.H., and Q.T. designed all experiments. J.C.C., J.O.-G., A.M.H., I.H., M.K., J.M.P., K.Q., B.T., and K.U. performed experiments. B.R.A., A.M.H., and K.Q. analysed the data. Genetic reagents and viral constructs were engineered by B.R.A., A.M.H., I.H., and J.S. A.M.H. and B.R.A. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer Information
Nature thanks L. de Lecea and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Extended data figures and tables
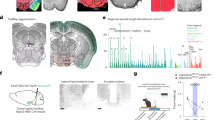
Extended Data Figure 1 Anatomy of the diagonal band of Broca.
a–c, The DBB is situated in the basal forebrain and consists of an anteriorly located vertical limb (VDB), which branches posteriorly into separate bilateral horizontal limbs (HDB). d, AAV-FLEX-tdTomato (pseudocoloured in green) injected bilaterally into the HDB to show expression throughout the full extent of the diagonal band (VDB and HDBs). e–g, Bilateral viral injections into the HDB (AAV-FLEX-Syn::EGFP shown as an example) are sufficient to target the full extent of the diagonal band (VDB and HDB). Scale bars, 100 μm. Schematic in a was adapted from images from the Allen Brain Institute Reference Atlas30.
Extended Data Figure 2 Time-dependent changes in body content and metabolic measures between non-ablated and DBB-ablated mice.
a, Oxygen consumption, represented as VO2, at various stages post DBB-ablation. Data are represented as mean ± s.e.m. *P < 0.05 by two-sided, unpaired Student’s t-test. b, c, Average total lean mass (b) or body fat (c) content at various stages post DBB-ablation. Data are represented as mean ± s.e.m. *P < 0.05 by two-sided, unpaired Student’s t-test. d–f, Blood cholesterol (d), leptin (e), insulin (f), and blood glucose concentration (g) at various stages post DBB-ablation. Data are represented as mean ± s.e.m. *P < 0.05 by two-sided, unpaired Student’s t-test. h, i, Representative non-ablated (h) or DBB-ablated (i) animals (n = 3 mice) dissected to show abdominal fat pads and enlarged fatty livers. j, k, Representative haematoxylin and eosin (H&E) stains showing fat deposition of the liver in non-ablated (j) or DBB-ablated (k) animals. Scale bar, 100 μm.
Extended Data Figure 3 Cholinergic DBB neurons are VGAT-positive.
a–c, Vgat-cre+/− mouse injected with AAV-FLEX-tdTomato into the HDB and stained with anti-ChAT antibody (green channel). Data suggest a high percentage of co-localization between VGAT-positive neurons and ChAT. Scale bar, 100 μm.
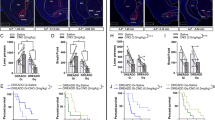
Extended Data Figure 4 hM4D-mediated inhibition of the cholinergic DBB increases food intake.
a, Whole-cell electrophysiological recordings from a representative cholinergic neuron expressing the hM4D inhibitory DREADD receptor before (top) and after (bottom) CNO treatment. b, Total two-hour food intake after an overnight fast (n = 11 mice). ‘Saline’ and ‘CNO’ groups represent separate trials of the same animals in the absence or presence of CNO, respectively. Data are represented as mean ± s.e.m. *P < 0.05 by two-sided, paired Student’s t-test.
Extended Data Figure 5 Representative cholinergic DBB-innervating projection sites in the brain.
a, b, Schematic (a) and representative image (b) showing injection site in the HDBs of a Chat-cre+/− mouse with AAV-FLEX-Syn::EGFP. Scale bar, 1 mm. c–e, Chat-cre+/− mouse injected bilaterally into the HDB with AAV-FLEX-Syn::EGFP and stained with an anti-ChAT antibody. Staining shows high co-localization of ChAT-positive neurons with the Cre-dependent AAV, demonstrating the efficacy of the Chat-cre mouse line. Of the neurons infected with virus, only a small fraction do not express ChAT. Scale bars, 100 μm (c–e) and 50 μm (c′–e′). f–h, Cholinergic projections from the DBB can be validated by known projection sites including the olfactory bulb. Scale bar, 200 μm; GL, glomerular layer; EPL, external plexiform layer; MCL, mitral cell layer; GCL, granule cell layer. i–k, Hippocampus. Scale bar, 200 μm. l–n, Amygdala (basolateral amygdala shown here). Scale bar, 100 μm. o–u, Also shown are areas of the ventral hypothalamus including, Median eminence (o–q), and arcuate nucleus (r–u). Scale bars, 100 μm. Schematic in a was adapted from an image from the Allen Brain Institute Reference Atlas30.
Extended Data Figure 6 Pomc expression is reduced in pair-fed DBB-ablated animals.
a, Relative Agrp and Pomc expression levels in arcuate nuclei between pair-fed non-ablated and DBB-ablated mice (n = 4 mice per group). Data are represented as mean ± s.e.m. *P < 0.05 by two-sided, unpaired Student’s t-test. b, Acetylcholine does not significantly alter arcuate NPY neuron firing. Data shown as mean ± s.e.m. of spike frequency in 8 acetylcholine-treated cells (1 recorded neuron per slice, 1–2 slices per mouse, n = 6 mice), comparisons made versus baseline values (1–4 min) by repeated measures ANOVA with Holm–Sidak multiple comparison. c, d, Acetylcholine significantly increases arcuate POMC neuron firing. Data shown as mean ± s.e.m. of spike frequency in 7 acetylcholine-treated cells (1 recorded neuron per slice, 1–2 slices per mouse, n = 4 mice), *P < 0.001 versus baseline values (1–4 min) by repeated measures ANOVA with Holm–Sidak multiple comparison. e, Representative voltage clamp recording from POMC-EGFP neuron after local administration of acetylcholine and after pharmacological manipulation of fast synaptic transmission, as well as nicotinic acetylcholine receptor blockade (20-s sweep with an inter-trial interval of 1 min and repeated for 5 sweeps each for baseline, synaptic blockers (CNQX, 2-amino-5-phosphonopentanoic acid (APV), GABAzine and tetrodotoxin (TTX)), and nicotinic blockers (mecamylamine (MEC), methyllycaconitine (MLA) and dihydro-β-erythroidine hydrobromide (DBHE)).
Extended Data Figure 7 Diffuse cholinergic DBB projections into the hypothalamus.
a, Site of AAV-FLEX-Syn::mRuby2 injections in the DBB. b–d, Representative images of cholinergic terminals (red) from the DBB into the hypothalamus. Scale bar, 100 μm. ME, median eminence. b′–d′, Higher magnification images from the section shown in the dotted squares in b–d. Scale bar, 50 μm. Closed arrowheads indicate close apposition or co-localization between cholinergic terminals and POMC (green) cell bodies, whereas open arrowheads show apposition or co-localization on or near POMC neuronal processes. Of note, cholinergic terminals also appear on non-POMC neurons. e, f, DAPI (e), anti-β-endorphin (f) (POMC neuron marker), g, h, AAV-FLEX-Syn::mRuby2 (injected into the DBB) (g), and merged (h) channels from f and g show innervation of the hypothalamus from cholinergic neurons in the DBB. Scale bar, 200 μm. Schematic in a was adapted from an image from the Allen Brain Institute Reference Atlas30.
Extended Data Figure 8 ChR2-mediated decrease in feeding is partially suppressed by nAChR blockade.
a, b, Total (a) and normalized (with respect to the mean ‘No-stim’ value; b) food intake after a two-hour re-feeding after an overnight fast under specified conditions. Comparisons between ‘No-Stim,’ ‘Stim,’ ‘No-Stim + Mec,’ and ‘Stim + Mec’ refer to separate trials conducted on the same animals (n = 5 mice). Data are represented as mean ± s.e.m. *P < 0.05 by two-sided, paired Student’s t-test.
Extended Data Figure 9 Arcuate acetylcholine receptor (AChR) expression profile.
a, Region-specific transcript expression profile of nicotinic and muscarinic AChRs in the arcuate nucleus, relative to the housekeeping gene, Gapdh (n = 4 mice). Data are represented as mean ± s.e.m.
Rights and permissions
About this article
Cite this article
Herman, A., Ortiz-Guzman, J., Kochukov, M. et al. A cholinergic basal forebrain feeding circuit modulates appetite suppression. Nature 538, 253–256 (2016). https://doi.org/10.1038/nature19789
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature19789
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.