Abstract
Morphine is an alkaloid from the opium poppy used to treat pain. The potentially lethal side effects of morphine and related opioids—which include fatal respiratory depression—are thought to be mediated by μ-opioid-receptor (μOR) signalling through the β-arrestin pathway or by actions at other receptors. Conversely, G-protein μOR signalling is thought to confer analgesia. Here we computationally dock over 3 million molecules against the μOR structure and identify new scaffolds unrelated to known opioids. Structure-based optimization yields PZM21—a potent Gi activator with exceptional selectivity for μOR and minimal β-arrestin-2 recruitment. Unlike morphine, PZM21 is more efficacious for the affective component of analgesia versus the reflexive component and is devoid of both respiratory depression and morphine-like reinforcing activity in mice at equi-analgesic doses. PZM21 thus serves as both a probe to disentangle μOR signalling and a therapeutic lead that is devoid of many of the side effects of current opioids.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lord, J. A. H., Waterfield, A. A., Hughes, J. & Kosterlitz, H. W. Endogenous opioid peptides: multiple agonists and receptors. Nature 267, 495–499 (1977)
Martin, W. R., Eades, C. G., Thompson, J. A., Huppler, R. E. & Gilbert, P. E. The effects of morphine- and nalorphine-like drugs in the nondependent and morphine-dependent chronic spinal dog. J. Pharmacol. Exp. Ther . 197, 517–532 (1976)
Hughes, J. et al. Identification of two related pentapeptides from the brain with potent opiate agonist activity. Nature 258, 577–580 (1975)
Bohn, L. M., Gainetdinov, R. R., Lin, F.-T., Lefkowitz, R. J. & Caron, M. G. μ-opioid receptor desensitization by β-arrestin-2 determines morphine tolerance but not dependence. Nature 408, 720–723 (2000)
Bohn, L. M. et al. Enhanced morphine analgesia in mice lacking β-arrestin 2. Science 286, 2495–2498 (1999)
Raehal, K. M., Walker, J. K. & Bohn, L. M. Morphine side effects in β-arrestin 2 knockout mice. J. Pharmacol. Exp. Ther. 314, 1195–1201 (2005)
DeWire, S. M. et al. A G protein-biased ligand at the μ-opioid receptor is potently analgesic with reduced gastrointestinal and respiratory dysfunction compared with morphine. J. Pharmacol. Exp. Ther. 344, 708–717 (2013)
Soergel, D. G. et al. Biased agonism of the μ-opioid receptor by TRV130 increases analgesia and reduces on-target adverse effects versus morphine: a randomized, double-blind, placebo-controlled, crossover study in healthy volunteers. Pain 155, 1829–1835 (2014)
Manglik, A. et al. Crystal structure of the μ-opioid receptor bound to a morphinan antagonist. Nature 485, 321–326 (2012)
Granier, S. et al. Structure of the δ-opioid receptor bound to naltrindole. Nature 485, 400–404 (2012)
Wu, H. et al. Structure of the human κ-opioid receptor in complex with JDTic. Nature 485, 327–332 (2012)
Thompson, A. A. et al. Structure of the nociceptin/orphanin FQ receptor in complex with a peptide mimetic. Nature 485, 395–399 (2012)
Carlsson, J. et al. Ligand discovery from a dopamine D3 receptor homology model and crystal structure. Nat. Chem. Biol. 7, 769–778 (2011)
de Graaf, C. et al. Crystal structure-based virtual screening for fragment-like ligands of the human histamine H1 receptor. J. Med. Chem. 54, 8195–8206 (2011)
Katritch, V. et al. Structure-based discovery of novel chemotypes for adenosine A2A receptor antagonists. J. Med. Chem. 53, 1799–1809 (2010)
Kolb, P. et al. Structure-based discovery of β2-adrenergic receptor ligands. Proc. Natl Acad. Sci. USA 106, 6843–6848 (2009)
Langmead, C. J. et al. Identification of novel adenosine A2A receptor antagonists by virtual screening. J. Med. Chem. 55, 1904–1909 (2012)
Powers, R. A., Morandi, F. & Shoichet, B. K. Structure-based discovery of a novel, noncovalent inhibitor of AmpC β-lactamase. Structure 10, 1013–1023 (2002)
Huang, X. P. et al. Allosteric ligands for the pharmacologically dark receptors GPR68 and GPR65. Nature 527, 477–483 (2015)
Irwin, J. J., Sterling, T., Mysinger, M. M., Bolstad, E. S. & Coleman, R. G. ZINC: a free tool to discover chemistry for biology. J. Chem. Inf. Model. 52, 1757–1768 (2012)
Mysinger, M. M. & Shoichet, B. K. Rapid context-dependent ligand desolvation in molecular docking. J. Chem. Inf. Model. 50, 1561–1573 (2010)
Mysinger, M. M. et al. Structure-based ligand discovery for the protein–protein interface of chemokine receptor CXCR4. Proc. Natl Acad. Sci. USA 109, 5517–5522 (2012)
Negri, A. et al. Discovery of a novel selective kappa-opioid receptor agonist using crystal structure-based virtual screening. J. Chem. Inf. Model. 53, 521–526 (2013)
Ballesteros, J. A. & Weinstein, H. Integrated methods for the construction of three-dimensional models and computational probing of structure-function relations in G protein-coupled receptors. Methods in Neurosciences 25, 366–428 (1995)
Gaulton, A. et al. ChEMBL: a large-scale bioactivity database for drug discovery. Nucleic Acids Res . 40, D1100–D1107 (2012)
Muchmore, S. W. et al. Application of belief theory to similarity data fusion for use in analog searching and lead hopping. J. Chem. Inf. Model. 48, 941–948 (2008)
Pasternak, G. W. & Pan, Y.-X. μ opioids and their receptors: evolution of a concept. Pharmacol. Rev. 65, 1257–1317 (2013)
Fenalti, G. et al. Structural basis for bifunctional peptide recognition at human δ-opioid receptor. Nat. Struct. Mol. Biol. 22, 265–268 (2015)
Huang, W. et al. Structural insights into μ-opioid receptor activation. Nature 524, 315–321 (2015)
Kroeze, W. K. et al. PRESTO-Tango as an open-source resource for interrogation of the druggable human GPCRome. Nat. Struct. Mol. Biol. 22, 362–369 (2015)
Kenakin, T. & Christopoulos, A. Signalling bias in new drug discovery: detection, quantification and therapeutic impact. Nat. Rev. Drug Discov. 12, 205–216 (2013)
Nickolls, S. A., Humphreys, S., Clark, M. & McMurray, G. Co-expression of GRK2 reveals a novel conformational state of the μ-opioid receptor. PLoS One 8, e83691 (2013)
Groer, C. E. et al. An opioid agonist that does not induce μ-opioid receptor–arrestin interactions or receptor internalization. Mol. Pharmacol. 71, 549–557 (2007)
Le Bars, D., Gozariu, M. & Cadden, S. W . Animal models of nociception. Pharmacol. Rev . 53, 597–652 (2001)
Li, C. et al. μ opioid receptor modulation of dopamine neurons in the periaqueductal gray/dorsal raphe: a role in regulation of pain. Neuropsychopharmacology 41, 2122–2132 (2016)
Han, S., Soleiman, M. T., Soden, M. E., Zweifel, L. S. & Palmiter, R. D. Elucidating an affective pain circuit that creates a threat memory. Cell 162, 363–374 (2015)
Gogas, K. R., Presley, R. W., Levine, J. D. & Basbaum, A. I. The antinociceptive action of supraspinal opioids results from an increase in descending inhibitory control: correlation of nociceptive behavior and c-fos expression. Neuroscience 42, 617–628 (1991)
Montandon, G. et al. G-protein-gated inwardly rectifying potassium channels modulate respiratory depression by opioids. Anesthesiology 124, 641–650 (2016)
Spanagel, R., Herz, A. & Shippenberg, T. S. Opposing tonically active endogenous opioid systems modulate the mesolimbic dopaminergic pathway. Proc. Natl Acad. Sci. USA 89, 2046–2050 (1992)
Bohn, L. M. et al. Enhanced rewarding properties of morphine, but not cocaine, in β(arrestin)-2 knock-out mice. J. Neurosci . 23, 10265–10273 (2003)
Tzschentke, T. M. Measuring reward with the conditioned place preference paradigm: a comprehensive review of drug effects, recent progress and new issues. Prog. Neurobiol. 56, 613–672 (1998)
Weiss, D. R. et al. Conformation guides molecular efficacy in docking screens of activated β-2 adrenergic G protein coupled receptor. ACS Chem. Biol. 8, 1018–1026 (2013)
Carlsson, J. et al. Structure-based discovery of A2A adenosine receptor ligands. J. Med. Chem. 53, 3748–3755 (2010)
Irwin, J. J. et al. Automated docking screens: a feasibility study. J. Med. Chem. 52, 5712–5720 (2009)
Besnard, J. et al. Automated design of ligands to polypharmacological profiles. Nature 492, 215–220 (2012)
Weichert, D. et al. Covalent agonists for studying G protein-coupled receptor activation. Proc. Natl Acad. Sci. USA 111, 10744–10748 (2014)
Möller, D. et al. Functionally selective dopamine D2, D3 receptor partial agonists. J. Med. Chem. 57, 4861–4875 (2014)
Hübner, H., Haubmann, C., Utz, W. & Gmeiner, P. Conjugated enynes as nonaromatic catechol bioisosteres: synthesis, binding experiments, and computational studies of novel dopamine receptor agonists recognizing preferentially the D3 subtype. J. Med. Chem. 43, 756–762 (2000)
Lane, J. R., Powney, B., Wise, A., Rees, S. & Milligan, G. G protein coupling and ligand selectivity of the D2L and D3 dopamine receptors. J. Pharmacol. Exp. Ther. 325, 319–330 (2008)
Jiang, L. I. et al. Use of a cAMP BRET sensor to characterize a novel regulation of cAMP by the sphingosine 1-phosphate/G13 pathway. J. Biol. Chem. 282, 10576–10584 (2007)
Nakajima, K.-i., Gimenez, L. D., Gurevich, V. & Wess, J. in Designer Receptors Exclusively Activated by Designer Drugs Vol. 108 Neuromethods (ed Thiel, G. ) Ch. 2, 29–48 (Springer New York, 2015)
Rajagopal, S. et al. Quantifying ligand bias at seven-transmembrane receptors. Mol. Pharmacol. 80, 367–377 (2011)
Rajagopal, S. Quantifying biased agonism: understanding the links between affinity and efficacy. Nat. Rev. Drug Discov. 12, 483 (2013)
Huang, X.-P., Mangano, T., Hufeisen, S., Setola, V. & Roth, B. L. Identification of human Ether-à-go-go related gene modulators by three screening platforms in an academic drug-discovery setting. Assay Drug Dev. Technol. 8, 727–742 (2010)
Balter, R. E. & Dykstra, L. A. Thermal sensitivity as a measure of spontaneous morphine withdrawal in mice. J. Pharmacol. Toxicol. Methods 67, 162–168 (2013)
Sorge, R. E. et al. Olfactory exposure to males, including men, causes stress and related analgesia in rodents. Nat. Methods 11, 629–632 (2014)
Woolf, C. J. Long term alterations in the excitability of the flexion reflex produced by peripheral tissue injury in the chronic decerebrate rat. Pain 18, 325–343 (1984)
Blanchard, R. J. & Blanchard, D. C. Passive and active reactions to fear-eliciting stimuli. J. Comp. Physiol. Psychol. 68, 129–135 (1969)
Bolles, R. C. Species-specific defense reactions and avoidance learning. Psychol. Rev. 77, 32 (1970)
Bolles, R. C. & Fanselow, M. S. A perceptual-defensive-recuperative model of fear and pain. Behav. Brain Sci. 3, 291–301 (1980)
Estes, W. K. Discriminative conditioning; effects of a Pavlovian conditioned stimulus upon a subsequently established operant response. J. Exp. Psychol. 38, 173–177 (1948)
Estes, W. K. & Skinner, B. F. Some quantitative properties of anxiety. J. Exp. Psychol. 29, 390 (1941)
Rescorla, R. A. & Lolordo, V. M. Inhibition of avoidance behavior. J. Comp. Physiol. Psychol. 59, 406–412 (1965)
Skinner, B. F. The behavior of organisms; an experimental analysis . (D. Appleton-Century Company, Incorporated, 1938)
Hunskaar, S. & Hole, K. The formalin test in mice: dissociation between inflammatory and non-inflammatory pain. Pain 30, 103–114 (1987)
Clougherty, J. E. et al. Chronic social stress and susceptibility to concentrated ambient fine particles in rats. Environ. Health Perspect. 118, 769–775 (2010)
Sanberg, P. R., Bunsey, M. D., Giordano, M. & Norman, A. B. The catalepsy test: its ups and downs. Behav. Neurosci. 102, 748–759 (1988)
Hiller, C. et al. Functionally selective dopamine D2/D3 receptor agonists comprising an enyne moiety. J. Med. Chem. 56, 5130–5141 (2013)
Case, D. et al. AMBER 15. San Francisco, CA: University of California (2015)
Goetz, A., Lanig, H., Gmeiner, P. & Clark, T. Molecular dynamics simulations of the effect of the G-protein and diffusible ligands on the β2-adrenergic receptor. J. Mol. Biol. 414, 611–623 (2011)
Wang, J., Wolf, R. M., Caldwell, J. W., Kollman, P. A. & Case, D. A. Development and testing of a general amber force field. J. Comput. Chem. 25, 1157–1174 (2004)
Hornak, V. et al. Comparison of multiple amber force fields and development of improved protein backbone parameters. Proteins 65, 712–725 (2006)
Bayly, C. I., Cieplak, P., Cornell, W. & Kollman, P. A. A well-behaved electrostatic potential based method using charge restraints for deriving atomic charges: the RESP model. J. Phys. Chem. 97, 10269–10280 (1993)
Berendsen, H., Grigera, J. & Straatsma, T. The missing term in effective pair potentials. J. Phys. Chem. 91, 6269–6271 (1987)
Kissin, I., Brown, P. T., Robinson, C. A. & Bradley, E. L. Acute tolerance in morphine analgesia continuous infusion and single injection in rats. Anesthesiology 74, 166–171 (1991)
Acknowledgements
Supported by the US National Institutes of Health grants GM106990 (B.K.K., B.K.S. and P.G.), DA036246 (B.K.K.), GM59957 (B.K.S.), and the National Institutes of Mental Health Psychoactive Drug Screening Program (B.L.R.) and DA017204 (B.L.R., D.A.), DA035764 (B.L.R.) and the Michael Hooker Distinguished Professorship (B.L.R.) and the German Research Foundation Grants Gm 13/10 and GRK 1910 (P.G). A.M. received support from the Stanford University Medical Scientist Training Program (T32GM007365) and the American Heart Association (12PRE8120001).
Author information
Authors and Affiliations
Contributions
A.M. and H.L. initiated the project. H.L. performed docking and identified compounds to be tested in the initial and analogue screens. A.M. performed binding studies to identify initial hits and devised structure-guided optimization strategies for subsequent analogues. D.K.A. performed in vivo studies, including analgesia assays, mouse plethysmography, faecal boli accumulation studies, open field locomotor assay, and conditioned place preference. J.D.M., M.F.S. and P.M.G. performed radioligand binding and signalling studies. X.P.H. performed signalling studies and assessed compound activity against the GPCRome. D.De., V.B., S.L. and H.H. synthesized compounds and determined affinities by radioligand binding and performed signalling studies. A.L. and A.M. docked PZM21 and TRV130 and R.C.K. simulated PZM21 binding to μOR. G.C. performed reflexive and affective analgesia studies of μOR knockout mice and was supervised by G.S. D.Du. performed pharmacokinetic studies. The manuscript was written by A.M., H.L. and B.K.S. with editing and suggestions from B.L.R. and input from D.K.A., B.K.K. and P.G. P.G. supervised chemical synthesis of compounds and the separation and identification of diastereomers, B.K.K. supervised testing of initial docking hits, B.L.R. supervised radioligand binding, signalling and in vivo studies and B.K.S. supervised the compound discovery and design. The project was conceived by A.M., H.L., B.K.K., P.G., B.K.S and B.L.R.
Corresponding authors
Ethics declarations
Competing interests
A.M., H.L., P.G., D.D., B.K.K., B.L.R. and B.K.S. have filed a provisional patent on PZM21 and related molecules. A.M., P.G., B.K.K., B.L.R. and B.K.S. are consultants and co-founders of Epiodyne, a company seeking to develop novel analgesics.
Additional information
Reviewer Information Nature thanks G. Henderson, E. Kelly, B. Kieffer and J. Meiler for their contribution to the peer review of this work.
Extended data figures and tables
Extended Data Figure 1 Docking poses of active compounds.
Seven of 23 experimentally tested compounds bound to the μOR with micromolar affinity. Their docked poses often occupy sites not exploited by the antagonist β-funaltrexamine. In each case, a canonical ionic interaction with D1473.32 is observed.
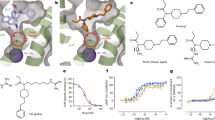
Extended Data Figure 2 Stereochemical structure-activity relationship.
a, As with the different stereoisomers of 12, variation of the chiral centres in compound PZM21 results in large changes in efficacy and potency. Data are mean ± s.e.m. of normalized results (n = 3 measurements). b, Structure–activity relationship of compound 12 and 21 stereoisomers with affinities displayed as pKi values and agonist potency and efficacy in a Gi/o Glosensor assay. c, d, PZM21 docked to active μOR shows a more extended conformation as compared to the inactive state. e, In the docked active state, the PZM21 thiophene extends into the specificity-determining region of opioid receptors. Key interacting residues here are highlighted as red lines and corresponding residues at the other human opioid receptors are indicated. f, Docked pose of TRV130 within the μOR site, showing minimal overlap in key pharmacophores with PZM21 besides the ionic interaction between the cationic amine and D1473.32. g, Molecular dynamics simulations of PZM21 in the inactive μOR state (grey and black traces) leads to a stable conformation with the thiophene positioned >10 Å away from N1272.63 (total of 2 μs of simulation time over three independent trajectories). In contrast, PZM21 adopts a more extended pose when simulated with active μOR, with an average distance of 6 Å between the thiophene and N1272.63. Other key interactions between μOR and PZM21 are also highlighted.
Extended Data Figure 3 Structure activity relationship defined by PZM21 analogues.
Eight analogues were synthesized to probe the binding orientation of PZM21 and their efficacy as agonists was tested in a CAMYEL-based Gi/o signalling assay. Analogues were compared to a parent reference compound (PZM22) with similar efficacy and potency to PZM21. In each case, the EC50 value for PZM22 is shown in black (1.8 nM) and the EC50 for the analogue is coloured. The covalent compound PZM29 binds to the μOR:N127C variant irreversibly, as evidenced by wash-resistant inhibition of radioligand binding. Signalling data are mean ± s.e.m. of normalized results (n = 3 measurements).
Extended Data Figure 4 Signalling properties of PZM21 at the opioid receptors.
Displayed are raw luminescence data from a Gi/o Glosensor assay. In agonist mode, agonists decrease luminescence while inverse agonists increase it by diminishing basal signalling. For each opioid receptor, a prototypical well-characterized agonist (black curves) and antagonist (red curves) were used to validate the assay. In antagonist mode, a competition reaction is performed with 50 nM agonist and an escalating amount of tested drug. Here, true antagonists increase the observed signal, consistent with their ability to compete with the agonist but not induce Gi signalling. Data are mean ± s.e.m. of non-normalized results (n = 3 measurements).
Extended Data Figure 5 PZM21 is selective for μOR.
a, Compound PZM21 was screened against 320 non-olfactory GPCRs for agonism in the arrestin recruitment TANGO assay. Each point shows luminescence normalized to basal level at a given GPCR, with vertical lines indicating the standard error of the mean. b, GPCRs for which PZM21 induces an increase in signal twofold over background were further tested in full dose–response mode. Several potential targets (GPR110, MCHR1R, PTGER1) did not show dose-dependent increase in signal and probably represent screening false positives. CXCR7 and SSTR4 did show dose-dependent signals at high concentrations of PZM21, and were further tested in non-arrestin signalling assays. c, PZM21 does not show a dose-dependent change in cAMP inhibition in a Gi/o Glosensor assay measuring SSTR4 activation, indicating that the single elevated point in b is probably a false positive result. d, e, Inhibition assays of hERG (d) and the dopamine transporter (DAT), norepinephrine transporter (NET), and serotonin transporter (SERT) (e) show that PZM21 has weak inhibitory activity ranging from 2–34 μM at these targets. For a, data are mean ± s.e.m. of non-normalized results (n = 4 measurements). For b–e, data are mean ± s.e.m. of normalized results (n = 3–6 measurements).
Extended Data Figure 6 Signalling profile of PZM21 and other μOR agonists.
a, PZM21 is an efficacious Gi and Go agonist in a GTPγS assay. b, Like other μOR agonists, PZM21 induces a dose-dependent decrease in cAMP levels that is sensitive to pertussis toxin, confirming Gi/o mediated signalling. c, Herkinorin is a Gi/o agonist and robustly recruits arrestin in a BRET assay performed in the absence of GRK2 overexpression. TRV130 or PZM21 show undetectable levels of arrestin recruitment in the same experiement. d, PZM21 and other opioids show no activity in a calcium-release assay, indicating no Gq-mediated second messenger signalling. The positive control TFLLR-NH2 efficiently activates the Gq coupled receptor PAR-1. e, PZM21 and TRV130 induce much decreased receptor internalization versus DAMGO and even morphine. f, Herkinorin and TRV130 are potent agonists of the κOR. PZM21 is a κOR antagonist (see Extended Data Fig. 4). g, In HEK293 cells, GRK2 expression levels have minimal effect on the potency and efficacy of the unbiased agonist DAMGO in a Gi/o activation assay. Increased GRK2 levels change the basal cAMP signal due to increased desentization of μOR, which lowers receptor basal activity and leads to elevated isoproterenol-induced cAMP. In an arrestin-recruitment BRET assay, increased GRK2 expression increases both the potency and maximal efficacy of the unbiased agonist DAMGO. This is likely because GRK2 mediated phosphorylation is required for efficient β-arrestin recruitment. h, Gi activation and arrestin recruitment in cells co-expressing 1.0 μg/15 cm2 of GRK2. Notably, PZM21 induces a higher maximal level of arrestin recruitment as compared to U2OS cells, which express very low levels of GRK2, but this level is significantly lower than morphine. Despite the lower efficacy for arrestin recruitment observed for morphine, TRV130 and PZM21 compared to DAMGO, a formal calculation of bias by the operational models fails to show that this effect is significant. i, Table of pEC50 values and Emax values for various signalling assays. All data are mean ± s.e.m. of results (n = 2–6 measurements).
Extended Data Figure 7 Additional in vivo studies of PZM21.
a, Analgesic responses measured in the hotplate assay were subcategorized into either affective or reflexive behaviours and scored separately. b, Morphine (n = 10 animals) induces changes in both behaviours, while PZM21 (n = 13 animals) only modulates the attending (affective) component. Knockout of the μOR ablates all analgesic responses by morphine and PZM21. c, PZM21 shows minimal cataleptic effect compared to morphine at different time points. The effect of haloperidol was included as a positive control. d, Pharmacokinetic studies of PZM21 (n = 3–4 animals for each time point) show central nervous system penetration of the compound, with a peak level of 197 ng of PZM21 per g of brain tissue. With a concomitant serum concentration of 1,253 ng/ml, this represents a serum:brain concentration ratio of 6.4. These levels are similar to those achieved by morphine, which shows a peak brain concentration of approximately 300 ng/g and a serum:brain concentration ratio of 3.7 30 min after subcutaneous injection75. e, Metabolism of PZM21 over 60 min exposure to mouse liver microsomes. Rotigotine and imipramine serve as positive controls for extensive phase I metabolism. The total amount of PZM21 and metabolite pool is slightly greater than 100% (101.8%) reflecting cumulative error in LC/MS analysis. f, A Gi/o signalling assay shows that none of the metabolites are measurably more potent activators of the μOR versus PZM21 alone. The metabolite pool after the 60-min incubation was used directly in the signalling assay. As a negative control, the pooled material from a reaction carried out in the absence of the key cofactor NADPH was used in the signalling assay. All data are mean ± s.e.m. For e, reactions were run in triplicate and the s.e.m. was calculated from individual measurements of each reaction.
Supplementary information
Supplementary Information
This file contains Supplementary Text and Data, Supplementary References and a Supplementary Table. (PDF 1531 kb)
Rights and permissions
About this article
Cite this article
Manglik, A., Lin, H., Aryal, D. et al. Structure-based discovery of opioid analgesics with reduced side effects. Nature 537, 185–190 (2016). https://doi.org/10.1038/nature19112
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature19112
This article is cited by
-
Ligand efficacy modulates conformational dynamics of the µ-opioid receptor
Nature (2024)
-
Profiling the proximal proteome of the activated μ-opioid receptor
Nature Chemical Biology (2024)
-
Structure, function and drug discovery of GPCR signaling
Molecular Biomedicine (2023)
-
Essential role of P-glycoprotein in the mechanism of action of oliceridine
Neuropsychopharmacology (2023)
-
CYX-5, a G-protein biassed MOP receptor agonist, DOP receptor antagonist and KOP receptor agonist, evokes constipation but not respiratory depression relative to morphine in rats
Pharmacological Reports (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.