Abstract
Thirst motivates animals to drink in order to maintain fluid balance. Thirst has conventionally been viewed as a homeostatic response to changes in blood volume or tonicity1,2,3. However, most drinking behaviour is regulated too rapidly to be controlled by blood composition directly, and instead seems to anticipate homeostatic imbalances before they arise4,5,6,7,8,9,10,11. How this is achieved remains unknown. Here we reveal an unexpected role for the subfornical organ (SFO) in the anticipatory regulation of thirst in mice. By monitoring deep-brain calcium dynamics, we show that thirst-promoting SFO neurons respond to inputs from the oral cavity during eating and drinking and then integrate these inputs with information about the composition of the blood. This integration allows SFO neurons to predict how ongoing food and water consumption will alter fluid balance in the future and then to adjust behaviour pre-emptively. Complementary optogenetic manipulations show that this anticipatory modulation is necessary for drinking in several contexts. These findings provide a neural mechanism to explain longstanding behavioural observations, including the prevalence of drinking during meals10,11, the rapid satiation of thirst7,8,9, and the fact that oral cooling is thirst-quenching12,13,14.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Lind, R. W., Thunhorst, R. L. & Johnson, A. K. The subfornical organ and the integration of multiple factors in thirst. Physiol. Behav. 32, 69–74 (1984)
McKinley, M. J. & Johnson, A. K. The physiological regulation of thirst and fluid intake. News Physiol. Sci. 19, 1–6 (2004)
Bourque, C. W. Central mechanisms of osmosensation and systemic osmoregulation. Nat. Rev. Neurosci. 9, 519–531 (2008)
Berridge, K. C. Motivation concepts in behavioral neuroscience. Physiol. Behav. 81, 179–209 (2004)
Woods, S. C. & Ramsay, D. S. Homeostasis: beyond Curt Richter. Appetite 49, 388–398 (2007)
Stricker, E. M. & Hoffmann, M. L. Presystemic signals in the control of thirst, salt appetite, and vasopressin secretion. Physiol. Behav. 91, 404–412 (2007)
Thrasher, T. N., Nistal-Herrera, J. F., Keil, L. C. & Ramsay, D. J. Satiety and inhibition of vasopressin secretion after drinking in dehydrated dogs. Am. J. Physiol. 240, E394–E401 (1981)
Bellows, R. T. Time factors in drinking in dogs. Am. J. Physiol. 125, 87–97 (1939)
Adolph, E. F., Barker, J. P. & Hoy, P. A. Multiple factors in thirst. Am. J. Physiol. 178, 538–562 (1954)
Oatley, K. & Toates, F. M. The passage of food through gut of rats and its uptake of fluid. Psychonomic Science 16, 225–226 (1969)
Fitzsimons, T. J. & Lemagnen, J. Eating as a regulatory control of drinking in rat. J. Comp. Physiol. Psychol. 67, 273–283 (1969)
Deaux, E. Thirst satiation and the temperature of ingested water. Science 181, 1166–1167 (1973)
Kapatos, G. & Gold, R. M. Tongue cooling during drinking: a regulator of water intake in rats. Science 176, 685–686 (1972)
Eccles, R., Du-Plessis, L., Dommels, Y. & Wilkinson, J. E. Cold pleasure. Why we like ice drinks, ice-lollies and ice cream. Appetite 71, 357–360 (2013)
Shizgal, P. B. & Hyman, S. E. in Principles of Neural Science (eds Kandel, E. R. et al.) 5th edn, 1095–1115 (McGraw-Hill, 2013)
Egan, G. et al. Neural correlates of the emergence of consciousness of thirst. Proc. Natl Acad. Sci. USA 100, 15241–15246 (2003)
Farrell, M. J. et al. Cortical activation and lamina terminalis functional connectivity during thirst and drinking in humans. Am. J. Physiol. Regul. Integr. Comp. Physiol. 301, R623–R631 (2011)
McKinley, M. J., Denton, D. A., Oldfield, B. J., De Oliveira, L. B. & Mathai, M. L. Water intake and the neural correlates of the consciousness of thirst. Semin. Nephrol. 26, 249–257 (2006)
Oka, Y., Ye, M. & Zuker, C. S. Thirst driving and suppressing signals encoded by distinct neural populations in the brain. Nature 520, 349–352 (2015)
Grob, M., Trottier, J. F., Drolet, G. & Mouginot, D. Characterization of the neurochemical content of neuronal populations of the lamina terminalis activated by acute hydromineral challenge. Neuroscience 122, 247–257 (2003)
Simpson, J. B. & Routtenberg, A. Subfornical organ: site of drinking elicitation by angiotensin II. Science 181, 1172–1175 (1973)
Betley, J. N. et al. Neurons for hunger and thirst transmit a negative-valence teaching signal. Nature 521, 180–185 (2015)
Gunaydin, L. A. et al. Natural neural projection dynamics underlying social behavior. Cell 157, 1535–1551 (2014)
Rowland, N. E. Brain mechanisms of mammalian fluid homeostasis: insights from use of immediate early gene mapping. Neurosci. Biobehav. Rev. 23, 49–63 (1998)
Szczepanńska-Sadowska, E. & Kozłowski, S. Equipotency of hypertonic solutions of mannitol and sodium chloride in eliciting thirst in the dog. Pflugers Arch. 358, 259–264 (1975)
Fitzsimons, J. T. Drinking by rats depleted of body fluid without increase in osmotic pressure. J. Physiol. (Lond.) 159, 297–309 (1961)
Chen, Y., Lin, Y. C., Kuo, T. W. & Knight, Z. A. Sensory detection of food rapidly modulates arcuate feeding circuits. Cell 160, 829–841 (2015)
Mandelblat-Cerf, Y. et al. Arcuate hypothalamic AgRP and putative POMC neurons show opposite changes in spiking across multiple timescales. eLife 4, (2015)
Mendelson, J. & Chillag, D. Tongue cooling: a new reward for thirsty rodents. Science 170, 1418–1421 (1970)
Miselis, R. R., Shapiro, R. E. & Hand, P. J. Subfornical organ efferents to neural systems for control of body water. Science 205, 1022–1025 (1979)
Smith, R. D., Chui, A. T., Wong, P. C., Herblin, W. F. & Timmermans, P. B. M. W. M. Pharmacology of nonpeptide angiotensin II receptor antagonists. Ann. Rev. Pharmacol. Toxicol. 32, 135–165 (1992)
Ondetti, M. A., Rubin, B. & Cushman, D. W. Design of specific inhibitors of angiotensin-converting enzyme: new class of orally active antihypertensive agents. Science 196, 441–444 (1977)
Hsiao, S. & Trankina, F. Thirst-hunger interaction. I. Effects of body-fluid restoration on food and water intake in water-deprived rats. J. Comp. Physiol. Psychol. 69, 448–453 (1969)
Acknowledgements
C.A.Z. is supported by an NSF Graduate Research Fellowship (grant no. 1144247) and a UCSF Discovery Fellowship. Z.A.K. is a New York Stem Cell Foundation-Robertson Investigator and acknowledges support from the New York Stem Cell Foundation, American Diabetes Association, Rita Allen, McKnight, Sloan, Brain and Behavior Research, Klingenstein, and Program for Breakthrough Biological Research Foundations. This work was supported by an NIH New Innovator Award (DP2-DK109533), R01-DK106399, and R01-NS094781, as well as the UCSF Diabetes and Obesity Centers (U01 DK089541).
Author information
Authors and Affiliations
Contributions
C.A.Z. and Z.A.K. conceived the project and designed the experiments. C.A.Z., Y.-C.L., D.E.L., L.G., E.L.H., G.E.D. and Y.C. performed stereotaxic surgery and histology. Y.-C.L. conducted acute slice experiments. C.A.Z. and Y.C. conducted photometry experiments. C.A.Z. conducted optogenetics experiments. C.A.Z. and D.E.L. conducted plasma composition experiments. Y.C. generated the synaptophysin-GCaMP6s construct. C.A.Z., Y.-C.L., Y.C. and Z.A.K. analysed the data. C.A.Z. and Z.A.K. prepared the manuscript with input from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer Information Nature thanks M. Krashes, M. McKinley and R. Palmiter for their contribution to the peer review of this work.
Extended data figures and tables
Extended Data Figure 1 Optogenetic activation of SFONos1 neurons is sufficient to promote drinking, but negative feedback inhibits excessive drinking during optogenetically and dehydration-induced drinking.
a–l, Optogenetic activation of SFONos1 neurons rapidly and specifically promotes drinking. a, Expression of mCherry in SFONos1 neurons from AAV5-EF1α-DIO-ChETATC-2A-mCherry. Scale bar, 100 μm. b, Representative recording showing rapid firing of SFONos1 neuron in response to photostimulation (20 Hz) in acute slice (1 out of 3 cells; blue lines, stimulation). c, Schematic of optogenetic set up for activating SFONos1 neurons. d, Rasters of drinking in response to optogenetic stimulation for seven trials each for four SFONos1::ChETATC mice (black lines, licks; blue boxes, stimulation). e, Averaged traces showing lick rate (n = 6 SFONos1::mCherry mice and 8 SFONos1::ChETATC mice). f, Averaged traces showing cumulative licks (n = 6 SFONos1::mCherry mice and 8 SFONos1::ChETATC mice). g, Quantification of drinking during stimulation protocol (****P < 0.0001, two-way repeated-measures ANOVA, n = 6 SFONos1::mCherry mice and 8 SFONos1::ChETATC mice). h, Licks during stimulation period across seven consecutive trials (not significant (n.s.), one-way repeated-measures ANOVA, n = 8 mice). i, Latency to first lick during stimulation period across seven consecutive trials (n.s., one-way repeated-measures ANOVA, n = 8 mice). j, Heat maps showing location of SFONos1::ChETATC mice during stimulation protocol (n = 4 mice). k, Quantification of time spent at lickometer during stimulation protocol (**P < 0.01, one-way repeated-measures ANOVA, n = 4 mice). l, Activation of SFONos1 neurons did not induce feeding (n.s., two-way repeated-measures ANOVA, n = 6 SFONos1::mCherry mice and 4 SFONos1::ChETATC mice). Panels m–q show that osmotic dilution does not inhibit excessive drinking during optogenetically induced drinking. m, SFONos1::ChETATC mice were stimulated, but with access to 150 mM NaCl instead of water. n, Lick rate (n = 8 mice). o, Cumulative licks (n = 8 mice). p, Comparison to stimulation with water access (n = 8 mice). q, Quantification (n.s., two-tailed Student’s t-test, n = 8 mice). Panels r–v show that channelrhodopsin failure does not explain the negative feedback that inhibits excessive drinking during optogenetically induced drinking. r, SFONos1::ChETATC mice were stimulated, but with delayed access to water instead of immediate access. s, Lick rate (n = 4 mice). t, Cumulative licks (n = 4 mice). u, Comparison to stimulation with water access (n = 4 mice). v, Quantification (n.s., two-tailed Student’s t-test, n = 4 mice). Panels w–af show that a negative feedback mechanism also inhibits excessive drinking during dehydration-induced drinking. w, Comparison of optogenetically and dehydration-induced drinking in SFONos1::ChETATC mice (n = 4 mice; P value colour bar represents independent two-tailed Student’s t-tests). x, Latency to first lick (n.s., two-tailed Student’s t-test, n = 4 mice). y, Cumulative licks (*P < 0.05, two-way repeated-measures ANOVA, n = 4 mice). z, Cumulative probability distribution for inter-lick interval, a measure of licking ‘speed’ (n = 4 mice). aa, Median inter-lick interval (**P < 0.01, two-tailed Student’s t-test, n = 4 mice). ab, Time constant (τ) for cumulative licks (*P < 0.05, two-tailed Student’s t-test, n = 4 mice). ac, Number of drinking bouts (**P < 0.01, two-way repeated-measures ANOVA, n = 4 mice). ad, Number of licks per drinking bout (**P < 0.01, two-way repeated-measures ANOVA, n = 4 mice). ae, Bout duration (**P < 0.01, two-way repeated-measures ANOVA, n = 4 mice). af, Inter-bout interval (n.s., two-way repeated-measures ANOVA, n = 4 mice). Values are mean ± s.e.m. (error bars or shaded area).
Extended Data Figure 2 GCaMP6s accurately reports SFONos1 neuron activity in acute slices.
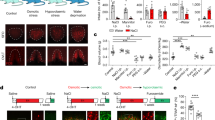
a, Expression of GCaMP6s in SFONos1 neurons from AAV5-EF1α-FLEX-GCaMP6s. Scale bar, 100 μm. b, Representative fluorescence images of a neuron given a 30 pA current injection for 700 m in acute slice (1 out of 9 cells). c, Representative traces showing calcium responses in response to 30 pA current injections of increasing duration to produce increasing numbers of action potentials (1 out of 9 cells). d, Relationship between number of action potentials and ΔF/F for the representative neuron in c (shaded area denotes 95% confidence interval). e, R2 and P value for linear relationship between number of action potentials and ΔF/F (n = 9 cells). Panels f–j show that SFONos1 neurons are homogeneously responsive to both angiotensin and salt challenge. f, Representative fluorescence images showing SFONos1 neuron activity before and during bath application of angiotensin (1 out of 3 experiments; red circles, identified neurons). g, 24 out of 27 (~90%) identified SFONos1 neurons were activated by bath application of angiotensin (red line, mean; grey lines, individual activated neurons). h, Quantification (****P < 0.0001, two-way repeated-measures ANOVA, n = 24 activated neurons). i, Experimental design to test whether a single population of SFO neurons is responsive to both angiotensin and salt challenge. j, Co-localization of Agtr1α::GFP and salt-challenge-induced cFos indicates that SFONos1 neurons are homogeneously responsive to both angiotensin and salt challenge. Scale bars, 100 μm. k, Experimental design to test whether a SFO neurons express the excitatory neuron marker CaMK2α. l, Co-localization of CaMK2α::mCherry and Nos1::GFP indicates that SFONos1 neurons are excitatory. Scale bar, 100 μm. Values are mean ± s.e.m. (error bars or shaded area).
Extended Data Figure 3 Regulation of SFONos1 neurons by homeostatic signals.
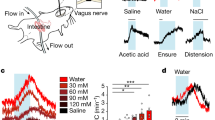
a, Recordings from SFONos1::GCaMP6s mice as they explored a behavioural chamber without access to food or water revealed dynamic fluctuations in fluorescence around a stable baseline (1 out of 8 mice); these fluctuations were absent from recordings from SFONos1::GFP mice (1 out of 3 mice). b, Quantification of response to peripheral injection of NaCl (averaged traces in Fig. 1b; **P < 0.01, ****P < 0.0001, two-way repeated-measures ANOVA, n = 5 mice). c, Time constant (τ) of rising and falling phases of response to peripheral injection of NaCl (n.s., two-way repeated-measures ANOVA, n = 5 mice). d, Representative recordings for five mice showing response to peripheral injection of NaCl or vehicle. e, SFONos1 neurons are activated by peripheral injection of angiotensin in a dose-dependent manner (n = 3 mice). f, Quantification (*P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, two-way repeated-measures ANOVA, n = 3 mice). g, The AT1R antagonist losartan abolished the response to peripheral injection of angiotensin (n = 3 mice). h, Quantification (**P < 0.01, two-way repeated-measures ANOVA, n = 3 mice). i, Schematic illustrating expected observations whether activation of SFONos1 neurons in response to peripheral injection PEG/isoproterenol and NaCl is angiotensin-dependent. j, Angiotensin blockers abolished the response to peripheral injection of PEG (quantification in Fig. 1f; n = 5 mice). k, Angiotensin blockers abolished the response to peripheral injection of isoproterenol (quantification in Fig. 1h; n = 5 mice). l, Angiotensin blockers did not abolish the response to peripheral injection of NaCl (quantification in Fig. 1c; n = 5 mice). m, Schematic illustrating expected observations if activation of SFONos1 neurons in response to peripheral injection NaCl is sodium-sensory or osmosensory. n, SFONos1 neurons are similarly activated by peripheral injection of equimolar concentrations of mannitol and NaCl (quantification in Fig. 1d; n = 5 mice). Values are mean ± s.e.m. (error bars or shaded area).
Extended Data Figure 4 SFONos1 neurons are necessary for drinking.
a, Expression of yellow fluorescent protein (YFP) in SFONos1 neurons from AAV5-EF1α-DIO-eArch3.0-YFP. Scale bar, 100 μm. b, Representative recording showing firing of SFONos1 neurons is blocked in response to photosilencing in acute slice (1 out of 3 cells; yellow line, laser). c, Averaged traces showing lick rate for experiment in Fig. 2j (n = 5 mice; green box, laser on). d, Quantification (*P < 0.05, two-way repeated-measures ANOVA, n = 5 mice). e, Averaged traces showing cumulative licks after water restriction for SFONos1::mCherry control mice (n = 5 mice; green box, laser on). f, Quantification (n.s., two-way repeated-measures ANOVA, n = 5 mice). g, Averaged traces showing lick rate after water restriction for SFONos1::mCherry control mice (n = 5 mice; green box, laser on). h, Quantification (n.s., two-way repeated-measures ANOVA, n = 5 mice). Values are mean ± s.e.m. (error bars or shaded area).
Extended Data Figure 5 Regulation of SFONos1 neurons by anticipatory signals.
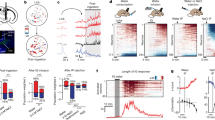
a, Representative recordings for five mice showing activation of SFONos1 neurons during salt challenge and rapid inhibition of SFONos1 neurons during drinking. b, PSTH of SFONos1 neuron activity and lick rate around the first lick in either the first drinking bout or all other drinking bouts following salt challenge (n = 5 mice). c, The decrease in SFONos1 neuron activity was greatest during the first drinking bout (ΔF/F at 20 s after first lick; **P < 0.01, two-tailed Student’s t-test, n = 5 mice). d, PSTH of SFONos1 neuron activity and lick rate around the last lick in either the first drinking bout or all other drinking bouts following salt challenge (n = 5 mice). e, Representative recording showing no inhibition of SFONos1 neurons during licking an empty bottle following salt challenge (1 out of 5 mice; red lines, licks; red boxes, drinking bouts). f, Representative recording showing rapid inhibition followed by ‘re-setting’ of SFONos1 neurons during drinking 300 mM NaCl following salt challenge (1 out of 5 mice; red lines, licks; red boxes, drinking bouts). g, SFONos1 neurons receive a post-ingestive error signal that reports the osmolarity of ingested fluids (averaged traces in Fig. 3j; ****P < 0.0001, two-way repeated-measures ANOVA, n = 5 mice). Panels h–j show that SFONos1 neurons do not transmit a teaching signal in a Pavlovian conditioning model. h, Schematic of Pavlovian conditioning model. i, SFONos1 neurons were not inhibited by cue presentation after 1 week of Pavlovian conditioning (n = 3 mice). j, PSTH of SFONos1 neuron activity and lick rate around the first lick in the first drinking bout either before or after Pavlovian conditioning (n = 3 mice). Panels k–n demonstrate that SFONos1 neurons are modulated by rapid anticipatory signals during drinking in the absence of homeostatic need. k, The activity of SFONos1 neurons was recorded while fully hydrated mice were given ad libitum access to sucrose. l, Representative recording showing modulation of SFONos1 neurons during sucrose drinking (1 out of 4 mice; red lines, licks). m, PSTH of SFONos1 neuron activity and lick rate around the first lick in all sucrose drinking bouts (n = 4 mice). n, PSTH of SFONos1 neuron activity and lick rate around the last lick in all sucrose drinking bouts (n = 4 mice). Values are mean ± s.e.m. (error bars or shaded area).
Extended Data Figure 6 Activation of SFONos1 neurons during eating does not require angiotensin signalling.
a, Experimental design to test whether angiotensin signalling is necessary for prandial thirst. b, Angiotensin blockers (INH) did not inhibit eating-induced activation of SFONos1 neurons or prandial drinking (n = 3 mice). c, Angiotensin blockers did not affect food consumption (n.s., two-tailed Student’s t-test, n = 3 mice). d, e, ARCAgRP neurons that control hunger are not reciprocally modulated by eating and drinking. d, Schematic of fibre photometry set up for recording the activity of ARCAgRP neurons. Scale bar, 100 μm. e, ARCAgRP neurons were rapidly inhibited when fasted mice were presented with chow, as previously reported27, but were unaffected when dehydrated mice were presented with water (n = 5 mice). Values are mean ± s.e.m. (error bars or shaded area).
Extended Data Figure 7 Silencing of SFONos1 neurons disinhibits feeding.
a, Experimental design to test whether prandial thirst inhibits food intake. b, Mice provided simultaneous access to water consumed more food after overnight fasting than mice without simultaneous access to water (**P < 0.01, ****P < 0.0001, two-way ANOVA, n = 10 mice per group), consistent with previous reports that thirst can inhibit hunger in rats33. c, Experimental design to test whether SFONos1 neurons mediate inhibition of food intake by prandial thirst. d, Silencing of SFONos1 neurons increased food intake when mice were provided with access to chow without simultaneous access to water after overnight fasting (****P < 0.0001, two-way repeated-measures ANOVA, n = 5 mice). Values are mean ± s.e.m. (error bars or shaded area).
Extended Data Figure 8 Projection mapping and retrograde tracing from SFO neurons.
a, Schematic of viral strategy for identifying projections from SFONos1 neurons using a fluorescent synaptophysin fusion protein. b, Representative images showing SFONos1 neuron somas in the SFO and axon terminals in the OVLT, MnPO, PVH and supraoptic nucleus (SON) (1 out of 1 mice; green, GFP; blue, DAPI). Scale bars, 100 μm. c, Schematic of strategy for retrograde tracing from SFO neurons using retrobeads. d, Representative images showing retrobeads injection site in the SFO and retrograde-labelled neurons in the medial septum (MS), OVLT, MnPO, ARC, MnR, dorsal raphe (DR), and locus coeruleus (LC) (1 out of 2 mice; red, rhodamine; blue, DAPI). Scale bars, 100 μm.
Extended Data Figure 9 Schematic for convergence of anticipatory and homeostatic signals at SFONos1 thirst neurons.
SFONos1 neurons monitor the composition of the blood by sensing plasma osmolarity and, via angiotensin, plasma volume and pressure. SFONos1 neurons predict the future state of the blood by integrating temperature-dependent inputs from the mouth and osmolarity-dependent inputs from the gut during drinking, and angiotensin- and osmolarity-independent inputs from the mouth/gut during eating. Illustration from National Institute of Diabetes and Digestive and Kidney Diseases, US National Institutes of Health.
Rights and permissions
About this article
Cite this article
Zimmerman, C., Lin, YC., Leib, D. et al. Thirst neurons anticipate the homeostatic consequences of eating and drinking. Nature 537, 680–684 (2016). https://doi.org/10.1038/nature18950
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature18950
This article is cited by
-
TMEM63B channel is the osmosensor required for thirst drive of interoceptive neurons
Cell Discovery (2024)
-
Thirst: neuroendocrine regulation in mammals
Veterinary Research Communications (2023)
-
CPT1A in AgRP neurons is required for sex-dependent regulation of feeding and thirst
Biology of Sex Differences (2023)
-
Exploring the neurobiology of the premonitory phase of migraine preclinically – a role for hypothalamic kappa opioid receptors?
The Journal of Headache and Pain (2022)
-
Dopamine subsystems that track internal states
Nature (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.