Abstract
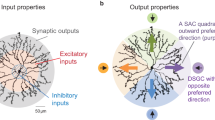
Directionally tuned signalling in starburst amacrine cell (SAC) dendrites lies at the heart of the circuit that detects the direction of moving stimuli in the mammalian retina. The relative contributions of intrinsic cellular properties and network connectivity to SAC direction selectivity remain unclear. Here we present a detailed connectomic reconstruction of SAC circuitry in mouse retina and describe two previously unknown features of synapse distributions along SAC dendrites: input and output synapses are segregated, with inputs restricted to proximal dendrites; and the distribution of inhibitory inputs is fundamentally different from that observed in rabbit retina. An anatomically constrained SAC network model suggests that SAC–SAC wiring differences between mouse and rabbit retina underlie distinct contributions of synaptic inhibition to velocity and contrast tuning and receptive field structure. In particular, the model indicates that mouse connectivity enables SACs to encode lower linear velocities that account for smaller eye diameter, thereby conserving angular velocity tuning. These predictions are confirmed with calcium imaging of mouse SAC dendrites responding to directional stimuli.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Barlow, H. B., Hill, R. M. & Levick, W. R. Retinal ganglion cells responding selectively to direction and speed of image motion in the rabbit. J. Physiol. 173, 377–407 (1964)
Famiglietti, E. V. Synaptic organization of starburst amacrine cells in rabbit retina: analysis of serial thin sections by electron microscopy and graphic reconstruction. J. Comp. Neurol. 309, 40–70 (1991)
Vaney, D. I., Collin, S. P. & Young, H. M. in Neurobiology of the Inner Retina (eds Weiler R. & Osborne N. N. ) 157–168 (Springer, 1989)
Briggman, K. L., Helmstaedter, M. & Denk, W. Wiring specificity in the direction-selectivity circuit of the retina. Nature 471, 183–188 (2011)
Wei, W., Hamby, A. M., Zhou, K. & Feller, M. B. Development of asymmetric inhibition underlying direction selectivity in the retina. Nature 469, 402–406 (2011)
Hausselt, S. E., Euler, T., Detwiler, P. B. & Denk, W. A dendrite-autonomous mechanism for direction selectivity in retinal starburst amacrine cells. PLoS Biol. 5, e185 (2007)
Euler, T., Detwiler, P. B. & Denk, W. Directionally selective calcium signals in dendrites of starburst amacrine cells. Nature 418, 845–852 (2002)
Lee, S. & Zhou, Z. J. The synaptic mechanism of direction selectivity in distal processes of starburst amacrine cells. Neuron 51, 787–799 (2006)
Tukker, J. J., Taylor, W. R. & Smith, R. G. Direction selectivity in a model of the starburst amacrine cell. Vis. Neurosci. 21, 611–625 (2004)
Gavrikov, K. E., Dmitriev, A. V., Keyser, K. T. & Mangel, S. C. Cation–chloride cotransporters mediate neural computation in the retina. Proc. Natl Acad. Sci. USA 100, 16047–16052 (2003)
Oesch, N. W. & Taylor, W. R. Tetrodotoxin-resistant sodium channels contribute to directional responses in starburst amacrine cells. PLoS One 5, e12447 (2010)
Kim, J. S. et al. Space-time wiring specificity supports direction selectivity in the retina. Nature 509, 331–336 (2014)
Taylor, W. R. & Smith, R. G. The role of starburst amacrine cells in visual signal processing. Vis. Neurosci. 29, 73–81 (2012)
Münch, T. A. & Werblin, F. S. Symmetric interactions within a homogeneous starburst cell network can lead to robust asymmetries in dendrites of starburst amacrine cells. J. Neurophysiol. 96, 471–477 (2006)
Enciso, G. A. et al. A model of direction selectivity in the starburst amacrine cell network. J. Comput. Neurosci. 28, 567–578 (2010)
Millar, T. J. & Morgan, I. G. Cholinergic amacrine cells in the rabbit retina synapse onto other cholinergic amacrine cells. Neurosci. Lett. 74, 281–285 (1987)
Denk, W. & Horstmann, H. Serial block-face scanning electron microscopy to reconstruct three-dimensional tissue nanostructure. PLoS Biol. 2, e329 (2004)
Dacheux, R. F., Chimento, M. F. & Amthor, F. R. Synaptic input to the on–off directionally selective ganglion cell in the rabbit retina. J. Comp. Neurol. 456, 267–278 (2003)
Roska, B. & Werblin, F. Vertical interactions across ten parallel, stacked representations in the mammalian retina. Nature 410, 583–587 (2001)
Baden, T., Berens, P., Bethge, M. & Euler, T. Spikes in mammalian bipolar cells support temporal layering of the inner retina. Curr. Biol. 23, 48–52 (2013)
Borghuis, B. G., Marvin, J. S., Looger, L. L. & Demb, J. B. Two-photon imaging of nonlinear glutamate release dynamics at bipolar cell synapses in the mouse retina. J. Neurosci. 33, 10972–10985 (2013)
Greene, M. J., Kim, J. S. & Seung, H. S. Analogous convergence of sustained and transient inputs in parallel on and off pathways for retinal motion computation. Cell Rep. 14, 1892–1900 (2016)
Ichinose, T., Fyk-Kolodziej, B. & Cohn, J. Roles of ON cone bipolar cell subtypes in temporal coding in the mouse retina. J. Neurosci. 34, 8761–8771 (2014)
Hoggarth, A. et al. Specific wiring of distinct amacrine cells in the directionally selective retinal circuit permits independent coding of direction and size. Neuron 86, 276–291 (2015)
Ishii, T. & Kaneda, M. ON-pathway-dominant glycinergic regulation of cholinergic amacrine cells in the mouse retina. J. Physiol. 592, 4235–4245 (2014)
Park, S. J. H., Kim, I.-J., Looger, L. L., Demb, J. B. & Borghuis, B. G. Excitatory synaptic inputs to mouse On-Off direction-selective retinal ganglion cells lack direction tuning. J. Neurosci. 34, 3976–3981 (2014)
Yonehara, K. et al. The first stage of cardinal direction selectivity is localized to the dendrites of retinal ganglion cells. Neuron 79, 1078–1085 (2013)
Chen, M., Lee, S., Park, S. J., Looger, L. L. & Zhou, Z. J. Receptive field properties of bipolar cell axon terminals in direction-selective sublaminas of the mouse retina. J. Neurophysiol. 112, 1950–1962 (2014)
Bozkir, G., Bozkir, M., Dogan, H., Aycan, K. & Güler, B. Measurements of axial length and radius of corneal curvature in the rabbit eye. Acta Med. Okayama 51, 9–11 (1997)
Park, H. et al. Assessment of axial length measurements in mouse eyes. Optometry Vision Sci. 89, 296–303 (2012)
Chan, Y. C. & Chiao, C. C. Effect of visual experience on the maturation of ON–OFF direction selective ganglion cells in the rabbit retina. Vision Res. 48, 2466–2475 (2008)
Weng, S., Sun, W. & He, S. Identification of ON–OFF direction-selective ganglion cells in the mouse retina. J. Physiol. (Lond.) 562, 915–923 (2005)
Denk, W., Strickler, J. H. & Webb, W. W. Two-photon laser scanning fluorescence microscopy. Science 248, 73–76 (1990)
Euler, T. et al. Eyecup scope—optical recordings of light stimulus-evoked fluorescence signals in the retina. Pflugers Arch. 457, 1393–1414 (2009)
Grzywacz, N. M. & Amthor, F. R. Robust directional computation in On–Off directionally selective ganglion cells of rabbit retina. Vis. Neurosci. 24, 647–661 (2007)
Morgan, J. L. & Lichtman, J. W. Why not connectomics? Nature Methods 10, 494–500 (2013)
Denk, W., Briggman, K. L. & Helmstaedter, M. Structural neurobiology: missing link to a mechanistic understanding of neural computation. Nat. Rev. Neurosci. 13, 351–358 (2012)
Vlasits, A. L. et al. A role for synaptic input distribution in a dendritic computation of motion direction in the retina. Neuron 89, 1317–1330 (2016)
Vaney, D. I. ‘Coronate’ amacrine cells in the rabbit retina have the ‘starburst’ dendritic morphology. Proc. R. Soc. Lond. B. 220, 501–508 (1984)
Tauchi, M. & Masland, R. H. The shape and arrangement of the cholinergic neurons in the rabbit retina. Proc. R. Soc. Lond. B. 223, 101–119 (1984)
Pérez De Sevilla Müller, L., Shelley, J. & Weiler, R. Displaced amacrine cells of the mouse retina. J. Comp. Neurol. 505, 177–189 (2007)
Keeley, P. W., Whitney, I. E., Raven, M. A. & Reese, B. E. Dendritic spread and functional coverage of starburst amacrine cells. J. Comp. Neurol. 505, 539–546 (2007)
Kostadinov, D. & Sanes, J. R. Protocadherin-dependent dendritic self-avoidance regulates neural connectivity and circuit function. eLife 4, (2015)
Helmstaedter, M. et al. Connectomic reconstruction of the inner plexiform layer in the mouse retina. Nature 500, 168–174 (2013)
Smith, R. G. NeuronC: a computational language for investigating functional architecture of neural circuits. J. Neurosci. Methods 43, 83–108 (1992)
Schachter, M. J., Oesch, N., Smith, R. G. & Taylor, W. R. Dendritic spikes amplify the synaptic signal to enhance detection of motion in a simulation of the direction-selective ganglion cell. PLOS Comput. Biol. 6, e1000899 (2010)
Pologruto, T. A., Sabatini, B. L. & Svoboda, K. ScanImage: flexible software for operating laser scanning microscopes. Biomed. Eng. Online 2, 13 (2003)
Acknowledgements
We thank W. Denk for supporting the collection of the serial block-face scanning electron microscopy data in his laboratory. This work was supported by NIH grants EY016607 and EY022070 (RGS), by the NINDS Intramural Research Program (NS003145; J.S.D.) and (NS003133; K.L.B.), the Max-Planck Society (K.L.B.), and the Pew Charitable Trusts (K.L.B.).
Author information
Authors and Affiliations
Contributions
H.D., R.G.S. and K.L.B. collected and analysed data; H.D., R.G.S., A.P.-P., J.S.D., and K.L.B. designed the study and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer Information Nature thanks G. Knott and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Extended data figures and tables
Extended Data Figure 1 EM data set, additional SAC reconstructions and rabbit connectivity.
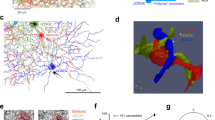
a, Conventionally stained serial block-face scanning electron microscopy volume of a mouse retina. b, Reconstructed ON-OFF DSGC. c–e, A second reconstructed ON and OFF SAC with annotated synapses locations. f, Annotation of the radial distribution of input and output synapses to and from approximately one-half of an OFF SAC dendritic arbor in rabbit retina. Data analysed from fig. 15 in ref. 2.
Extended Data Figure 2 Classification of OFF bipolar cells.
a, Types 1/2 and types 3/4 separated by IPL depth. b, Types 1 and 2 separate by stratification width and axonal arborization area (convex hull). c, Types 3a, 3b and 4 separate by stratification depth and axonal arborization area. d, Mosaic patterns and stratification profiles of OFF bipolar cells. e, The number of synapses (mean ± s.d.) each bipolar cell, by type, formed with each SAC. f, Location of bipolar cell synapses onto a second OFF SAC, colour-coded by bipolar cell type. g, The IPL depth of each synapse versus the radial distance relative to the soma.
Extended Data Figure 3 Classification of ON bipolar cells.
a, Type 5 and type 7 biploar cells separated by IPL depth. b, Types 5o (outer), 5t (thick) and 5i (inner) further subdivide based on IPL depth and stratification width. c, Mosaic patterns and stratification profiles of ON bipolar cells. d, Summary of the number of synapses (mean ± s.d.) each bipolar cell, by type, formed with each SAC. e, Location of bipolar cell synapses onto a second ON SAC, colour-coded by bipolar cell type. f, The IPL depth of each synapse versus the radial distance relative to the soma.
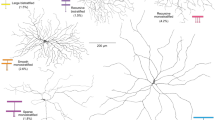
Extended Data Figure 4 Amacrine cell types presynaptic to SACs.
a, b, SACs presynaptic to the second pair of mouse SACs colour-coded by absolute orientation. c, d, Wide-field amacrine cells presynaptic to SACs. e, Narrow-field amacrine cells presynaptic to ON SACs.
Extended Data Figure 5 Relative angles between presynaptic and postsynaptic SAC dendrites.
a, Schematic of the relative angle measurement: parallel wiring = 0°, anti-parallel wiring = 180°. b, Locations of SAC input synapses colour-coded by relative angle. Grey locations indicate AC synapses that were not analysed. c, Cumulative distributions of the relative angles between each presynaptic and postsynaptic OFF SAC dendrite for synapses (black) and proximities (grey). Dashed line indicates a uniform distribution. d, Relative angle for each synapse was uncorrelated with the radial distance from the postsynaptic somas (r = 0.07, P = 0.16). Scale bar, 50 μm.
Extended Data Figure 6 Identities of neurons postsynaptic to SAC output synapses.
a, Percentage of output synapses formed with different postsynaptic cell types, colour-coded by postsynaptic cell class: ganglion cells (GC) (blue), SACs (red), bipolar cells (BC) (cyan), and wide-field amacrine cells (WAC) (green). b, Locations of 83 annotatedoutput synapses on 1 ON SAC dendrite fragment. c, Locations of 110 annotated output synapses on 2 OFF SAC dendrite fragments. Scale bar, 50 μm.
Extended Data Figure 7 Single SAC model.
a, Dendrite diameters sampled from an ON SAC (grey) and an OFF SAC (black) at different radial distances from their respective somas. b, Single SAC morphology used in all simulations. c, Somatic voltage clamp simulation showed poor space clamp of even proximal dendrites. Voltage traces measured at a different distances (20–150 μm) from the soma. d, Somatic (solid line) and distal dendrite (dashed line) voltage time series in response to an annulus moving centrifugally or centripetally. The addition of active conductances to SAC dendrites (see Extended Data Table 1) rendered somatic voltage recordings directionally selective for centrifugal compared to centripetal stimulation, consistent with electrophysiological measurements. Scale bar, 50 μm.
Extended Data Figure 8 Velocity tuning of rabbit and mouse direction selectivity circuits.
a, Schematic of the difference in axial diameters and subtended angle on the retina of rabbit and mouse eyes. b, Linear velocity tuning curves from rabbit and mouse ON–OFF DSGCs. c, Angular velocity tuning curves from rabbit and mouse ON–OFF DSGCs. Data analysed from fig. 2F of ref. 31 and fig. 1D of ref. 32.
Rights and permissions
About this article
Cite this article
Ding, H., Smith, R., Poleg-Polsky, A. et al. Species-specific wiring for direction selectivity in the mammalian retina. Nature 535, 105–110 (2016). https://doi.org/10.1038/nature18609
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature18609
This article is cited by
-
A presynaptic source drives differing levels of surround suppression in two mouse retinal ganglion cell types
Nature Communications (2024)
-
Dendritic mGluR2 and perisomatic Kv3 signaling regulate dendritic computation of mouse starburst amacrine cells
Nature Communications (2024)
-
Heterogeneous receptor expression underlies non-uniform peptidergic modulation of olfaction in Drosophila
Nature Communications (2023)
-
A circuit suppressing retinal drive to the optokinetic system during fast image motion
Nature Communications (2023)
-
Direction Selectivity of TmY Neurites in Drosophila
Neuroscience Bulletin (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.