Abstract
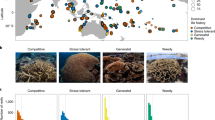
Ongoing declines in the structure and function of the world’s coral reefs1,2 require novel approaches to sustain these ecosystems and the millions of people who depend on them3. A presently unexplored approach that draws on theory and practice in human health and rural development4,5 is to systematically identify and learn from the ‘outliers’—places where ecosystems are substantially better (‘bright spots’) or worse (‘dark spots’) than expected, given the environmental conditions and socioeconomic drivers they are exposed to. Here we compile data from more than 2,500 reefs worldwide and develop a Bayesian hierarchical model to generate expectations of how standing stocks of reef fish biomass are related to 18 socioeconomic drivers and environmental conditions. We identify 15 bright spots and 35 dark spots among our global survey of coral reefs, defined as sites that have biomass levels more than two standard deviations from expectations. Importantly, bright spots are not simply comprised of remote areas with low fishing pressure; they include localities where human populations and use of ecosystem resources is high, potentially providing insights into how communities have successfully confronted strong drivers of change. Conversely, dark spots are not necessarily the sites with the lowest absolute biomass and even include some remote, uninhabited locations often considered near pristine6. We surveyed local experts about social, institutional, and environmental conditions at these sites to reveal that bright spots are characterized by strong sociocultural institutions such as customary taboos and marine tenure, high levels of local engagement in management, high dependence on marine resources, and beneficial environmental conditions such as deep-water refuges. Alternatively, dark spots are characterized by intensive capture and storage technology and a recent history of environmental shocks. Our results suggest that investments in strengthening fisheries governance, particularly aspects such as participation and property rights, could facilitate innovative conservation actions that help communities defy expectations of global reef degradation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Pandolfi, J. M. et al. Global trajectories of the long-term decline of coral reef ecosystems. Science 301, 955–958 (2003)
Bellwood, D. R., Hughes, T. P., Folke, C. & Nyström, M. Confronting the coral reef crisis. Nature 429, 827–833 (2004)
Hughes, T. P., Bellwood, D. R., Folke, C., Steneck, R. S. & Wilson, J. New paradigms for supporting the resilience of marine ecosystems. Trends Ecol. Evol. 20, 380–386 (2005)
Sternin, M. et al. in The Hearth Nutrition Model: Applications in Haiti, Vietnam, and Bangladesh. (eds O Wollinka, E Keeley, B Burkhalter, & N Bashir ) 49–61 (VA: BASICS, 1997)
Pretty, J. N. et al. Resource-conserving agriculture increases yields in developing countries. Environ. Sci. Technol. 40, 1114–1119 (2006)
Knowlton, N. & Jackson, J. B. C. Shifting baselines, local impacts, and global change on coral reefs. PLoS Biol. 6, e54 (2008)
Naeem, S., Duffy, J. E. & Zavaleta, E. The functions of biological diversity in an age of extinction. Science 336, 1401–1406 (2012)
Devillers, R. et al. Reinventing residual reserves in the sea: are we favouring ease of establishment over need for protection? Aquat. Conserv. 25, 480–504 (2014)
Pressey, R. L., Visconti, P. & Ferraro, P. J. Making parks make a difference: poor alignment of policy, planning and management with protected-area impact, and ways forward. Philos. Trans. R. Soc. B 370, 20140280 (2015)
Pascale, R. T. & Sternin, J. Your company’s secret change agents. Harv. Bus. Rev. 83, 72–81, 153 (2005)
Levinson, F. J., Barney, J., Bassett, L. & Schultink, W. Utilization of positive deviance analysis in evaluating community-based nutrition programs: an application to the Dular program in Bihar, India. Food Nutr. Bull. 28, 259–265 (2007)
McClanahan, T. R. et al. Critical thresholds and tangible targets for ecosystem-based management of coral reef fisheries. Proc. Natl Acad. Sci. USA 108, 17230–17233 (2011)
York, R. et al. Footprints on the earth: The environmental consequences of modernity. Am. Sociol. Rev. 68, 279–300 (2003)
Lambin, E. F. et al. The causes of land-use and land-cover change: moving beyond the myths. Glob. Environ. Change 11, 261–269 (2001)
Cinner, J. E. et al. Comanagement of coral reef social-ecological systems. Proc. Natl Acad. Sci. USA 109, 5219–5222 (2012)
Hughes, T. P., Huang, H. & Young, M. A. The wicked problem of China’s disappearing coral reefs. Conserv. Biol. 27, 261–269 (2013)
Dodd, S. C. The interactance hypothesis: a gravity model fitting physical masses and human groups. Am. Sociol. Rev. 15, 245–256 (1950)
Wittemyer, G., Elsen, P., Bean, W. T., Burton, A. C. & Brashares, J. S. Accelerated human population growth at protected area edges. Science 321, 123–126 (2008)
Noble, A. et al. in Bright spots demonstrate community successes in African agriculture (ed. F. W. T. Penning de Vries ) 7 (International Water Management Institute, 2005)
Cinner, J. et al. Periodic closures as adaptive coral reef management in the Indo-Pacific. Ecol. Soc. 11, 31 (2006)
Lindfield, S. J. et al. Mesophotic depths as refuge areas for fishery-targeted species on coral reefs. Coral Reefs 35, 125–137 (2016)
Cinner, J. E. et al. A framework for understanding climate change impacts on coral reef social–ecological systems. Reg. Environ. Change 16, 1133–1146 (2016)
Cinner, J. E. Social-ecological traps in reef fisheries. Glob. Environ. Change 21, 835–839 (2011)
O’Rourke, D. The science of sustainable supply chains. Science 344, 1124–1127 (2014)
Sampson, G. S. et al. Sustainability. Secure sustainable seafood from developing countries. Science 348, 504–506 (2015)
Schmitt, K. M. & Kramer, D. B. Road development and market access on Nicaragua’s Atlantic coast: implications for household fishing and farming practices. Environ. Conserv. 36, 289–300 (2009)
Falk, A. & Szech, N. Morals and markets. Science 340, 707–711 (2013)
Sandel, M. J. What Money Can't Buy: The Moral Limits Of Markets. (Macmillan, 2012)
Ostrom, E. Governing the Commons: The Evolution of Institutions for Collective Action. (Cambridge University Press, 1990)
Graham, N. A., Jennings, S., MacNeil, M. A., Mouillot, D. & Wilson, S. K. Predicting climate-driven regime shifts versus rebound potential in coral reefs. Nature 518, 94–97 (2015)
Daw, T. et al. The Spatial Behaviour of Artisanal Fishers: Implications for Fisheries Management and Development (Fishers in Space). (WIOMSA, 2011)
MacNeil, M. A. et al. Recovery potential of the world’s coral reef fishes. Nature 520, 341–344 (2015)
Mora, C. et al. Global human footprint on the linkage between biodiversity and ecosystem functioning in reef fishes. PLoS Biol. 9, e1000606 (2011)
Edwards, C. B. et al. Global assessment of the status of coral reef herbivorous fishes: evidence for fishing effects. Proc. Biol. Sci. 281, 20131835 (2014)
Froese, R. & Pauly, D. FishBase. http://www.fishbase.org (2014)
Center for International Earth Science Information Network (CIESIN). Gridded population of the world. Version 3 (GPWv3)http://sedac.ciesin.columbia.edu/gpw (2005)
MacNeil, M. A. & Connolly, S. R. in Ecology of Fishes on Coral Reefs (ed C. Mora ) Ch. 12, 116–126 (2015).
Mora, C. et al. Ecology. Coral reefs and the global network of Marine Protected Areas. Science 312, 1750–1751 (2006)
Ravenstein, E. G. The laws of migration. J. R. Stat. Soc. 48, 167–235 (1885)
Anderson, J. E. A theoretical foundation for the gravity equation. Am. Econ. Rev. 69, 106–116 (1979)
Anderson, J. E. The Gravity Model. (National Bureau of Economic Research, 2010)
Lukermann, F. & Porter, P. W. Gravity and potential models in economic geography. Ann. Assoc. Am. Geogr. 50, 493–504 (1960)
Nelson, A. Travel Time to Major Cities: A Global Map of Accessibility. (Ispra, Italy, 2008)
Bartholomé, E. et al. GLC 2000: Global Land Cover Mapping for the Year 2000: Project Status November 2002. (Institute for Environment and Sustainability, 2002)
R Core Team. R: A language and environment for statistical computing. https://www.R-project.org (2016)
Dijkstra, E. W. A note on two problems in connexion with graphs. Numer. Math. 1, 269–271 (1959)
Black, W. R. An analysis of gravity model distance exponents. Transportation 2, 299–312 (1973)
Emran, M. S. & Shilpi, F. The Extent of the Market and Stages of Agricultural Specialization. Vol. 4534 (World Bank Publications, 2008)
Cinner, J. E., Graham, N. A., Huchery, C. & Macneil, M. A. Global effects of local human population density and distance to markets on the condition of coral reef fisheries. Conserv. Biol. 27, 453–458 (2013)
Teh, L. S. L., Teh, L. C. & Sumaila, U. R. A Global Estimate of the Number of Coral Reef Fishers. PLoS One 8, e65397 (2013)
Andréfouët, S. et al. in 10th International Coral Reef Symposium (eds Y. Suzuki et al. ) 1732–1745 (Japanese Coral Reef Society, 2006)
Maina, J., McClanahan, T. R., Venus, V., Ateweberhan, M. & Madin, J. Global gradients of coral exposure to environmental stresses and implications for local management. PLoS One 6, e23064 (2011)
Gelman, A. et al. Bayesian Data Analysis. Vol. 2 (Taylor & Francis, 2014)
Patil, A., Huard, D. & Fonnesbeck, C. J. PyMC: Bayesian stochastic modelling in Python. J. Stat. Softw. 35, 1–81 (2010)
Gelman, A. & Rubin, D. B. Inference from iterative simulation using multiple sequences. Stat. Sci. 7, 457–472 (1992)
Acknowledgements
The ARC Centre of Excellence for Coral Reef Studies, Stanford University, and University of Montpellier funded working group meetings. This work was supported by J.E.C.’s Pew Fellowship in Marine Conservation and ARC Australian Research Fellowship. Thanks to M. Barnes for constructive comments.
Author information
Authors and Affiliations
Contributions
J.E.C. conceived of the study with support from M.A.M., N.A.J.G., T.R.M., J.K., C.Hu., D.M., C.M., E.H.A., and C.C.Hi.; C.Hu. managed the database; M.A.M., J.E.C., and D.M. developed and implemented the analyses; J.E.C. led the manuscript with M.A.M. and N.A.J.G. All other authors contributed data and made substantive contributions to the text.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
This is the Social-Ecological Research Frontiers (SERF) working group contribution no. 11.
Reviewer Information Nature thanks S. Qian, B. Walker and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Extended data figures and tables
Extended Data Figure 1 Marginal relationships between reef fish biomass and social drivers.
a, Local population growth; b, market gravity; c, nearest settlement gravity; d, tourism; e, nation/state population size; f, Human Development Index; g, high compliance marine reserve (0 is fished baseline); h, restricted fishing (0 is fished baseline); i, low-compliance marine reserve (0 is fished baseline); j, voice and accountability; k, reef fish landings; l, ocean productivity; m, depth (−1 = 0–4 m, 0 = 4–10 m, 1 = >10 m); n, reef flat (0 is reef slope baseline); o, reef crest flat (0 is reef slope baseline); p, lagoon/back reef flat (0 is reef slope baseline). All variables displayed on the x axis are standardized. Red lines are the marginal trend line for each parameter as estimated by the full model. Grey lines are 100 simulations of the marginal trend line sampled from the posterior distributions of the intercept and parameter slope, analogous to conventional confidence intervals. Two asterisks indicate that 95% of the posterior density is in either a positive or negative direction (Fig. 1b–d); a single asterisk indicates that 75% of the posterior density is in either a positive or negative direction.
Extended Data Figure 2 Correlation plot of candidate continuous covariates before accounting for collinearity (Extended Data Table 4).
Collinearity between continuous and categorical covariates (including biogeographic region, habitat, protection status, and depth) were analysed using box plots.
Extended Data Figure 3 Model fit statistics.
Top, Bayesian P values (BpV) for the full model indicating goodness of fit, based on posterior discrepancy. Points are Freeman–Tukey differences between observed and expected values, and simulated and expected values within the MCMC scheme (n = 10,000). Plot shows no evidence for lack of fit between the model and the data. Bottom, Posterior distribution for the degrees of freedom parameter (ν) in our supplementary analysis of candidate distributions. The highest posterior density of 3.46, with 97.5% of the total posterior density below 4 provides strong evidence in favour of a non-central t distribution relative to a normal distribution and supports the use of 3.5 for ν.
Extended Data Figure 4 Box plot of deviation from expected as a function of the presence or absence of key social and environmental conditions expected to produce bright spots.
Boxes range from the first to third quartile and whiskers extend to the highest value that is within 1.5× the inter-quartile range (that is, distance between the first and third quartiles). Data beyond the end of the whiskers are outliers, which are plotted as points.
Supplementary information
Supplementary Information
This file contains Supplementary Text and Data and an additional reference. (PDF 511 kb)
Rights and permissions
About this article
Cite this article
Cinner, J., Huchery, C., MacNeil, M. et al. Bright spots among the world’s coral reefs. Nature 535, 416–419 (2016). https://doi.org/10.1038/nature18607
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature18607
This article is cited by
-
Viral predation pressure on coral reefs
BMC Biology (2023)
-
Temperate functional niche availability not resident-invader competition shapes tropicalisation in reef fishes
Nature Communications (2023)
-
Coral reefs benefit from reduced land–sea impacts under ocean warming
Nature (2023)
-
Nutrient supply from marine small-scale fisheries
Scientific Reports (2023)
-
Asynchrony in coral community structure contributes to reef-scale community stability
Scientific Reports (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.