Abstract
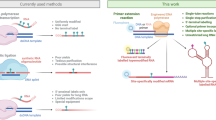
Enzymes that contain metal ions—that is, metalloenzymes—possess the reactivity of a transition metal centre and the potential of molecular evolution to modulate the reactivity and substrate-selectivity of the system1. By exploiting substrate promiscuity and protein engineering, the scope of reactions catalysed by native metalloenzymes has been expanded recently to include abiological transformations2,3. However, this strategy is limited by the inherent reactivity of metal centres in native metalloenzymes. To overcome this limitation, artificial metalloproteins have been created by incorporating complete, noble-metal complexes within proteins lacking native metal sites1,4,5. The interactions of the substrate with the protein in these systems are, however, distinct from those with the native protein because the metal complex occupies the substrate binding site. At the intersection of these approaches lies a third strategy, in which the native metal of a metalloenzyme is replaced with an abiological metal with reactivity different from that of the metal in a native protein6,7,8. This strategy could create artificial enzymes for abiological catalysis within the natural substrate binding site of an enzyme that can be subjected to directed evolution. Here we report the formal replacement of iron in Fe-porphyrin IX (Fe-PIX) proteins with abiological, noble metals to create enzymes that catalyse reactions not catalysed by native Fe-enzymes or other metalloenzymes9,10. In particular, we prepared modified myoglobins containing an Ir(Me) site that catalyse the functionalization of C–H bonds to form C–C bonds by carbene insertion and add carbenes to both β-substituted vinylarenes and unactivated aliphatic α-olefins. We conducted directed evolution of the Ir(Me)-myoglobin and generated mutants that form either enantiomer of the products of C–H insertion and catalyse the enantio- and diastereoselective cyclopropanation of unactivated olefins. The presented method of preparing artificial haem proteins containing abiological metal porphyrins sets the stage for the generation of artificial enzymes from innumerable combinations of PIX-protein scaffolds and unnatural metal cofactors to catalyse a wide range of abiological transformations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lewis, J. C. Artificial metalloenzymes and metallopeptide catalysts for organic synthesis. ACS Catal. 3, 2954–2975 (2013)
Farwell, C. C., Zhang, R. K., McIntosh, J. A., Hyster, T. K. & Arnold, F. H. Enantioselective enzyme-catalyzed aziridination enabled by active-site evolution of a cytochrome P450. ACS Cent. Sci. 1, 89–93 (2015)
Hyster, T. K. & Arnold, F. H. P450BM3-axial mutations: a gateway to non-natural reactivity. Isr. J. Chem. 55, 14–20 (2015)
Ringenberg, M. R. & Ward, T. R. Merging the best of two worlds: artificial metalloenzymes for enantioselective catalysis. Chem. Commun. 47, 8470–8476 (2011)
Ward, T. R. Artificial metalloenzymes based on the biotin−avidin technology: enantioselective catalysis and beyond. Acc. Chem. Res. 44, 47–57 (2011)
Jing, Q. & Kazlauskas, R. J. Regioselective hydroformylation of styrene using rhodium-substituted carbonic anhydrase. ChemCatChem 2, 953–957 (2010)
Abe, S. et al. Polymerization of phenylacetylene by rhodium complexes within a discrete space of apo-ferritin. J. Am. Chem. Soc. 131, 6958–6960 (2009)
Key, H. M., Clark, D. S. & Hartwig, J. F. Generation, characterization, and tunable reactivity of organometallic fragments bound to a protein ligand. J. Am. Chem. Soc. 137, 8261–8268 (2015)
Coelho, P. S., Brustad, E. M., Kannan, A. & Arnold, F. H. Olefin cyclopropanation via carbene transfer catalyzed by engineered cytochrome P450 enzymes. Science 339, 307–310 (2013)
Bordeaux, M., Tyagi, V. & Fasan, R. Highly diastereoselective and enantioselective olefin cyclopropanation using engineered myoglobin-based catalysts. Angew. Chem. Int. Ed. 54, 1744–1748 (2015)
Ortiz de Montellano, P. Hydrocarbon hydroxylation by cytochrome P450 enzymes. Chem. Rev. 110, 932–948 (2010)
Peters, M. W., Meinhold, P., Glieder, A. & Arnold, F. H. Regio- and enantioselective alkane hydroxylation with engineered cytochromes P450 BM-3. J. Am. Chem. Soc. 125, 13442–13450 (2003)
Roiban, G.-D. & Reetz, M. T. Expanding the toolbox of organic chemists: directed evolution of P450 monooxygenases as catalysts in regio- and stereoselective oxidative hydroxylation. Chem. Commun. 51, 2208–2224 (2015)
Tyagi, V., Bonn, R. B. & Fasan, R. Intermolecular carbene S-H insertion catalyzed by engineered myoglobin-based catalysts. Chem. Sci. 6, 2488–2494 (2015)
Chan, K. H., Guan, X., Lo, V. K. Y. & Che, C.-M. Elevated catalytic activity of ruthenium(II)–porphyrin-catalyzed carbene/nitrene transfer and insertion reactions with N-heterocyclic carbene ligands. Angew. Chem. Int. Ed. 53, 2982–2987 (2014)
Maxwell, J. L., O’Malley, S., Brown, K. C. & Kodadek, T. Shape-selective and asymmetric cyclopropanation of alkenes catalyzed by rhodium porphyrins. Organometallics 11, 645–652 (1992)
Anding, B. J., Ellern, A. & Woo, L. K. Olefin cyclopropanation catalyzed by iridium(III) porphyrin complexes. Organometallics 31, 3628–3635 (2012)
Carey, J. R. et al. A site-selective dual anchoring strategy for artificial metalloprotein design. J. Am. Chem. Soc. 126, 10812–10813 (2004)
Ohashi, M. et al. Preparation of artificial metalloenzymes by insertion of chromium(III) Schiff base complexes into apomyoglobin mutants. Angew. Chem. Int. Ed. 42, 1005–1008 (2003)
Oohora, K., Kihira, Y., Mizohata, E., Inoue, T. & Hayashi, T. C(sp3)–H bond hydroxylation catalyzed by myoglobin reconstituted with manganese porphycene. J. Am. Chem. Soc. 135, 17282–17285 (2013)
Bordeaux, M., Singh, R. & Fasan, R. Intramolecular C(sp3)–H amination of arylsulfonyl azides with engineered and artificial myoglobin-based catalysts. Bioorg. Med. Chem. 22, 5697–5704 (2014)
Teale, F. W. Cleavage of the haem-protein link by acid methylethylketone. Biochim. Biophys. Acta 35, 543 (1959)
Lelyveld, V. S., Brustad, E., Arnold, F. H. & Jasanoff, A. Metal-substituted protein MRI contrast agents engineered for enhanced relaxivity and ligand sensitivity. J. Am. Chem. Soc. 133, 649–651 (2011)
Kawakami, N., Shoji, O. & Watanabe, Y. Single-step reconstitution of apo-hemoproteins at the disruption stage of Escherichia coli cells. ChemBioChem 13, 2045–2047 (2012)
Woodward, J. J., Martin, N. I. & Marletta, M. A. An Escherichia coli expression-based method for heme substitution. Nat. Methods 4, 43–45 (2007)
Paulson, D. R., Addison, A. W., Dolphin, D. & James, B. R. Preparation of ruthenium(II) and ruthenium(III) myoglobin and the reaction of dioxygen, and carbon monoxide, with ruthenium(II) myoglobin. J. Biol. Chem. 254, 7002–7006 (1979)
DelProposto, J., Majmudar, C. Y., Smith, J. L. & Brown, W. C. Mocr: a novel fusion tag for enhancing solubility that is compatible with structural biology applications. Protein Expr. Purif. 63, 40–49 (2009)
Ilie, A. & Reetz, M. T. Directed evolution of artificial metalloenzymes. Isr. J. Chem. 55, 51–60 (2015)
Suematsu, H., Kanchiku, S., Uchida, T. & Katsuki, T. Construction of aryliridium-salen complexes: enantio- and cis-selective cyclopropanation of conjugated and nonconjugated olefins. J. Am. Chem. Soc. 130, 10327–10337 (2008)
Watson, H. C. The stereochemistry of the protein myoglobin. Protein Stereochem. 4, 299 (1969)
Acknowledgements
This work was supported by the Director, Office of Science, of the US Department of Energy under contract no. DE-AC02- 05CH11231, by the NSF (graduate research fellowship to H.M.K.), and the NWO Netherlands Organization for Scientific Research (Rubicon postdoctoral fellowship no. 680-50-1306 to P.D.). We thank the QB3 MacroLab facility (sub-cloning), the UC Berkeley DNA Sequencing Facility (plasmid sequencing), T. Iavarone and the QB3 Mass Spectrometry Facility (supported by NIH grant 1S10RR022393-01) for native NS-ESI-MS data and analysis, and H. Zhao (University of Illinois-Champaign Urbana) for the P411-CIS gene.
Author information
Authors and Affiliations
Contributions
H.M.K., P.D., and J.F.H. conceived the work, designed the initial experiments, and interpreted the data. All authors discussed the results to design subsequent experiments. H.M.K. and P.D. performed all the experiments. H.M.K., P.D., and J.F.H. wrote the manuscript and all authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
Competing Interests: The authors have filed through the Lawrence Berkeley National Laboratory a provisional patent application (U.S. Patent Application Ser. No: 62/241,487) that is based on the results presented here.
Supplementary information
Supplementary Information
This file contains Supplementary Methods, Supplementary Figures 1-29, Supplementary Tables 1-16, NMR Spectra and additional references. (PDF 49843 kb)
Rights and permissions
About this article
Cite this article
Key, H., Dydio, P., Clark, D. et al. Abiological catalysis by artificial haem proteins containing noble metals in place of iron. Nature 534, 534–537 (2016). https://doi.org/10.1038/nature17968
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature17968
This article is cited by
-
Chemodivergent C(sp3)–H and C(sp2)–H cyanomethylation using engineered carbene transferases
Nature Catalysis (2023)
-
Multifaceted personality and roles of heme enzymes in industrial biotechnology
3 Biotech (2023)
-
Silver-catalyzed site-selective C(sp3)−H benzylation of ethers with N-triftosylhydrazones
Nature Communications (2022)
-
Data-informed discovery of hydrolytic nanozymes
Nature Communications (2022)
-
Hydride surprise
Nature Chemistry (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.