Abstract
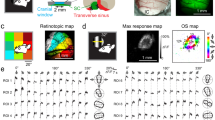
Circuits in the visual cortex integrate the information derived from separate ON (light-responsive) and OFF (dark-responsive) pathways to construct orderly columnar representations of stimulus orientation and visual space1,2,3,4,5,6,7. How this transformation is achieved to meet the specific topographic constraints of each representation remains unclear. Here we report several novel features of ON–OFF convergence visualized by mapping the receptive fields of layer 2/3 neurons in the tree shrew (Tupaia belangeri) visual cortex using two-photon imaging of GCaMP6 calcium signals. We show that the spatially separate ON and OFF subfields of simple cells in layer 2/3 exhibit topologically distinct relationships with the maps of visual space and orientation preference. The centres of OFF subfields for neurons in a given region of cortex are confined to a compact region of visual space and display a smooth visuotopic progression. By contrast, the centres of the ON subfields are distributed over a wider region of visual space, display substantial visuotopic scatter, and have an orientation-specific displacement consistent with orientation preference map structure. As a result, cortical columns exhibit an invariant aggregate receptive field structure: an OFF-dominated central region flanked by ON-dominated subfields. This distinct arrangement of ON and OFF inputs enables continuity in the mapping of both orientation and visual space and the generation of a columnar map of absolute spatial phase.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hubel, D. H. & Wiesel, T. N. Receptive fields, binocular interaction and functional architecture in the cat’s visual cortex. J. Physiol. (Lond.) 160, 106–154 (1962)
Ferster, D., Chung, S. & Wheat, H. Orientation selectivity of thalamic input to simple cells of cat visual cortex. Nature 380, 249–252 (1996)
Jin, J. Z. et al. On and off domains of geniculate afferents in cat primary visual cortex. Nature Neurosci. 11, 88–94 (2008)
Jin, J., Wang, Y., Swadlow, H. A. & Alonso, J. M. Population receptive fields of ON and OFF thalamic inputs to an orientation column in visual cortex. Nature Neurosci. 14, 232–238 (2011)
Bosking, W. H., Crowley, J. C. & Fitzpatrick, D. Spatial coding of position and orientation in primary visual cortex. Nature Neurosci. 5, 874–882 (2002)
Reid, R. C. & Alonso, J. M. Specificity of monosynaptic connections from thalamus to visual cortex. Nature 378, 281–284 (1995)
Alonso, J. M., Usrey, W. M. & Reid, R. C. Rules of connectivity between geniculate cells and simple cells in cat primary visual cortex. J. Neurosci. 21, 4002–4015 (2001)
Fan, Y. et al. Genome of the Chinese tree shrew. Nat. Commun. 4, 1426 (2013)
Rockland, K. S. & Lund, J. S. Widespread periodic intrinsic connections in tree shrew visual cortex (Area 17). Science 215, 1532–1534 (1982)
Bosking, W. H., Zhang, Y., Schofield, B. & Fitzpatrick, D. Orientation selectivity and the arrangement of horizontal connections in tree shrew striate cortex. J. Neurosci. 17, 2112–2127 (1997)
Van Hooser, S. D. et al. Transformation of receptive field properties from lateral geniculate nucleus to superficial V1 in the tree shrew. J. Neurosci. 33, 11494–11505 (2013)
Kretz, R., Rager, G. & Norton, T. T. Laminar organization of ON and OFF regions and ocular dominance in the striate cortex of the tree shrew (Tupaia belangeri). J. Comp. Neurol. 251, 135–145 (1986)
Muly, E. C. & Fitzpatrick, D. The morphological basis for binocular and ON/OFF convergence in tree shrew striate cortex. J. Neurosci. 12, 1319–1334 (1992)
Jones, J. P. & Palmer, L. A. The two-dimensional spatial structure of simple receptive fields in cat striate cortex. J. Neurophysiol. 58, 1187–1211 (1987)
Lampl, I., Anderson, J. S., Gillespie, D. C. & Ferster, D. Prediction of orientation selectivity from receptive field architecture in simple cells of cat visual cortex. Neuron 30, 263–274 (2001)
Ringach, D. L. Spatial structure and symmetry of simple-cell receptive fields in macaque primary visual cortex. J. Neurophysiol. 88, 455–463 (2002)
Yeh, C. I., Xing, D., Williams, P. E. & Shapley, R. M. Stimulus ensemble and cortical layer determine V1 spatial receptive fields. Proc. Natl Acad. Sci. USA 106, 14652–14657 (2009)
Chichilnisky, E. J. & Kalmar, R. S. Functional asymmetries in ON and OFF ganglion cells of primate retina. J. Neurosci. 22, 2737–2747 (2002)
Devries, S. H. & Baylor, D. A. Mosaic arrangement of ganglion cell receptive fields in rabbit retina. J. Neurophysiol. 78, 2048–2060 (1997)
Ratliff, C. P., Borghuis, B. G., Kao, Y. H., Sterling, P. & Balasubramanian, V. Retina is structured to process an excess of darkness in natural scenes. Proc. Natl Acad. Sci. USA 107, 17368–17373 (2010)
Miller, K. D. A model for the development of simple cell receptive fields and the ordered arrangement of orientation columns through activity-dependent competition between ON- and OFF-center inputs. J. Neurosci. 14, 409–441 (1994)
Ringach, D. L. Haphazard wiring of simple receptive fields and orientation columns in visual cortex. J. Neurophysiol. 92, 468–476 (2004)
Oppenheim, A. V. & Lim, J. S. The importance of phase in signals. Proc. IEEE 69, 529–541 (1981)
Pollen, D. A. & Ronner, S. F. Phase relationships between adjacent simple cells in the visual cortex. Science 212, 1409–1411 (1981)
Adelson, E. H. & Bergen, J. R. Spatiotemporal energy models for the perception of motion. J. Opt. Soc. Am. A 2, 284–299 (1985)
Kara, P. & Boyd, J. D. A micro-architecture for binocular disparity and ocular dominance in visual cortex. Nature 458, 627–631 (2009)
Smith, G. B., Whitney, D. E. & Fitzpatrick, D. Modular representation of luminance polarity in the superficial layers of primary visual cortex. Neuron 88, 805–818 (2015)
Wang, Y. et al. Columnar organization of spatial phase in visual cortex. Nature Neurosci. 18, 97–103 (2015)
Smith, S. L. & Häusser, M. Parallel processing of visual space by neighboring neurons in mouse visual cortex. Nature Neurosci. 13, 1144–1149 (2010)
Cossell, L. et al. Functional organization of excitatory synaptic strength in primary visual cortex. Nature 518, 399–403 (2015)
Chen, T. W. et al. Ultrasensitive fluorescent proteins for imaging neuronal activity. Nature 499, 295–300 (2013)
Pologruto, T. A., Sabatini, B. L. & Svoboda, K. ScanImage: flexible software for operating laser scanning microscopes. Biomed. Eng. Online 2, 13 (2003)
Sage, D., Prodanov, D., Tinevez, J.Y. & Schindelin, J. MIJ: Making interoperability between ImageJ and Matlab possible. in ImageJ User & Developer Conference, 24–26 October 2012, Luxembourg
Peirce, J. W. PsychoPy–Psychophysics software in Python. J. Neurosci. Methods 162, 8–13 (2007)
Seelig, J. D. & Jayaraman, V. Feature detection and orientation tuning in the Drosophila central complex. Nature 503, 262–266 (2013)
Vogelstein, J. T. et al. Fast nonnegative deconvolution for spike train inference from population calcium imaging. J. Neurophysiol. 104, 3691–3704 (2010)
Jones, J. P. & Palmer, L. A. An evaluation of the two-dimensional Gabor filter model of simple receptive fields in cat striate cortex. J. Neurophysiol. 58, 1233–1258 (1987)
Nauhaus, I. et al. Orthogonal micro-organization of orientation and spatial frequency in primate primary visual cortex. Nature Neurosci. 15, 1683–1690 (2012)
Huang, X. et al. Optogenetic assessment of horizontal interactions in primary visual cortex. J. Neurosci. 34, 4976–4990 (2014)
Swindale, N. V., Shoham, D., Grinvald, A., Bonhoeffer, T. & Hubener, M. Visual cortex maps are optimized for uniform coverage. Nature Neurosci. 3, 822–826 (2000)
Acknowledgements
We thank members of the Fitzpatrick laboratory for discussions and comments, D. Ouimet and K. Diah for animal technical support, T. Walker for technical assistance, A. Jacob for histology assistance, D. Wilson for advice on two-photon imaging, and D. Whitney for advice on epi-fluorescence imaging. This work was supported by grants from the US National Institutes of Health and funding from Max Planck Florida Institute for Neuroscience and Max Planck Society to D.F.
Author information
Authors and Affiliations
Contributions
K.-S.L. and D.F. designed the experiments. X.H. helped to collect data in the initial stage. K.-S.L performed all the experiments and analysed all the data with assistance from X.H. K.-S.L. and D.F. wrote the manuscript with input from X.H.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Robust receptive field estimation from GCaMP6 calcium signal in layer 2/3 neurons.
a, Somatic locations of seven example cells (circles) overlaid on the two-photon field of view. b, Raw calcium trace, spatiotemporal receptive field and SNR curves from an example cell in a. c–h, Six different ways to infer the onset time and response strength of neural activity were compared with the original method described in Methods for six of the example cells in a. Processed calcium trace before starting inference (blue) and the inferred response (red) are shown on the left. Receptive fields and SNR curves derived from original and alternative methods are shown on the right. i–l, Change in peak SNR (i) and peak time (j), receptive field similarity index (k) and deviation of the receptive field and subfield centre estimation (l) illustrating that the main conclusions regarding receptive field structure and fine visuotopic organization are not altered by the signal processing method employed (n = 143 cells from 3 animals). All error bars indicate s.e.m.
Extended Data Figure 2 Cell type categorization in tree shrew primary visual cortex layer 2/3.
a, Distribution of ON/OFF segregation index values for simple and complex cells (see Methods). A value of 0.6 was used to delineate the two classes. b, Distribution of ON/OFF ratio values for simple and complex cells. In both a and b, the proportions are based on the total number of cells; however, the single-sign cell population is not shown in the plots. c, Percentage of different classes of neurons in tree shrew visual cortex layer 2/3.
Extended Data Figure 3 Cortical spread of light- and dark-evoked activity in epi-fluorescence imaging.
a, Wide-field epi-fluorescence imaging of visual cortex reveals a similar visuotopic progression for the zones of activity found for static light and dark bar stimuli at different locations in elevation. b, The bandwidth of the normalized cortical activity pattern, characterized by half width at half maximum (HWHM), shows that light stimuli evoke broader cortical activity patterns than dark stimuli at the same visuotopic location (n = 21 stimulus-evoked response maps from 4 animals, P = 9.6 × 10−5, rank-sum test). Error bars indicate s.e.m.
Extended Data Figure 4 ON and OFF receptive field organization of single-sign cells.
a, The cortical volume and orientation map of an example imaging area. b, The ON and OFF centres from single-sign cells display an arrangement similar to that of simple cells. The bottom plot shows that the distribution pattern of ON and OFF receptive fields is consistent with that of the ON and OFF subfields of simple cells (n = 8 imaging areas from 7 animals, Kruskal–Wallis test; compare with Fig. 1c, d; letters indicate groups with statistically significant difference, P < 0.01). c, The visuotopic organization of ON and OFF receptive field centres was similar to that of simple cell ON and OFF subfields. d, The relationship between cortical distance and visuotopic position, demonstrating the difference in visuotopic precision for ON and OFF receptive fields (linear regression). Deviations of the experimental results from the linear fit and explained variance of the smooth visuotopy (n = 16 visuotopic maps, combining elevation and azimuth results from 8 imaging areas, **P < 0.0001, rank-sum test) are consistent with the results from simple cell ON- and OFF subfields. e, Only the displacement of the population ON receptive field centre, but not that of the OFF receptive field centre, can predict the orientation tuning of the orientation column (circular correlation, n = 68 cortical columns, P = 9.51 × 10−3 for ON; n = 89 cortical columns, P = 0.586 for OFF). All error bars indicate s.e.m.
Extended Data Figure 5 Visuotopic arrangement of ON and OFF subfields is independent of orientation map structure.
a, Example orientation map and local heterogeneity index map. The local heterogeneity index was used to compare ON and OFF subfield arrangement for cortical regions with different orientation map structure. a.u., arbitrary units. b, Top, illustration comparing the visuotopic displacement of OFF subfields to the theoretical prediction from a smooth visuotopic map. Bottom, illustration comparing the visuotopic displacement of ON subfields to the orientation map. c, Top, visuotopic distortion of OFF subfield centres in relation to the structure of the orientation map. There is no relationship between local heterogeneity and the visuotopic precision of OFF subfields (linear regression, n = 1,811 cells from 7 animals, P = 8.2 × 10−2). Bottom, axial mismatch of ON subfield centres in relation to the structure of the orientation map. There is no relationship between local heterogeneity and the axial displacement of ON subfield centres (linear regression, n = 1,811 cells from 7 animals, P = 9.6 × 10−2). d, Examples of the ON and OFF subfield centre distributions from 80-μm circular regions (black circles) centred on three distinct regions of the orientation map.
Extended Data Figure 6 Contribution of simple cells at different depths to aggregate receptive field of cortical column.
a, An example orientation column at four depths, with two-photon images on the left and the corresponding orientation maps on the right. b, Simple cell receptive fields from these four cortical depths. Each RF was normalized by the strongest subfield. The averages of the RFs within each depth appear similar. All the RFs within the orientation column were pooled into an aggregate receptive field (ARF) and then fitted with a 2D Gabor function. c, Nine further examples of ARFs from different orientation columns display the same organization: OFF subfield in the centre with ON subfields flanking on two sides.
Extended Data Figure 7 Characterizing spatial phase tuning, phase column and phase map.
a, The phase tuning of an example cell (black) and its Gaussian fit (red) compared with the phase tuning curve predicted from its receptive field structure (grey) and its Gaussian fit (yellow). Dashed line depicts the preferred phase derived from the Gaussian fit to the experimental data. b, Relationship between absolute phase prediction from receptive field structure and absolute phase tuning measurement (n = 179 cells from 2 animals, P = 1.8 × 10−18, circular regression). c, Phase preference of the orientation column is well predicted by the phase parameter of the Gabor fit to the ARF (n = 73 cortical columns from 5 animals, P = 1.7 × 10−10, circular regression). d, Example two-photon phase maps derived from pixel tuning at three cortical depths for both horizontal and vertical orientations. e, Comparison of phase preference from different cortical depths (red asterisks in d) showing the consistency of columnar structure for spatial phase (rank-sum test for R2 from circular regression, n = 36 pairs of maps at different depths from 2 animals, P = 8.2 × 10−18). f, Large-scale functional maps visualized by epi-fluorescence imaging. The phase map with full orientation coverage (right) was constructed from four individual phase maps measured independently with four orientations (0°, 45°, 90°, 135°). The phase maps for single orientations with corresponding visuotopic maps are shown separately in the lower two rows. g, The statistical structure of functional maps (orientation, phase, visuotopy, and phase with four orientations) summarized by the relationship between the change in cortical distance and the average change in preferred feature (left). Summary comparison of clustering and periodicity of the preferred features of four functional maps from six animals (right). Each map exhibits distinct clustering and periodicity (n = 32 sample regions from 6 animals, Kruskal–Wallis test with post-hoc test using Dunn’s method, letters indicate groups with statistically significant difference, P < 0.05). All error bars indicate s.e.m.
Extended Data Figure 8 Simulation based on experimental observations to evaluate completeness and uniformity of coverage for orientation and phase representations.
a, The large-scale orientation preference map derived from intrinsic signal imaging and corresponding phase map predicted from experimental observations (see Methods). b, Distribution of ON and OFF subfield centres in visual space predicted from the visuotopic precision and orientation-specific displacement demonstrated in this study. Although the distribution of the ON subfield centres in visual space appears uneven, complete coverage of visual space is achieved when the actual size of the ON subfields is considered (black circle). c, Illustration of two of the visual stimuli (8° stimulus in the centre, 0.5° stimulus to the left) used to simulate the evoked response map. d, Theoretical stimulus-evoked orientation and phase response maps for sample 0.5° stimulus shown in c (see Methods). e, Histograms showing the distribution of preferred orientation and phase values for pixels activated in d, calculated by counts of the pixels in the responsive region (left) or weighted by the strength of the responses (right). f, Theoretical stimulus-evoked orientation and phase response maps for sample 8° stimulus shown in c (see Methods). g, Histograms showing the distribution of preferred orientation and phase values for pixels activated in f, calculated by counts of the pixels in the responsive region (left) or weighted by the strength of the responses (right). h, Completeness (top) and uniformity (middle, bottom) of coverage simulated with visual stimuli of various sizes and positions. Complete coverage can be achieved with 1° stimuli, while coverage uniformity continues to improve with increases in stimulus size. The results of spatial phase were always the average results obtained with four different orientations. Error bars indicate s.e.m.
Supplementary information
Supplementary Data
This file contains the statistical details of experimental analysis. (PDF 91 kb)
Rights and permissions
About this article
Cite this article
Lee, KS., Huang, X. & Fitzpatrick, D. Topology of ON and OFF inputs in visual cortex enables an invariant columnar architecture. Nature 533, 90–94 (2016). https://doi.org/10.1038/nature17941
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature17941
This article is cited by
-
The Cousa objective: a long-working distance air objective for multiphoton imaging in vivo
Nature Methods (2024)
-
A theory of cortical map formation in the visual brain
Nature Communications (2022)
-
ON/OFF domains shape receptive field structure in mouse visual cortex
Nature Communications (2022)
-
Mechanism underpinning the sharpening of orientation and spatial frequency selectivities in the tree shrew (Tupaia belangeri) primary visual cortex
Brain Structure and Function (2022)
-
Inter-mosaic coordination of retinal receptive fields
Nature (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.