Abstract
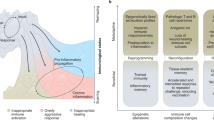
Our current understanding of immunology was largely defined in laboratory mice, partly because they are inbred and genetically homogeneous, can be genetically manipulated, allow kinetic tissue analyses to be carried out from the onset of disease, and permit the use of tractable disease models. Comparably reductionist experiments are neither technically nor ethically possible in humans. However, there is growing concern that laboratory mice do not reflect relevant aspects of the human immune system, which may account for failures to translate disease treatments from bench to bedside1,2,3,4,5,6,7,8. Laboratory mice live in abnormally hygienic specific pathogen free (SPF) barrier facilities. Here we show that standard laboratory mouse husbandry has profound effects on the immune system and that environmental changes produce mice with immune systems closer to those of adult humans. Laboratory mice—like newborn, but not adult, humans—lack effector-differentiated and mucosally distributed memory T cells. These cell populations were present in free-living barn populations of feral mice and pet store mice with diverse microbial experience, and were induced in laboratory mice after co-housing with pet store mice, suggesting that the environment is involved in the induction of these cells. Altering the living conditions of mice profoundly affected the cellular composition of the innate and adaptive immune systems, resulted in global changes in blood cell gene expression to patterns that more closely reflected the immune signatures of adult humans rather than neonates, altered resistance to infection, and influenced T-cell differentiation in response to a de novo viral infection. These data highlight the effects of environment on the basal immune state and response to infection and suggest that restoring physiological microbial exposure in laboratory mice could provide a relevant tool for modelling immunological events in free-living organisms, including humans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Mestas, J. & Hughes, C. C. W. Of mice and not men: differences between mouse and human immunology. J. Immunol. 172, 2731–2738 (2004)
Seok, J. et al. Genomic responses in mouse models poorly mimic human inflammatory diseases. Proc. Natl Acad. Sci. USA 110, 3507–3512 (2013)
Shay, T. et al. Conservation and divergence in the transcriptional programs of the human and mouse immune systems. Proc. Natl Acad. Sci. USA 110, 2946–2951 (2013)
Mak, I. W., Evaniew, N. & Ghert, M. Lost in translation: animal models and clinical trials in cancer treatment. Am. J. Transl. Res. 6, 114–118 (2014)
Rivera, J. & Tessarollo, L. Genetic background and the dilemma of translating mouse studies to humans. Immunity 28, 1–4 (2008)
Payne, K. J. & Crooks, G. M. Immune-cell lineage commitment: translation from mice to humans. Immunity 26, 674–677 (2007)
von Herrath, M. G. & Nepom, G. T. Lost in translation: barriers to implementing clinical immunotherapeutics for autoimmunity. J. Exp. Med. 202, 1159–1162 (2005)
Takao, K. & Miyakawa, T. Genomic responses in mouse models greatly mimic human inflammatory diseases. Proc. Natl Acad. Sci. USA 112, 1167–1172 (2015)
Schenkel, J. M. & Masopust, D. Tissue-resident memory T cells. Immunity 41, 886–897 (2014)
Thome, J. J. C. et al. Early-life compartmentalization of human T cell differentiation and regulatory function in mucosal and lymphoid tissues. Nature Med. (2016)
Thome, J. J. C. et al. Spatial map of human T cell compartmentalization and maintenance over decades of life. Cell 159, 814–828 (2014)
Machado, C. S., Rodrigues, M. A. & Maffei, H. V. Gut intraepithelial lymphocyte counts in neonates, infants and children. Acta Paediatr. 83, 1264–1267 (1994)
Sallusto, F., Geginat, J. & Lanzavecchia, A. Central memory and effector memory T cell subsets: function, generation, and maintenance. Annu. Rev. Immunol. 22, 745–763 (2004)
Olson, J. A., McDonald-Hyman, C., Jameson, S. C. & Hamilton, S. E. Effector-like CD8+ T cells in the memory population mediate potent protective immunity. Immunity 38, 1250–1260 (2013)
Mouse Genome Sequencing Consortium Initial sequencing and comparative analysis of the mouse genome. Nature 420, 520–562 (2002)
Pritchett-Corning, K. R., Cosentino, J. & Clifford, C. B. Contemporary prevalence of infectious agents in laboratory mice and rats. Lab. Anim. 43, 165–173 (2009)
Pedersen, A. B. & Babayan, S. A. Wild immunology. Mol. Ecol. 20, 872–880 (2011)
Maizels, R. M. & Nussey, D. H. Into the wild: digging at immunology’s evolutionary roots. Nature Immunol. 14, 879–883 (2013)
Cadwell, K. The virome in host health and disease. Immunity 42, 805–813 (2015)
Virgin, H. W., Wherry, E. J. & Ahmed, R. Redefining chronic viral infection. Cell 138, 30–50 (2009)
Votavova, H. et al. Transcriptome alterations in maternal and fetal cells induced by tobacco smoke. Placenta 32, 763–770 (2011)
Godec, J. et al. Compendium of immune signatures identifies conserved and species-specific biology in response to inflammation. Immunity 44, 194–206 (2016)
Kaech, S. M. et al. Selective expression of the interleukin 7 receptor identifies effector CD8 T cells that give rise to long-lived memory cells. Nature Immunol. 4, 1191–1198 (2003)
Joshi, N. S. et al. Inflammation directs memory precursor and short-lived effector CD8+ T cell fates via the graded expression of T-bet transcription factor. Immunity 27, 281–295 (2007)
Jordan, M. B., Hildeman, D., Kappler, J. & Marrack, P. An animal model of hemophagocytic lymphohistiocytosis (HLH): CD8+ T cells and interferon gamma are essential for the disorder. Blood 104, 735–743 (2004)
Selin, L. K. et al. Memory of mice and men: CD8+ T-cell cross-reactivity and heterologous immunity. Immunol. Rev. 211, 164–181 (2006)
Sun, J. C., Ugolini, S. & Vivier, E. Immunological memory within the innate immune system. EMBO J. 33, 1295–1303 (2014)
Adams, A. B., Pearson, T. C. & Larsen, C. P. Heterologous immunity: an overlooked barrier to tolerance. Immunol. Rev. 196, 147–160 (2003)
Taurog, J. D. et al. The germfree state prevents development of gut and joint inflammatory disease in HLA-B27 transgenic rats. J. Exp. Med. 180, 2359–2364 (1994)
Pozzilli, P., Signore, A., Williams, A. J. K. & Beales, P. E. NOD mouse colonies around the world- recent facts and figures. Immunol. Today 14, 193–196 (1993)
Hamilton, S. E., Wolkers, M. C., Schoenberger, S. P. & Jameson, S. C. The generation of protective memory-like CD8+ T cells during homeostatic proliferation requires CD4+ T cells. Nature Immunol. 7, 475–481 (2006)
Hamilton, S. E., Schenkel, J. M., Akue, A. D. & Jameson, S. C. IL-2 complex treatment can protect naive mice from bacterial and viral infection. J. Immunol. 1950, 6584–6590 (2010)
Pope, C. et al. Organ-specific regulation of the CD8 T cell response to Listeria monocytogenes infection. J. Immunol. 1950, 3402–3409 (2001)
Gordon, E. B. et al. Inhibiting the Mammalian target of rapamycin blocks the development of experimental cerebral malaria. MBio 6, e00725 (2015)
Balfour, H. H. et al. Behavioral, virologic, and immunologic factors associated with acquisition and severity of primary Epstein-Barr virus infection in university students. J. Infect. Dis. 207, 80–88 (2013)
Odumade, O. A. et al. Primary Epstein-Barr virus infection does not erode preexisting CD8+ T cell memory in humans. J. Exp. Med. 209, 471–478 (2012)
Anderson, K. G. et al. Intravascular staining for discrimination of vascular and tissue leukocytes. Nature Protocols 9, 209–222 (2014)
Beura, L. K. et al. Lymphocytic choriomeningitis virus persistence promotes effector-like memory differentiation and enhances mucosal T cell distribution. J. Leukoc. Biol. 97, 217–225 (2015)
Schenkel, J. M., Fraser, K. A., Vezys, V. & Masopust, D. Sensing and alarm function of resident memory CD8+ T cells. Nature Immunol. 14, 509–513 (2013)
Steinert, E. M. et al. Quantifying memory CD8 T cells reveals regionalization of immunosurveillance. Cell 161, 737–749 (2015)
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl Acad. Sci. USA 102, 15545–15550 (2005)
Eden, E., Navon, R., Steinfeld, I., Lipson, D. & Yakhini, Z. GOrilla: a tool for discovery and visualization of enriched GO terms in ranked gene lists. BMC Bioinformatics 10, 48 (2009)
Acknowledgements
This study was supported by National Institutes of Health grants 1R01AI111671, R01AI084913 (to D.M.), R01AI116678, R01AI075168 (to S.C.J.) and a BSL-3 suite rental waiver grant from the University of Minnesota. We thank R. Ahmed for providing reagents for pilot studies, P. Southern and D. McKenna for tissue samples or cord blood, and all members of the BSL-3 mouse team (University of Minnesota).
Author information
Authors and Affiliations
Contributions
L.K.B., S.E.H., J.M.S., O.A.O., K.A.C., E.A.T., K.A.F, P.C.R, V.V., and D.M. performed the experiments and analysed the data. K.B. and W.N.H. analysed the transcriptome data. M.K.J., A.F.-M., and R.P.S. provided input on research design. L.K.B., S.E.H., W.N.H., S.C.J., and D.M. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Frequency of CD8+ T-cell subsets in newborn versus adult humans.
CD8+ T-cell subsets were defined in adult PBMCs (n = 13) and cord blood PBMCs (n = 8) by fluorescence flow cytometry based on the following markers: naive, CD45RAhiCCR7hi; TCM, CD45RAloCCR7hi; TEM, CD45RAloCCR7lo; TEMRA, CD45RAhiCCR7lo. Significance was determined using unpaired two-sided t-test. ***P < 0.001, ****P < 0.0001; error bars indicate mean ± s.e.m.
Extended Data Figure 2 Co-housing laboratory mice with pet store mice induces accumulation of TRM-phenotype CD8+ T cells and other innate cells in tissues of laboratory mice.
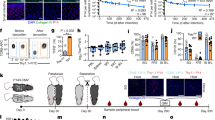
a, CD8+ T-cell density within the indicated tissues of adult laboratory mice (n = 5) and co-housed mice (n = 7). Representative immunofluorescence staining, CD8β (red), DAPI (nuclei, blue); scale bars, 50 μm. b, Phenotype of CD8+ T cells was compared between laboratory mice (n = 9) and age-matched laboratory mice that were co-housed (n = 9, representative flow cytometry plots shown). Samples gated on CD44hi cells isolated from the indicated tissue (vasculature populations were excluded, see Methods). c, Enumeration of CD11b+ granulocytes and Ly6Chi inflammatory monocytes in spleens of laboratory (n = 6) and co-housed (n = 6) mice. Significance was determined using unpaired two-sided Mann–Whitney U-test. **P < 0.01; error bars indicate mean ± s.e.m.
Extended Data Figure 3 LEM metagene analysis.
For each comparison, standard GSEA was performed using the ImmSigDB database of gene-sets. Genes in the top 150 enriched sets (FDR < 0.001, ranked by P value) were filtered to only leading edge genes and subsequently clustered into groups (metagenes) using an NMF algorithm. Hierarchical clustering of genes within individual metagenes was performed to obtain the final heatmap. Metagenes with qualitatively discernible ‘blocks’ of gene-set membership were annotated according to the identity of corresponding enriched gene-sets. Heatmaps for adult versus neonatal, pet store versus laboratory, co-housed versus laboratory, neonatal versus adult, laboratory versus pet store, and laboratory versus co-housed comparisons are shown. Individual genes within each metagene are listed in Supplementary Table 1. Pairwise overlaps between metagenes from different comparisons are visualized in Fig. 4c.
Extended Data Figure 4 Environment altered antimicrobial resistance and CD8+ T-cell differentiation.
Laboratory mice were co-housed with pet store mice as described in Figure 3. a, Bacterial load in the spleen 3 days after challenge with 8.5 × 104 c.f.u. of L. monocytogenes (LM) in laboratory (n = 8), LM-immune (n = 9), co-housed (n = 9) and pet store mice (n = 9) in two independent experiments. b, Survival of laboratory mice (n = 15), co-housed mice (n = 19) and pet store mice (n = 15) after challenge with 106 P. berghei ANKA parasitized RBCs in two independent experiments. c, Laboratory (n = 9) and co-housed (n = 8) mice were infected with LCMV. Four weeks later, LCMV-specific CD8+ T cells (identified with H-2Db/gp33 MHC I tetramers) were evaluated for expression of the indicated markers. Top row, gated on live CD8α+ T cells. Bottom three rows, gated on live CD8α+ H-2Db/gp33+ T cells. Significance was determined using Kruskal–Wallis (ANOVA) test (a) and log-rank (Mantel–Cox) test (b). *P < 0.05, ***P < 0.001, ****P < 0.0001; error bars indicate mean ± s.e.m.
Supplementary information
Supplementary Table 1
Genes belonging to all indicated metagenes identified are shown in this table. (XLSX 81 kb)
Supplementary Table 2
Adult vs. Neonatal metagene 1, Petstore vs. Laboratory metagene 2, and Cohoused vs. Laboratory metagene 1 are three metagenes exhibiting consistent and conserved enrichment of type-I interferon response-related GO terms. Genes are separated into columns depending on whether they belong uniquely to a single metagene, are shared between two specific metagenes, or shared between all three type-I interferon response-related metagenes. (XLSX 33 kb)
Rights and permissions
About this article
Cite this article
Beura, L., Hamilton, S., Bi, K. et al. Normalizing the environment recapitulates adult human immune traits in laboratory mice. Nature 532, 512–516 (2016). https://doi.org/10.1038/nature17655
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature17655
This article is cited by
-
Phosphorylation of insulin receptor substrates (IRS-1 and IRS-2) is attenuated following cecal ligation and puncture in mice
Molecular Medicine (2023)
-
Comparison of the effect of autoclaved and non-autoclaved live soil exposure on the mouse immune system
BMC Immunology (2023)
-
GPR43 stimulation on TCRαβ+ intraepithelial colonic lymphocytes inhibits the recruitment of encephalitogenic T-cells into the central nervous system and attenuates the development of autoimmunity
Journal of Neuroinflammation (2023)
-
Microbial experience through housing in a farmyard-type environment alters intestinal barrier properties in mouse colons
Scientific Reports (2023)
-
Advancing translational research for colorectal immuno-oncology
British Journal of Cancer (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.