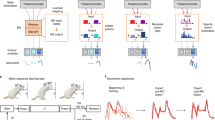
Abstract
For goal-directed behaviour it is critical that we can both select the appropriate action and learn to modify the underlying movements (for example, the pitch of a note or velocity of a reach) to improve outcomes. The basal ganglia are a critical nexus where circuits necessary for the production of behaviour, such as the neocortex and thalamus, are integrated with reward signalling1 to reinforce successful, purposive actions2. The dorsal striatum, a major input structure of basal ganglia, is composed of two opponent pathways, direct and indirect, thought to select actions that elicit positive outcomes and suppress actions that do not, respectively3,4. Activity-dependent plasticity modulated by reward is thought to be sufficient for selecting actions in the striatum5,6. Although perturbations of basal ganglia function produce profound changes in movement7, it remains unknown whether activity-dependent plasticity is sufficient to produce learned changes in movement kinematics, such as velocity. Here we use cell-type-specific stimulation in mice delivered in closed loop during movement to demonstrate that activity in either the direct or indirect pathway is sufficient to produce specific and sustained increases or decreases in velocity, without affecting action selection or motivation. These behavioural changes were a form of learning that accumulated over trials, persisted after the cessation of stimulation, and were abolished in the presence of dopamine antagonists. Our results reveal that the direct and indirect pathways can each bidirectionally control movement velocity, demonstrating unprecedented specificity and flexibility in the control of volition by the basal ganglia.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Dudman, J. T. & Gerfen, C. R. in The Rat Nervous System (ed. G Paxinos ) Ch. 17, (Elsevier, 2015)
Balleine, B. W., Liljeholm, M. & Ostlund, S. B. The integrative function of the basal ganglia in instrumental conditioning. Behav. Brain Res. 199, 43–52 (2009)
Mink, J. W. The basal ganglia: focused selection and inhibition of competing motor programs. Prog. Neurobiol. 50, 381–425 (1996)
Frank, M. J. Computational models of motivated action selection in corticostriatal circuits. Curr. Opin. Neurobiol. 21, 381–386 (2011)
Gurney, K. N., Humphries, M. D. & Redgrave, P. A new framework for cortico-striatal plasticity: behavioural theory meets in vitro data at the reinforcement-action interface. PLoS Biol. 13, e1002034 (2015)
Schultz, W. Behavioral theories and the neurophysiology of reward. Annu. Rev. Psychol. 57, 87–115 (2006)
Desmurget, M. & Turner, R. S. Motor sequences and the basal ganglia: kinematics, not habits. J. Neurosci. 30, 7685–7690 (2010)
Tumer, E. C. & Brainard, M. S. Performance variability enables adaptive plasticity of ‘crystallized’ adult birdsong. Nature 450, 1240–1244 (2007)
Andalman, A. S. & Fee, M. S. A basal ganglia-forebrain circuit in the songbird biases motor output to avoid vocal errors. Proc. Natl Acad. Sci. USA 106, 12518–12523 (2009)
Gerfen, C. R. et al. D1 and D2 dopamine receptor-regulated gene expression of striatonigral and striatopallidal neurons. Science 250, 1429–1432 (1990)
Kravitz, A. V. et al. Regulation of parkinsonian motor behaviours by optogenetic control of basal ganglia circuitry. Nature 466, 622–626 (2010)
Collins, A. G. & Frank, M. J. Opponent actor learning (OpAL): modeling interactive effects of striatal dopamine on reinforcement learning and choice incentive. Psychol. Rev. 121, 337–366 (2014)
Kravitz, A. V., Tye, L. D. & Kreitzer, A. C. Distinct roles for direct and indirect pathway striatal neurons in reinforcement. Nature Neurosci. 15, 816–818 (2012)
Tai, L. H., Lee, A. M., Benavidez, N., Bonci, A. & Wilbrecht, L. Transient stimulation of distinct subpopulations of striatal neurons mimics changes in action value. Nature Neurosci. 15, 1281–1289 (2012)
Niv, Y., Daw, N. D., Joel, D. & Dayan, P. Tonic dopamine: opportunity costs and the control of response vigor. Psychopharmacology (Berl.) 191, 507–520 (2007)
Sutton, R. S. & Barto, A. G. Reinforcement Learning: an Introduction. (MIT Press, 1998)
Yin, H. H., Ostlund, S. B., Knowlton, B. J. & Balleine, B. W. in Eur. J. Neurosci. 22, 513–523 (2005)
Paninski, L., Fellows, M. R., Hatsopoulos, N. G. & Donoghue, J. P. Spatiotemporal tuning of motor cortical neurons for hand position and velocity. J. Neurophysiol. 91, 515–532 (2004)
Churchland, M. M. & Shenoy, K. V. Temporal complexity and heterogeneity of single-neuron activity in premotor and motor cortex. J. Neurophysiol. 97, 4235–4257 (2007)
Moran, D. W. & Schwartz, A. B. Motor cortical representation of speed and direction during reaching. J. Neurophysiol. 82, 2676–2692 (1999)
Panigrahi, B. et al. Dopamine is required for the neural representation and control of movement vigor. Cell 162, 1418–1430 (2015)
Pawlak, V. & Kerr, J. N. Dopamine receptor activation is required for corticostriatal spike-timing-dependent plasticity. J. Neurosci. 28, 2435–2446 (2008)
Shen, W., Flajolet, M., Greengard, P. & Surmeier, D. J. Dichotomous dopaminergic control of striatal synaptic plasticity. Science 321, 848–851 (2008)
Cooper, L. N. & Bear, M. F. The BCM theory of synapse modification at 30: interaction of theory with experiment. Nature Rev. Neurosci. 13, 798–810 (2012)
Izhikevich, E. M. & Desai, N. S. Relating STDP to BCM. Neural Comput. 15, 1511–1523 (2003)
Turner, R. S. & Desmurget, M. Basal ganglia contributions to motor control: a vigorous tutor. Curr. Opin. Neurobiol. 20, 704–716 (2010)
Wall, N. R., De La Parra, M., Callaway, E. M. & Kreitzer, A. C. Differential innervation of direct- and indirect-pathway striatal projection neurons. Neuron 79, 347–360 (2013)
Mazzoni, P., Hristova, A. & Krakauer, J. W. Why don’t we move faster? Parkinson’s disease, movement vigor, and implicit motivation. J. Neurosci. 27, 7105–7116 (2007)
Phillips, A. G. & Fibiger, H. C. The role of dopamine in maintaining intracranial self-stimulation in the ventral tegmentum, nucleus accumbens, and medial prefrontal cortex. Can. J. Psychol. 32, 58–66 (1978)
Gallistel, C. R. & Gibbon, J. Time, rate, and conditioning. Psychol. Rev. 107, 289–344 (2000)
Gerfen, C. R., Paletzki, R. & Heintz, N. GENSAT BAC Cre-recombinase driver lines to study the functional organization of cerebral cortical and basal ganglia circuits. Neuron 80, 1368–1383 (2013)
Gong, S. et al. Targeting Cre recombinase to specific neuron populations with bacterial artificial chromosome constructs. J. Neurosci. 27, 9817–9823 (2007)
Madisen, L. et al. A toolbox of Cre-dependent optogenetic transgenic mice for light-induced activation and silencing. Nature Neurosci. 15, 793–802 (2012)
Deacon, R. M. Measuring the strength of mice. J. Vis. Exp. (2013)
Osborne, J. E. & Dudman, J. T. RIVETS: a mechanical system for in vivo and in vitro electrophysiology and imaging. PLoS ONE 9, e89007 (2014)
Pan, W. X., Mao, T. & Dudman, J. T. Inputs to the dorsal striatum of the mouse reflect the parallel circuit architecture of the forebrain. Front. Neuroanat. 4, 147 (2010)
Paxinos, G. & Franklin, K. The Mouse Brain in Stereotaxic Coordinates. (Gulf Professional Publishing, 2004)
Tennant, K. A. et al. The organization of the forelimb representation of the C57BL/6 mouse motor cortex as defined by intracortical microstimulation and cytoarchitecture. Cereb. Cortex 21, 865–876 (2011)
Stujenske, J. M., Spellman, T. & Gordon, J. A. Modeling the spatiotemporal dynamics of light and heat propagation for in vivo optogenetics. Cell Rep. 12, 525–534 (2015)
Brown, J., Pan, W. X. & Dudman, J. T. The inhibitory microcircuit of the substantia nigra provides feedback gain control of the basal ganglia output. Elife 3, e02397 (2014)
Acknowledgements
This work was supported by funding from the Howard Hughes Medical Institute. J.T.D. is a Group Leader at Janelia Research Campus. We thank A. Lee, A. Karpova, N. Spruston, and members of our laboratory for critical reading and feedback on the manuscript. We also thank M. Frank for helpful discussions of the OpAL model.
Author information
Authors and Affiliations
Contributions
E.A.Y. performed the experiments and analysed the data. E.A.Y. and J.T.D. designed the experiments, performed the modelling, and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
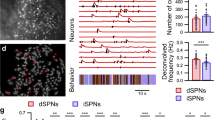
Extended Data Figure 1 Anatomical localization of stimulated neurons and their corticostriatal inputs.
a, Schematic showing the histological reconstruction of optic fibre endpoints in dMSN (cyan) and iMSN (red) mice. Hemispheric segregation for display purposes only; actual implants were randomized left and right. b, Location of retrobead tracer injection just below the coordinates of fibre implant terminus. c, Location of the labelled projection neurons innervating the retrobead injection site corresponding to the rostral forelimb area (RFA, orange) and caudal forelimb area (CFA, yellow). d, Results of simulation of light intensity and spread through the brain based upon the model of Stujenske et al.39. Light intensity drops below 10% peak intensity 285 μm below the fibre. At this depth the lateral fall-off is a drop to 1% peak intensity 250 μm. Contour lines for 10% intensity and 1% intensity are overlaid in white and grey, respectively. e, Average photostimulation-evoked activity of individual neurons (each row = 1 neuron) sorted by electrode shank. We observed minimal fidelity in the light-evoked responses of neurons on the farthest shank consistent with the predicted fall-off in light intensity from simulations. Blue dashes at top identify timing of laser pulses. Robust entrainment of spiking to photostimulation was also observed in vitro (Extended Data Fig. 7).
Extended Data Figure 2 Selective stimulation of MSNs produces changes in peak velocity.
a, Density plot of movement trajectories showing the percentage of movements that passed through a given amplitude throughout time, aligned to movement onset. Particularly in the magnified plot on the right, we can see that very few movement trajectories passed near the central 0.2 cm within the first 100 ms. b, Peak velocity distributions for sham and stimulation data sets for dMSN stimulation (blue, left column), and iMSN stimulation (red, middle column). Top row shows complete distributions across animals. Note that no particular part of the distribution is pronounced following stimulation. Bottom row shows means for all experimental sessions by animal. Animal number is for indexing purposes only. Most experiments were carried out concurrently across animals. The right column shows the contrast ratio (difference divided by sum) for dMSN and iMSN stimulation effects (blue and red, respectively). These data show a steady mean shift in the data; for instance, iMSN stimulation (red) is positive for velocities slower than the sham (control) mean (an increase in frequency), and is negative for velocity values greater than the sham mean (a decrease in frequency). We have curtailed the contrast ratio plot where too few values existed to achieve reliable estimates (55 cm s−1). c, Autocorrelation of movement velocities for dMSN stim (blue), iMSN (red), and sham (black) data. d, e, Same analyses as b, c, but for those sessions in which stimulation occurred on the slower, lower third of movement velocities.
Extended Data Figure 3 Trained animals can adjust amplitude to changing task requirements.
a, Reach amplitudes from a sample of seven sessions in two mice in which the eccentricity of the threshold to receive reward was suddenly increased at random. Green field identifies reaches performed with the increased amplitude threshold. Shaded area represents standard error of the mean. b, Success rate before (black) and after (green) the jump in amplitude threshold. c, Distribution of reach amplitudes across sessions for pre- (green) and post- (black) amplitude threshold jump. These data indicate that the mean amplitude was not saturated and suggest that behaviour remains outcome-dependent (that is, goal-directed).
Extended Data Figure 4 Variance of movement velocity does not change throughout a session.
Each session was z-scored and the standard deviation for each movement number is plotted for dMSN (cyan) and iMSN (red) stimulation.
Extended Data Figure 5 Non-selective stimulation does not affect motor control or initiation.
a, b, ‘All-stim’ (a; 15 stimulation and 17 sham sessions from 4 dMSN mice; 17 stimulation and 20 sham sessions from 4 iMSN mice) and ‘random stim’ (b; 11 stimulation and 15 sham dMSN sessions from 3 dMSN mice, 8 stimulation and 12 sham sessions from 3 iMSN mice) summary data showing (top) the mean velocity as a function of movement number for dMSN (cyan) and iMSN (red) stimulation sessions and (bottom) histograms of the inter-move interval (IMI; left) and lick rate (LR; right). Shaded area indicates standard error of the mean. We found no differences between sham (black lines) and stimulation (coloured lines) sessions for either dMSN stimulation (cyan) or iMSN stimulation (red). c, Plot of average trajectory position aligned to stimulation onset for random dMSN (top) and iMSN stimulation (middle) for stimulation (coloured) and sham (black) sessions (in sham sessions, timing was randomly chosen, but no stimulation was given). Stimulation did not systematically induce forelimb movement. For reference, the bottom trace displays closed-loop dMSNs aligned to stimulation onset. Grey field represents the 450 ms stimulation period.
Extended Data Figure 6 A corticostriatal circuit model that implements the MeSH learning rule.
a, In the left panel, we present a schematic of the corticostriatal pathway consistent with known anatomical data1. Descending cortical outputs, largely from layer V of the neocortex, project subcortically and intracortically elaborating axon collaterals onto direct (blue) and indirect (red) MSNs in the dorsal striatum. By typical convention we assume that dMSNs have a net positive effect on behaviour (increase in velocity in this case) and iMSNs have a net inhibitory effect (decrease in velocity). These pathways are combined at the basal ganglia output nucleus (substantia nigra pars reticulata; not shown) and then combined with cortical drive to produce the net movement velocity. The model assumes that both dMSNs and iMSNs are positively correlated with cortical activity and with movement velocity. We assume a monotonic relationship between cortical activity and movement velocity20. The model is initialized at a presumptive steady-state in which weights between cortical inputs and dMSN and iMSN units are noisy, but distributed around 0.5 and bounded [0,1]. Most simulations were performed with 100 cortical units and 250 dMSNs and iMSNs each. Under all conditions weights are subject to updating according to a balanced plasticity rule (inset) in which inactive units are subject to depression and active units are subject to potentiation. All synapses drift back towards a mean of 0.5 thereby implementing a homeostatic set point to the weight distribution. Finally, photostimulation is assumed to enhance (90% increase) the magnitude of both depression and potentiation on stimulated trials in the stimulated population. Random sets of cortical inputs are assumed to be active on any given reach and are drawn from a gamma (shape parameters: 8, 63) distribution that gives a distribution of velocities similar to that observed experimentally. Further details of the model are provided in the Methods. b, Example simulations of 100 trials (first 50 receive stimulation according to conditions described in legend followed by 50 unstimulated recovery trials). Curves reflect averages and standard errors of 100 repetitions of the simulated condition. Other conventions as in main figures. c, Schematic of dendrite of MSN containing synapses active during limb movements. Synaptic plasticity enhanced by stimulation (inset in panel a) produces a net bias in synaptic weights when delivered in closed-loop. This bias can become uniform by permuting the active synapses on each simulated trial.
Extended Data Figure 7 D1+ and D2+ MSNs can follow repetitive stimulus trains of ~20 Hz photostimulation in vitro.
Example MSNs recorded in vitro in brain slices containing the DMS. Upper row are two example D1+ dMSNs recorded from DMS of a Drd1a-Cre::Ai32 mouse. Lower row are two example D2+ dMSN recorded from DMS of a Drd2a-Cre::Ai32 mouse. 3 example traces shown from each. Spiking is evoked by increasing current injection (traces selected for approximately similar evoked spiking rate) and ~500 ms later by a train of 5 pulses of blue light of increasing duty cycle. All cells recorded were able to follow rapid phasic stimulation and action potentials were reliably evoked on every stimulus of these 20 Hz trains (approximately similar to the stimulus trains used in stimulation experiments described in the text). Examples were selected from seven neurons from three D1+ animals, and four neurons from three D2+ animals that were recorded for this particular stimulation design.
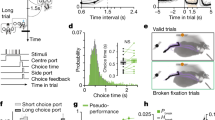
Extended Data Figure 8 Characterization of movement onset and reach initiation threshold crossing time.
A sample reach from a sham block of 50 is shown, with eccentricity in black and velocity in blue (in right panel). The reach start is identified with the greed dot, the threshold crossing, 9 ms later when the reach eccentricity surpassed 0.1 cm, is identified with the magenta dot. The beginning of the reach (green) was assessed offline for each reach and was determined to be the first time point, sampled at 1 kHz, constituting the increasing velocity associated with that reach.
Supplementary information
Supplementary Information
This file contains a Supplementary Discussion and additional references. (PDF 1113 kb)
dMSN upper third stimulation
Infrared video recording of a Drd1a-cre::Ai32 mouse performing task while head-fixed during stimulation session for upper 1/3 stimulation protocols. Black line indicates eccentricity. Green line indicates outward velocity. Blue bar indicates period of stimulation. Video angle may cause joystick to appear closer to mouse platform than it is (2.7cm away). (MOV 20331 kb)
iMSN upper third stimulation
Infrared video recording of a Drd2-cre::Ai32 mouse performing task while head-fixed during stimulation session for upper 1/3 stimulation protocols. Black line indicates eccentricity. Green line indicates outward velocity. Blue bar indicates period of stimulation. (MOV 22144 kb)
dMSN lower third stimulation
Infrared video recording of a Drd1a-cre::Ai32 mouse performing task while head-fixed during stimulation session for lower 1/3 stimulation protocols. Black line indicates eccentricity. Green line indicates outward velocity. Blue bar indicates period of stimulation. (MOV 20353 kb)
iMSN lower third stimulation
Infrared video recording of a Drd2-cre::Ai32 mouse performing task while head-fixed during stimulation session for lower 1/3 stimulation protocols. Black line indicates eccentricity. Green line indicates outward velocity. Blue bar indicates period of stimulation. (MOV 21403 kb)
Rights and permissions
About this article
Cite this article
Yttri, E., Dudman, J. Opponent and bidirectional control of movement velocity in the basal ganglia. Nature 533, 402–406 (2016). https://doi.org/10.1038/nature17639
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature17639
This article is cited by
-
The respective activation and silencing of striatal direct and indirect pathway neurons support behavior encoding
Nature Communications (2023)
-
Cilia in the Striatum Mediate Timing-Dependent Functions
Molecular Neurobiology (2023)
-
Locomotion activates PKA through dopamine and adenosine in striatal neurons
Nature (2022)
-
Reinforcement-learning in fronto-striatal circuits
Neuropsychopharmacology (2022)
-
Opponent control of behavior by dorsomedial striatal pathways depends on task demands and internal state
Nature Neuroscience (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.