Abstract
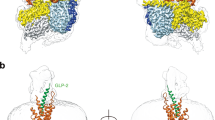
Glucagon is a 29-amino-acid peptide released from the α-cells of the islet of Langerhans, which has a key role in glucose homeostasis1. Glucagon action is transduced by the class B G-protein-coupled glucagon receptor (GCGR), which is located on liver, kidney, intestinal smooth muscle, brain, adipose tissue, heart and pancreas cells, and this receptor has been considered an important drug target in the treatment of diabetes. Administration of recently identified small-molecule GCGR antagonists in patients with type 2 diabetes results in a substantial reduction of fasting and postprandial glucose concentrations2. Although an X-ray structure of the transmembrane domain of the GCGR3 has previously been solved, the ligand (NNC0640) was not resolved. Here we report the 2.5 Å structure of human GCGR in complex with the antagonist MK-0893 (ref. 4), which is found to bind to an allosteric site outside the seven transmembrane (7TM) helical bundle in a position between TM6 and TM7 extending into the lipid bilayer. Mutagenesis of key residues identified in the X-ray structure confirms their role in the binding of MK-0893 to the receptor. The unexpected position of the binding site for MK-0893, which is structurally similar to other GCGR antagonists, suggests that glucagon activation of the receptor is prevented by restriction of the outward helical movement of TM6 required for G-protein coupling. Structural knowledge of class B receptors is limited, with only one other ligand-binding site defined—for the corticotropin-releasing hormone receptor 1 (CRF1R)—which was located deep within the 7TM bundle5. We describe a completely novel allosteric binding site for class B receptors, providing an opportunity for structure-based drug design for this receptor class and furthering our understanding of the mechanisms of activation of these receptors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
12 May 2016
The MK-0893 structure in Fig. 1b was corrected.
References
Ahrén, B. Glucagon—early breakthroughs and recent discoveries. Peptides 67, 74–81 (2015)
Bagger, J. I., Knop, F. K., Holst, J. J. & Vilsbøll, T. Glucagon antagonism as a potential therapeutic target in type 2 diabetes. Diabetes Obes. Metab. 13, 965–971 (2011)
Siu, F. Y et al. Structure of the human glucagon class B G-protein-coupled receptor. Nature 499, 444–449 (2013)
Xiong, Y. et al. Discovery of a novel glucagon receptor antagonist N-[(4-(1S)-1-[3-(3, 5-dichlorophenyl)-5-(6-methoxynaphthalen-2-yl)-1H-pyrazol-1-yl]ethylphenyl)carbonyl]-β-alanine (MK-0893) for the treatment of type II diabetes. J. Med. Chem. 55, 6137–6148 (2012)
Hollenstein, K. et al. Structure of class B GPCR corticotropin-releasing factor receptor 1. Nature 499, 438–443 (2013)
Serrano-Vega, M. J., Magnani, F., Shibata, Y. & Tate, C. G. Conformational thermostabilization of the β1-adrenergic receptor in a detergent-resistant form. Proc. Natl Acad. Sci. USA 105, 877–882 (2008)
Shibata, Y. et al. Thermostabilization of the neurotensin receptor NTS1. J. Mol. Biol. 390, 262–277 (2009)
Robertson, N. et al. The properties of thermostabilised G protein-coupled receptors (StaRs) and their use in drug discovery. Neuropharmacology 60, 36–44 (2011)
Ballesteros, J. A. & Weinstein, H. Integrated methods for the construction of three-dimensional models and computational probing of structure-function relations in G protein-coupled receptors. Methods Neurosci. 25, 366–428 (1995)
Wootten, D., Simms, J., Miller, L. J., Christopoulos, A. & Sexton, P. M. Polar transmembrane interactions drive formation of ligand-specific and signal pathway-biased family B G protein-coupled receptor conformations. Proc. Natl Acad. Sci. USA 110, 5211–5216 (2013)
Hollenstein, K. et al. Insights into the structure of class B GPCRs. Trends Pharmacol. Sci. 35, 12–22 (2014)
Deupi, X. & Standfuss, J. Structural insights into agonist-induced activation of G-protein-coupled receptors. Curr. Opin. Struct. Biol. 21, 541–551 (2011)
Zhang, D. et al. Two disparate ligand-binding sites in the human P2Y1 receptor. Nature 520, 317–321 (2015)
Tehan, B. G., Bortolato, A., Blaney, F. E., Weir, M. P. & Mason, J. S. Unifying family A GPCR theories of activation. Pharmacol. Ther. 143, 51–60 (2014)
Acknowledgements
We thank D. Hall and J. Waterman at I04, Diamond Light Source, Oxford, UK for technical support. We thank colleagues at Heptares Therapeutics Ltd for suggestions and comments, specifically R. K. Y. Cheng.
Author information
Authors and Affiliations
Contributions
D.L. and A.J. devised the strategy and carried out the conformational thermostabilization of the receptor and construct engineering. H.K. established procedures for, and H.K. and S.M.S. carried out expression and optimized purification of the final construct. H.K., S.M.S. and A.S.D. established the platform/protocols for LCP crystallization, harvested crystals, collected and processed X-ray diffraction data, and solved and refined the structure. N.J.R. supported thermostabilization. M.K. and J.C.E. supported expression and purification of the final StaR. A.H.B., I.T. and A.J.H.B. carried out and analysed the pharmacology data. Computational analysis of the structure and modelling was carried out by A.B. S.P.A. identified and sourced the chemical compounds used in the study. Project management was carried out by A.J., R.M.C., M.W. and F.H.M. The manuscript was prepared by A.S.D., A.J. and F.H.M. All authors contributed to the final editing and approval of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The work described here was carried out by employees of Heptares Therapeutics Limited.
Extended data figures and tables
Extended Data Figure 1 Packing interactions in the GCGR-StaR(136–417)–T4L primitive orthorhombic crystal system.
a, View along b-axis, GCGR-StaR TMD in blue ribbon representation, T4L in magenta ribbon representation, MK-0893 in stick representation with carbon, nitrogen, oxygen and chlorine atoms coloured yellow, blue, red and green, respectively. b, View as in a rotated 90° to view along c-axis. c, Close-up of the hydrophobic/shape complementarity interaction of the dichlorophenyl ‘head’ group of MK-0893 with residues on TM4 of a symmetry-related copy.
Extended Data Figure 2 Molecular dynamics analysis of the GCGR–MK-0893 complex.
a, Structural alignment of the wild-type GCGR–MK-0893 complex at 0, 4, 8, 12, 16 and 20 ns molecular dynamics, colour-coded from blue to red as indicated by the arrow. Protein is shown as ribbon, MK-0893 as sticks. b, Extracellular view of the ligand conformations during the molecular dynamics simulation, colour-coded as in a. c, Root-mean-squared deviation (r.m.s.d.) in Å for protein Cα (blue) and ligand heavy atom (pink) during the simulation, after structural alignment to the initial model. d, Number of protein–ligand contacts during the simulation: top, fluctuation of the total number of contacts over time; bottom, individual residues interacting with the ligand at a particular time are shown as rectangles colour-coded based on the number of contacts, from white (no contacts) to dark red (four contacts). e, Two-dimensional representation of the ligand–protein contacts. Green circles represent hydrophobic, cyan represents polar and purple are charged residues. Interactions that occur more than 20% of the simulation time in the selected trajectory are shown and the percentage frequency is marked. Hydrogen bonds are shown as pink arrows. A π–cation interaction is shown as a red line. The part of the ligand buried within the membrane is surrounded by a continuous line, while the water-exposed atoms (corresponding to the propionic acid moiety) are indicated by grey circles. f, GRID (Molecular Discovery) analysis of the ligand-binding site. The shape of the pocket is shown as grey mesh, hydrophilic and hydrophobic hotspots are shown using green and yellow transparent surfaces, respectively. This panel was prepared in Vida (OpenEye).
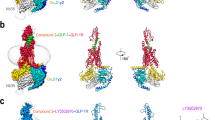
Extended Data Figure 4 Structural superposition of GCGR structures.
a, Structural superposition of the GCGR-StaR(136–417)–MK-0893 structure and GCGR–NNC0640 structure (PDB accession 4L6R) in blue and orange ribbon representation, respectively. b, Close-up view of the intracellular ends of TM5, TM6 and TM7 in 4L6R, as viewed from within the membrane. A PEG molecule modelled in the 4L6R coordinates is shown in stick representation with carbon coloured grey and oxygen coloured red. m|Fo| − Δ|Fc| map calculated using the 4L6R structure factors and 4L6R coordinates with the PEG molecule omitted, difference density is rendered at 2.5σ. c, View as in b, with the GCGR-StaR(136–417)–MK-0893 structure overlaid with MK-0893 in stick representation with carbon, nitrogen, oxygen and chlorine atoms coloured yellow, blue, red and green, respectively. d, Representation as in c, close-up and tilted towards the view from the cytoplasm.
Extended Data Figure 5 Conservation of residues in the MK-0893 bipartite allosteric pocket.
a, Sequence alignment of 15 human GPCR class B members across residues constituting the MK-0893 bipartite binding site. UniProt accession numbers are given. b, The GCGR MK-0893 allosteric binding site is shown in surface representation with residues in a coloured in rainbow spectrum according to conservation level (red = 100%; blue = 0%). MK-0893 is in stick representation with carbon, nitrogen, oxygen and chlorine atoms coloured purple, blue, red and green, respectively.
Extended Data Figure 6 Expression of GCGR mutants.
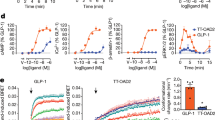
Cell-surface expression of GCGR mutants determined using FACS. Data are expressed as the ratio of APC (cell surface expression) to GFP (total expression) and calculated as percentage of wild type. Experiments were carried out in triplicate and error bars indicate s.e.m. P values are derived from an unpaired two-tailed t-test.
Extended Data Figure 7 Saturation binding analysis of mutants with [3H]MK-0893.
a–g, Saturation binding of [3H]MK-0893 to membrane containing the indicated variants of GCGR. Data are representative of three independent experiments. pKd values are average of three independent experiments and error bars represent s.e.m. P values are calculated from a two-tailed t-test. The data set for R346E and N404A did not fit the one-site binding unambiguously due to near complete loss of specific binding.
Extended Data Figure 8 Effect of binding-site mutations in functional assay.
Cells expressing either wild type or the indicated mutants were stimulated with a concentration range of glucagon in the presence of increasing concentrations of MK-0893. After determination of levels of cAMP generated, the data were analysed by global fitting to the half-maximum effective concentration (EC50) allosteric shift equation. pKB is the negative log of the antagonist affinity that mediates the allosteric inhibition of the glucagon response. Data are an average of three independent experiments and error bars represent s.e.m. P values are derived from an unpaired two-tailed t-test.
Supplementary information
Supplementary Information
This file contains Supplementary Methods and additional references. (PDF 527 kb)
Rights and permissions
About this article
Cite this article
Jazayeri, A., Doré, A., Lamb, D. et al. Extra-helical binding site of a glucagon receptor antagonist. Nature 533, 274–277 (2016). https://doi.org/10.1038/nature17414
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature17414
This article is cited by
-
Structural basis for recognition of antihistamine drug by human histamine receptor
Nature Communications (2022)
-
G protein-coupled receptors: structure- and function-based drug discovery
Signal Transduction and Targeted Therapy (2021)
-
Crystal structure of dopamine D1 receptor in complex with G protein and a non-catechol agonist
Nature Communications (2021)
-
Ligand binding at the protein–lipid interface: strategic considerations for drug design
Nature Reviews Drug Discovery (2021)
-
Biologically active compounds and drugs in the tetrazole series
Chemistry of Heterocyclic Compounds (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.