Abstract
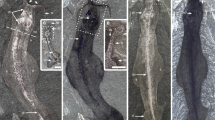
Problematic fossils, extinct taxa of enigmatic morphology that cannot be assigned to a known major group, were once a major issue in palaeontology. A long-favoured solution to the ‘problem of the problematica’1, particularly the ‘weird wonders’2 of the Cambrian Burgess Shale, was to consider them representatives of extinct phyla. A combination of new evidence and modern approaches to phylogenetic analysis has now resolved the affinities of most of these forms. Perhaps the most notable exception is Tullimonstrum gregarium3, popularly known as the Tully monster, a large soft-bodied organism from the late Carboniferous Mazon Creek biota (approximately 309–307 million years ago) of Illinois, USA, which was designated the official state fossil of Illinois in 1989. Its phylogenetic position has remained uncertain and it has been compared with nemerteans4,5, polychaetes4, gastropods4, conodonts6, and the stem arthropod Opabinia4. Here we review the morphology of Tullimonstrum based on an analysis of more than 1,200 specimens. We find that the anterior proboscis ends in a buccal apparatus containing teeth, the eyes project laterally on a long rigid bar, and the elongate segmented body bears a caudal fin with dorsal and ventral lobes3,4,5,6. We describe new evidence for a notochord, cartilaginous arcualia, gill pouches, articulations within the proboscis, and multiple tooth rows adjacent to the mouth. This combination of characters, supported by phylogenetic analysis, identifies Tullimonstrum as a vertebrate, and places it on the stem lineage to lampreys (Petromyzontida). In addition to increasing the known morphological disparity of extinct lampreys7,8,9, a chordate affinity for T. gregarium resolves the nature of a soft-bodied fossil which has been debated for more than 50 years.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Bengtson, S. in Problematic Fossil Taxa (eds Hoffman, A. & Nitecki, M. H. ) 3–11 (Oxford Univ. Press, 1986)
Gould, S. J. Wonderful Life: The Burgess Shale and the Nature of History (WW Norton, 1990)
Richardson, E. S. Jr. Wormlike fossil from the Pennsylvanian of Illinois. Science 151, 75–76 (1966)
Foster, M. in Mazon Creek Fossils (ed. Nitecki, M. H. ) 269–301 (Academic, 1979)
Johnson, R. G. & Richardson, E. S. Pennsylvanian invertebrates of the Mazon Creek Area, Illinois: the morphology and affinities of Tullimonstrum. Fieldiana Geol. 12, 119–149 (1969)
Beall, B. in The Early Evolution of Metazoa and the Significance of Problematic Taxa (eds Simonetta, A. M. & Conway Morris, S. ) 271–286 (Cambridge Univ. Press, 1991)
Janvier, P. & Sansom, R. S. in Hagfish Biology (eds Edwards, S. L. & Goss, G. G. ) 73–94 (CRC, 2015)
Bardack, D. & Zangerl, R. in The Biology of Lampreys (eds Hardisty, M. W. & Potter, I. C. ) Vol. 1, 67–84 (Academic, 1971)
Janvier, P. Early Vertebrates (Oxford Monographs on Geology and Geophysics Vol. 33) (Oxford Univ. Press, 1996)
Bardack, D. & Richardson, E. Jr. New agnathous fishes from the Pennsylvanian of Illinois. Fieldiana Geol. 33, 489–510 (1977)
Janvier, P. The phylogeny of the Craniata, with particular reference to the significance of fossil “agnathans”. J. Vert. Paleont. 1, 121–159 (1981)
Baird, G. C., Sroka, S. D., Shabica, C. W. & Kuecher, G. J. Taphonomy of Middle Pennsylvanian Mazon Creek area fossil localities, northeast Illinois: significance of exceptional fossil preservation in syngenetic concretions. Palaios 1, 271–285 (1986)
Jacobs, D. K. et al. Molluscan engrailed expression, serial organization, and shell evolution. Evol. Dev. 2, 340–347 (2000)
Sansom, R. S., Gabbott, S. E. & Purnell, M. A. Atlas of vertebrate decay: a visual and taphonomic guide to fossil interpretation. Palaeontology 56, 457–474 (2013)
Yalden, D. Feeding mechanisms as evidence for cyclostome monophyly. Zool. J. Linn. Soc. 84, 291–300 (1985)
Alibardi, L. & Segalla, A. The process of cornification in the horny teeth of the lamprey involves proteins in the keratin range and other keratin-associated proteins. Zool. Stud. 50, 416–425 (2011)
Scheltema, A. H., Kerth, K. & Kuzirian, A. M. Original molluscan radula: comparisons among Aplacophora, Polyplacophora, Gastropoda, and the Cambrian fossil Wiwaxia corrugata . J. Morphol. 257, 219–245 (2003)
Morris, S. C. & Caron, J.-B. A primitive fish from the Cambrian of North America. Nature 512, 419–422 (2014)
Meeuwig, M. H., Bayer, J. M. & Reiche, R. A. Morphometric discrimination of early life stage Lampetra tridentata and L. richardsoni (Petromyzonidae) from the Columbia River Basin. J. Morphol. 267, 623–633 (2006)
McDowall, R. M. Jordan’s and other ecogeographical rules, and the vertebral number in fishes. J. Biogeogr. 35, 501–508 (2008)
Janvier, P. Facts and fancies about early fossil chordates and vertebrates. Nature 520, 483–489 (2015)
Bardack, D. First fossil hagfish (Myxinoidea): a record from the Pennsylvanian of Illinois. Science 254, 701–703 (1991)
Vogt, S. MAPS: a set of software tools for analysis and visualization of 3D X-ray fluorescence data sets. J. Phys. IV Fr. 104, 635–638 (2003)
Goloboff, P. A., Farris, J. A. & Nixon, K. C. TNT, a free program for phylogenetic analysis. Cladistics 24, 774–786 (2008)
Felsenstein, J. Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39, 783–791 (1985)
Farris, J. S., Albert, V. A., Källersjö, M., Lipscomb, D. & Kluge, A. G. Parsimony jackknifing outperforms neighbor-joining. Cladistics 12, 99–124 (1996)
Bremer, K. Branch support and tree stability. Cladistics 10, 295–304 (1994)
Maddison, W. P. & Maddison, D. R. Mesquite: a modular system for evolutionary analysis v.3.02 (2015)
Acknowledgements
We thank affiliates of the Field Museum of Natural History: J. Voight and J. Wittry for discussion; P. Heck for help with scanning electron microscopy and energy-dispersive spectroscopy; and N. Karpus for specimen photography. This research used resources of the Advanced Photon Source, a US Department of Energy Office of Science User Facility operated for the US Department of Energy Office of Science by Argonne National Laboratory under contract number DE-AC02-06CH11357. The Field Museum of Natural History, the Sedgwick Museum in Cambridge, UK, and C. Eaton at the University of Wisconsin-Madison Geology Museum provided access to specimens. Access to the software TNT for phylogenetic analysis was provided by the Willi Hennig Society. Funding was provided by a Field Museum visiting scholarship to V.E.M. and by the NASA Astrobiology Institute (NNA13AA90A) Foundations of Complex Life, Evolution, Preservation and Detection on Earth and Beyond.
Author information
Authors and Affiliations
Contributions
V.E.M. conceived the study and wrote the initial draft. V.E.M., E.E.S., L.G.T., J.C.L., and D.E.G.B. developed the project. V.E.M., E.E.S., J.C.L., L.G.T., S.M., S.L., P.M., C.D.W., E.G.C., and R.P.A. analysed and measured specimens. J.C.L. ran the phylogenetic analysis. E.E.S., C.S., L.F., and S.V. performed the synchrotron analysis. S.M. created the reconstruction. H.P. dissected modern taxa for comparative purposes. E.R.L., E.E.S., and S.M. photographed comparative fossil taxa. All authors reviewed and edited the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Details of myomeres, myomere separations, and gills in Tullimonstrum.
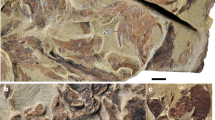
Anterior to the right. Scale bars, 10 mm. Insets are line drawings of the specimens, with colours as in Fig. 1d. a, b, W-shaped myomeres. a, FMNH PE32436, narrow myomere separations. b, FMNH PE32386, wider myomere separations. Note the folded tail and the dorsal fin which extends from approximately the fifth myomere (arrow). c, FMNH PE32423, chevron-shaped myomeres and dorsal fin extending from the fifth most posterior myomere (arrow). The tail appears unusually narrow because of an atypical vertical orientation. d, FMNH PE7063, straight myomeres separated only at the edges and corresponding to repeated arcualia (arrows). e, FMNH PE32395, myomeres separated only at the margins of the body except in the tail where they are completely separated (arrows). f, FMNH PE10654, myomeres separated in the tail. Note the axially positioned notochord in the tail (black arrow), and the asymmetric fin with rays (white arrow). g, FMNH PE10601, myomere separations that resemble gill pouches but differ in their W-shape, lighter colour, and maximum width at the margin of the body. Note the arcualia (arrows) accentuated in white by kaolinite (arrows). h, FMNH PE45366, gill pouches (arrows) that are elliptical, darker in colour, and widest immediately adjacent to the notochord (reddish brown in the fossil). The eyebar is indicated by a white arrow. i, j, Linear regressions on segmentation variables. The assumptions for linear regression were tested using the R package gvlma, and all were met. The data are presented in Supplementary Table 2. Data were included for all specimens for which segments could be counted and measured, except for one outlier, which was removed from the calculations with no change to the P values or R2 values of the regressions. i, Regression between body size and average myomere size, n = 25, R2 = 0.57, P = 4.92 × 10−10. j, Regression between body size and number of myomeres in the body, n = 25, R2 = 0.05, P = 0.19.
Extended Data Figure 2 Dorsoventral position of axial structures in Tullimonstrum.
Specimens preserved obliquely (as indicated by asymmetric preservation of the eyebar, its midpoint indicated by white arrow); the offset of medial structures relative to the axis of the specimen indicates their dorsoventral position. Anterior to the right. Insets are line drawings of the specimens, with colours as in Fig. 1d. Scale bars, 1 cm. a, FMNH PE40113, displacement of the centre of the eyebar, the notochord (black arrow), and the dorsal fin (red arrow) in the same direction, indicating that these are all dorsal structures. The notochord follows the curvature of the body. The tail bends ventrally at the posterior tip (blue arrow) and the dorsal lobe (DL) of the caudal fin is longer than but not as deep as the ventral lobe (VL). b, c, Comparison of the position of additional medial features with that of the centre of the eyebar (which indicates the notochord position even when the notochord is not preserved). b, FMNH PE9864, displacement of the central bulb of the eyebar and the gut trace (black arrow), in opposite directions, indicating that the gut trace is ventral. c, FMNH PE24567, displacement of the central bulb of the eyebar and the arcualia (black arrow) in the same direction, indicating that the arcualia are dorsal.
Extended Data Figure 3 The tri-lobed brain of Tullimonstrum.
a–c, Anterior to the top; d, anterior to the right. Scale bars, 10 mm. a, b, Yellow arrow, olfactory lobe; orange arrow, optic lobe; red arrow, otic lobe. a, FMNH PE45350, the three lobes of the brain, the large olfactory lobe anterior to the eyebar, the central optic lobe on the eyebar, and the large otic lobe posterior to it. b, FMNH PE22103, a faint trace of the anterior olfactory lobe, the central optic lobe preserved in association with the eyebar, and the posterior otic lobe immediately adjacent to the notochord (black arrow). c, Comparison of the brain of Tullimonstrum (above, based on FMNH PE45350 in a) with the brain of a typical lamprey (below) showing the olfactory lobe(s) in yellow, the optic lobes and optic nerve connections in orange, the cerebellum/medulla in red, and the notochord in grey. d, FMNH PE39890, bilobed central optic lobe (orange arrow) on the eyebar. Note the faint dark trace of the oesophagus in the proboscis (black arrow) and the crescent-shaped naris (white arrow).
Extended Data Figure 4 Buccal apparatus of Tullimonstrum.
All scale bars, 5 mm. Angled arrows indicate the lingual organ, and vertical arrows indicate teeth (not all of which are marked). a, FMNH PE45419, dark stain in the base of the apparatus that may represent a remnant of the lingual organ. The base and bifurcate structure are both asymmetric; the thicker (presumably dorsal) element of the bifurcate structure occurs on the same side as the dorsal bulge in the base. Three teeth (indicated by arrows) lie within the base rather than along the bifurcate structure. Teeth are present as far as the distal-most end of the bifurcate structure. The teeth are preserved as three-dimensional moulds and casts, occasionally as pyrite infills, and are situated on raised soft tissue areas (blue arrow in bifurcate structure). b, FMNH PE28739, lingual organ and two rows of teeth associated with the ventral element of the bifurcate structure. Two teeth in each row are indicated by arrows; the rows are offset and indicated by black and yellow arrows. Teeth are preserved as moulds. c, FMNH PE31057, teeth with white kaolinite infill of negative-relief moulds. Other teeth are preserved as casts or moulds, beyond the distal-most preserved outline of the bifurcate structure. At the distal-most end, the teeth of each element of the bifurcate structure are offset (alternating), whereas for most of the length of the buccal apparatus they are not offset. The apparent asymmetry of the elements of the buccal apparatus is exaggerated as the kaolinite does not reveal the complete outline. d, A Tullimonstrum tooth in three dimensions as a slightly hooked hollow cone with a bulbous base. e–i, The three-dimensional tooth in d is digitally ‘rotated’ and ‘sliced’, to show how a two-dimensional representation of a three-dimensional structure may result in a variety of morphologies. These morphologies match those seen in preserved Tullimonstrum teeth, suggesting that the three-dimensional reconstruction in d might represent the original shape.
Extended Data Figure 5 Elemental maps of Tullimonstrum.
a, e, i, Photographs and (b–d, f–h) synchrotron analysis of FMNH PE10504 and (j–l) FMNH PE45419, showing the distribution of calcium, copper, and zinc in (b–d) the body, (f–h) the eye (corresponding to the box d), and (j–l) the buccal mass. Scale bars, 10 mm (a–d, i–l) and 1 mm (e–h, m–o). Zinc shows a high concentration in the body and eye, but the buccal apparatus is characterized by enrichment in copper relative to the matrix. Note that the imaged buccal apparatus bears teeth, but they are characterized by enrichments distinct from those of the bifurcate structure. m–o, SEM/EDS analysis of (m) the teeth (at the spot indicated by the black arrow in d, where the proboscis folds back over the body) and (o) the eye of FMNH PE10504: yellow, sulphur; red, iron; showing the pyrite preservation (teeth indicated by arrows), and (n) the teeth of FMNH PE45426: green, calcium; blue, aluminium; pink, silicon; indicating clay mineral preservation of the teeth (in purple, indicated by arrows).
Extended Data Figure 6 Elemental maps of comparative taxa.
For each taxon, an icon shows the specimen regions analysed for Zn enrichment (brighter regions) with synchrotron (a, c, d, h, i, k) or for general elemental composition with SEM/EDS (b, e–g, j, l): green box, eyes; blue box, soft body tissue; red box, feeding structures. In SEM/EDS maps (b, e–g, j, l): red, iron; yellow, sulphur; green, calcium; pink, silicon; purple, phosphorus; blue, aluminium. The EDS cannot capture carbon, so a thin layer of it appears identical to the matrix, and a thick layer appears black. All scale bars 1 mm, except f which is 0.5 mm. a–c, FMNH PF8719, chordate Esconichthys apopyris; (a) eyes enriched in zinc, (b) preserved in pyrite, and (c) body lacking zinc enrichment. d, FMNH PE 39321, unidentified cephalopod, radula and soft tissue lacking zinc enrichment (dark areas are due to relief). e, f, FMNH PF7499, chordate Elonichthys peltigerus; (e) eyes lacking pyrite and (f) teeth preserved in calcium, sulfur, and phosphorus interpreted to reflect the original apatite. g, FMNH PE12371, polychaete Fossundecima konecniorum, jaw apparatus (two light-coloured triangular areas, each with an arrow, on a darker background) preserved in a thin carbon film. h, FMNH PE30643, cnidarian Essexella asherae, tissue lacking zinc enrichment. i, FMNH PE 30054, Etacystis communis, unidentified soft-bodied organism, possibly cnidarian, soft tissue may be slightly enriched in zinc. j, FMNH PE21951, holothurian Achistrum sp., teeth of circular jaw preserved in calcium reflecting the original (calcite) composition. k, FMNH PE15530, crustacean Lobetelson mclaughlinae, eyes lacking zinc enrichment. l, FMNH PE23336, horseshoe crab Euproops danae, telson preserved as a thick carbon film.
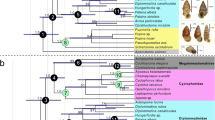
Extended Data Figure 7 Phylogenetic analysis.
Strict consensus of the 18 most parsimonious trees retrieved under equal weighting (consistency index = 0.549, retention index = 0.614, rescaled consistency index = 0.337). Jacknife support above each node in plain text, Bremer support emboldened and in parentheses, and bootstrap values beneath each node in italics.
Extended Data Figure 8 Internal structures of Gilpichthys and Tullimonstrum.
Anterior to right. a, FMNH PF8410, Gilpichthys, gut (arrow) preserved in a fashion similar to typical Mazon Creek coprolites. Scale bar, 5 mm. b, FMNH PE10638, Tullimonstrum with preserved tectal cartilages (arrow). Scale bar, 10 mm. c, FMNH PE39169, Tullimonstrum with liver (arrow) preserved as a diffuse dark circular structure. Inset is a line drawing: red, notochord; brown, liver. Scale bar, 10 mm.
Supplementary information
Supplementary Information
This file contains a Supplementary Discussion, which includes morphological interpretations, phylogenetic analysis, a data matrix for the phylogenetic analysis and systematic palaeontology. Additional references are also included. (PDF 329 kb)
Supplementary Table 1
This file contains a list of specimens included in this study, with the presence and absence of major morphological features. (XLSX 98 kb)
Supplementary Table 2
This file contains the data for the linear regressions presented in Extended Data Figure 1. (XLSX 12 kb)
Rights and permissions
About this article
Cite this article
McCoy, V., Saupe, E., Lamsdell, J. et al. The ‘Tully monster’ is a vertebrate. Nature 532, 496–499 (2016). https://doi.org/10.1038/nature16992
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature16992
This article is cited by
-
Homology thinking reconciles the conceptual conflict between typological and population thinking
Biology & Philosophy (2021)
-
Fin modules: an evolutionary perspective on appendage disparity in basal vertebrates
BMC Biology (2017)
-
Palaeospondylus as a primitive hagfish
Zoological Letters (2016)
-
Getting the measure of a monster
Nature (2016)
-
Preservation and phylogeny of Cambrian ecdysozoans tested by experimental decay of Priapulus
Scientific Reports (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.