Abstract
Sighs are long, deep breaths expressing sadness, relief or exhaustion. Sighs also occur spontaneously every few minutes to reinflate alveoli, and sighing increases under hypoxia, stress, and certain psychiatric conditions. Here we use molecular, genetic, and pharmacologic approaches to identify a peptidergic sigh control circuit in murine brain. Small neural subpopulations in a key breathing control centre, the retrotrapezoid nucleus/parafacial respiratory group (RTN/pFRG), express bombesin-like neuropeptide genes neuromedin B (Nmb) or gastrin-releasing peptide (Grp). These project to the preBötzinger Complex (preBötC), the respiratory rhythm generator, which expresses NMB and GRP receptors in overlapping subsets of ~200 neurons. Introducing either neuropeptide into preBötC or onto preBötC slices, induced sighing or in vitro sigh activity, whereas elimination or inhibition of either receptor reduced basal sighing, and inhibition of both abolished it. Ablating receptor-expressing neurons eliminated basal and hypoxia-induced sighing, but left breathing otherwise intact initially. We propose that these overlapping peptidergic pathways comprise the core of a sigh control circuit that integrates physiological and perhaps emotional input to transform normal breaths into sighs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Haldane, J. S., Meakins, J. C. & Priestley, J. G. The effects of shallow breathing. J. Physiol. (Lond.) 52, 433–453 (1919)
McCutcheon, F. H. Atmospheric respiration and the complex cycles in mammalian breathing mechanisms. J. Cell. Physiol. 41, 291–303 (1953)
Knowlton, G. C. & Larrabee, M. G. A unitary analysis of pulmonary volume receptors. Am. J. Physiol. 147, 100–114 (1946)
Reynolds, L. B. Characteristics of an inspiration-augmenting reflex in anesthetized cats. J. Appl. Physiol. 17, 683–688 (1962)
Bartlett, D. Origin and regulation of spontaneous deep breaths. Respir. Physiol. 12, 230–238 (1971)
Maytum, C. K. Sighing dyspnea: A clinical syndrome. J. Allergy 10, 50–55 (1938)
Smith, J. C., Ellenberger, H. H., Ballanyi, K., Richter, D. W. & Feldman, J. L. Pre-Bötzinger complex: a brainstem region that may generate respiratory rhythm in mammals. Science 254, 726–729 (1991)
Gray, P. A., Janczewski, W. A., Mellen, N., McCrimmon, D. R. & Feldman, J. L. Normal breathing requires preBötzinger complex neurokinin-1 receptor-expressing neurons. Nature Neurosci. 4, 927–930 (2001)
Tan, W. et al. Silencing preBötzinger complex somatostatin-expressing neurons induces persistent apnea in awake rat. Nature Neurosci. 11, 538–540 (2008)
Feldman, J. L., Del Negro, C. A. & Gray, P. A. Understanding the rhythm of breathing: so near, yet so far. Annu. Rev. Physiol. 75, 423–452 (2013)
Kam, K., Worrell, J. W., Janczewski, W. A., Cui, Y. & Feldman, J. L. Distinct inspiratory rhythm and pattern generating mechanisms in the preBötzinger complex. J. Neurosci. 33, 9235–9245 (2013)
Lieske, S. P., Thoby-Brisson, M., Telgkamp, P. & Ramirez, J. M. Reconfiguration of the neural network controlling multiple breathing patterns: eupnea, sighs and gasps. Nature Neurosci. 3, 600–607 (2000)
Ruangkittisakul, A. et al. Generation of eupnea and sighs by a spatiochemically organized inspiratory network. J. Neurosci. 28, 2447–2458 (2008)
Caughey, J. L. Jr. Analysis of breathing patterns. Am. Rev. Tuberc. 48, 382 (1943)
Niewoehner, D. E., Levine, A. S. & Morley, J. E. Central effects of neuropeptides on ventilation in the rat. Peptides 4, 277–281 (1983)
Ramirez, J. M. The integrative role of the sigh in psychology, physiology, pathology, and neurobiology. Prog. Brain Res. 209, 91–129 (2014)
Janczewski, W. A., Pagliardini, S., Cui, Y. & Feldman, J. L. Sighing after vagotomy. Proc. Society of Neuroscience Conference (New Orleans, 2012)
Smith, J. C., Morrison, D. E., Ellenberger, H. H., Otto, M. R. & Feldman, J. L. Brainstem projections to the major respiratory neuron populations in the medulla of the cat. J. Comp. Neurol. 281, 69–96 (1989)
Onimaru, H. & Homma, I. A novel functional neuron group for respiratory rhythm generation in the ventral medulla. J. Neurosci. 23, 1478–1486 (2003)
Diez-Roux, G. et al. A high resolution atlas of the transcriptome in the mouse embryo. PLoS Biol. 9, e1000582 (2011)
Tomer, R. et al. Advanced CLARITY for rapid and high-resolution imaging of intact tissues. Nature Protocols 9, 1682–1697 (2014)
Mulkey, D. K. et al. Respiratory control by ventral surface chemoreceptor neurons in rats. Nature Neurosci. 7, 1360–1369 (2004)
Stornetta, R. L. et al. Expression of Phox2b by brainstem neurons involved in chemosensory integration in the adult rat. J. Neurosci. 26, 10305–10314 (2006)
Guyenet, P. G. & Bayliss, D. A. Neural control of breathing and CO2 homeostasis. Neuron 87, 946–961 (2015)
Lazarenko, R. M. et al. Acid sensitivity and ultrastructure of the retrotrapezoid nucleus in Phox2b–EGFP transgenic mice. J. Comp. Neurol. 517, 69–86 (2009)
Bendixen, H. H., Smith, G. M. & Mead, J. Pattern of ventilation in young adults. J. Appl. Phyiol . 19, 195–198 (1964)
Jensen, R. T. et al., International Union of Pharmacology. LXVIII. Mammalian bombesin receptors: nomenclature, distribution, pharmacology, signaling, and functions in normal and disease states. Pharmacol. Rev. 60, 1–42 (2008)
Guyenet, P. G., Stornetta, R. L. & Bayliss, D. A. Central respiratory chemoreception. J. Comp. Neurol. 518, 3883–3906 (2010)
Lee, H., Naughton, N. N., Woods, J. H. & Ko, M. C. Characterization of scratching responses in rats following centrally administered morphine or bombesin. Behav. Pharmacol. 14, 501–508 (2003)
Pagliardini, S. et al. Active expiration induced by excitation of ventral medulla in adult anesthetized rats. J. Neurosci. 31, 2895–2905 (2011)
Ohki-Hamazaki, H. et al. Functional properties of two bombesin-like peptide receptors revealed by the analysis of mice lacking neuromedin B receptor. J. Neurosci. 19, 948–954 (1999)
Wada, E. et al. Generation and characterization of mice lacking gastrin-releasing peptide receptor. Biochem. Biophys. Res. Commun. 239, 28–33 (1997)
Orbuch, M. et al. Discovery of a novel class of neuromedin B receptor antagonists, substituted somatostatin analogues. Mol. Pharmacol. 44, 841–850 (1993)
Qin, Y. et al. Inhibitory effect of bombesin receptor antagonist RC-3095 on the growth of human pancreatic cancer cells in vivo and in vitro. Cancer Res. 54, 1035–1041 (1994)
Acknowledgements
We thank M. Sunday for providing the Nmbr−/− and Grpr−/− mice, Y. Zhang for providing rat tissues, and K. Wada and E. Wada for plasmid constructs for in situ hybridization probes. We also thank members of the Krasnow and Feldman laboratories for comments. This work was supported by the Howard Hughes Medical Institute (M.A.K.), NIH grants HL70029, HL40959 and NS72211 (J.L.F.), a Walter V. and Idun Berry postdoctoral fellowship (P.L.), the NIH Medical Scientist Training Program (K.Y.), and CIHR and AIHS postdoctoral fellowships (S.P.). M.A.K. is an investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
W.A.J. and S.P. performed experiments showing the effects on sighing of bombesin injection into the preBötC and ablation of receptor-expressing neurons with bombesin–saporin. K.Y. performed the screen that discovered Nmb expression in the respiratory centres. P.L. performed experiments identifying and characterizing expression of Nmb, Grp and their receptors. W.A.J., P.L., and K.Y. performed genetic and pharmacology experiments on Nmb and Grp pathways. K.K. performed in vitro slice experiments. W.A.J., K.K., P.L., S.P., and K.Y. analysed data. J.L.F., W.A.J., K.K., M.A.K., P.L., and K.Y. conceived experiments, interpreted data and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Expression of Nmb in rodent brain.
a, b, Sagittal sections of P7 mouse (a) and P7 rat (b) brain showing RTN/pFRG region probed for Nmb mRNA expression (purple) by in situ hybridization as in Fig. 1. Scale bars, 100 μm. c, d, Nmb expression as in a showing regions outside ventrolateral medulla. Nmb is expressed in mouse olfactory bulb (c) and hippocampus (d). Scale bars, 200 μm (c) and 100 μm (d). e–h, Section through RTN/pFRG brain region of P0 transgenic Nmb-GFP mouse immunostained for GFP (green) and probed for Nmb mRNA (red) by in situ hybridization. Blue, DAPI nuclear stain. Nmb-GFP and Nmb mRNA are mainly co-expressed in the same cells. Scale bar, 100 μm.
Extended Data Figure 2 Serial confocal preBötC sections showing Nmb-GFP projections contain puncta of NMB.
a–d, Serial confocal optical sections (0.6 μm apart) through preBötC brain region of Nmb-GFP mouse immunostained for GFP (green), NMB (red), preBötC marker SST (white), and DAPI (blue) as in Fig. 2h. Note the GFP-positive projection with a puncta of NMB (yellow, open arrows in b, c) directly abutting an SST positive neuron (asterisk). Most NMB puncta (open arrowheads) were detected within GFP-positive projections as expected, and only a small fraction of NMB puncta (closed arrowhead) were detected outside them; NMB outside Nmb-GFP projections could be secreted protein or the rare Nmb-expressing cells that do not co-express the Nmb-GFP transgene (see Extended Data Fig. 1e–h). Scale bar, 20 μm.
Extended Data Figure 3 Sighing after surgery and bilateral injection of saline into preBötC.
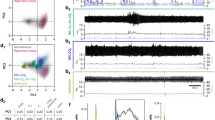
a, Example of a sigh in a breathing activity trace of a urethane anaesthetized rat after surgery as in Fig. 2a–c. VT, tidal volume; ∫Dia, integrated diaphragm activity; Dia, raw diaphragm activity trace. b, Sigh rate before (control) and after (saline) bilateral saline injection into preBötC. There is no effect of saline injection (data are mean ± s.d., n = 5, P = 0.83 by paired t-test). c–f, Breathing activity trace as in a (but also showing airflow). Note stereotyped waveform of sighs (d–f). Bars, 1 min (c), 1 s (d–f).
Extended Data Figure 4 Effects on sighing in individual rats following bilateral injection into preBötC of NMB, GRP and both NMB/GRP.
a–e, Raster plot of sighs (upper) and instantaneous sigh rates (lower) before and after NMB injection for the five experiments (a–e) shown in Fig. 2d. f–j, Raster plot of sighs (upper) and instantaneous sigh rates (lower) before and after GRP injection for the five experiments (f–j) shown in Fig. 3c. k–o, Raster plot of sighs (upper) and instantaneous sigh rates (lower) before and after NMB/GRP injection for the five experiments (k–o) shown in Fig. 4i. Grey, injection period; arrowhead in raster plots, maximum instantaneous sigh rate; numbers, basal (left) and maximal instantaneous sigh rate (right) and fold induction (in parentheses) after neuropeptide injection.
Extended Data Figure 5 Effect of NMB and GRP on rhythmic activity of preBötC slice.
a, Neuronal activity trace(∫XII, black; ∫preBötC population activity, grey) of preBötC slice containing 30 nM NMB, as in Fig. 2e. Note the extreme effect of NMB in which every burst (‘breath’) in the trace is a doublet (‘sigh’, asterisk). Bar, 5 s. b, c, NMB increases the doublet rate by increasing the fraction of total events that are doublets (b) and decreasing the interval following a doublet (c). Data are mean ± s.d., *P < 0.05 by paired t-test, n = 7. d, e, GRP also increases the doublet rate by increasing the fraction of total events that are doublets (d) and decreasing the interval following a doublet (e). Data are mean ± s.d., *P < 0.05 by paired t-test, n = 9. Note that post-doublet intervals are significantly longer than post-burst intervals under all conditions, consistent with longer post-sigh apneas in vivo.
Extended Data Figure 6 Effects on sighing in individual rats following bilateral injection of BIM23042, RC3095 and BIM23042/RC3095 into preBötC.
a–d, Raster plot of sighs (upper) and binned sigh rates (lower; bin size 4 min; slide 30 s) before and after injection of the NMBR antagonist BIM23042 for the four experiments shown in Fig. 2h. e–h, Raster plot of sighs (upper) and binned sigh rates (lower; bin size 4 min; slide 30 s) before and after injection of the GRPR antagonist RC3095 for the four experiments shown in Fig. 3f. i–n, Raster plot of sighs (upper) and binned sigh rates (lower; bin size 4 min; slide 30 s) before and after BIM23042 and RC3095 injection for the six experiments shown in Fig. 4j. Grey, injection period; numbers, longest inter-sigh intervals (s, seconds) following injection.
Extended Data Figure 7 Specificity of antagonists BIM23042 and RC3095 in preBötC slice.
a, BIM 23042 (100 nM) blocks the effect of NMB (10 nM), but not GRP (3 nM) in preBötC slices. Data are mean ± s.d., *P < 0.05 by paired t-test, n = 7. b, RC3095 (100 nM) shows the opposite specificity, blocking the effect of GRP (3 nM), but not NMB (10 nM). Data are mean ± s.d., *P < 0.05 by paired t-test, n = 9.
Extended Data Figure 8 Expression of Grp in rodent brain.
a, b, In situ hybridization of mouse brain slices as in Fig. 3a showing expression of Grp (purple) in parabrachial nucleus (PBN) (a) and nucleus tractus solitarius (NTS) (b). Scale bar, 200 μm. c–e, In situ hybridization of rat brain slices showing expression of Grp in PBN (c), NTS (d), RTN/pFRG (e). Scale bar, 200 μm. f, Tiled image showing GRP-positive projection (red) from RTN/pFRG region to preBötC region containing SST-positive neuron (green). Scale bar, 20 μm. g–i, Serial confocal optical sections (0.8 μm apart) through mouse preBötC stained for GRP (red) and SST (green) focusing on short segment of GRP-positive projection where a GRP puncta (red) directly abuts (arrowhead) an SST-positive neuron. Scale bar, 10 μm.
Extended Data Figure 9 Effect of bombesin injection on sighing following BBN–SAP-induced ablation of NMBR-expressing and GRPR-expressing preBötC neurons.
a, b, 10 min plethysmography traces of a control rat (a) and a day 5 BBN–SAP injected rat (b) during eupneic breathing (left). Indicated parts (10 s) of traces are expanded at right. Note presence of sighs with stereotyped waveform in control rat, and no sighs detectable in BBN–SAP injected rat. c, Sigh rate before (control) and after 10 μg bombesin injection (BBN) into the cisterna magna of rats before BBN–SAP injection (WT) and at day 4 and day 6 after BBN–SAP injection (BBN–SAP) into the preBötC to ablate NMBR-expressing and GRPR-expressing neurons as in Fig. 5a, b. Values shown are mean ± s.d. (WT, n = 10; BBN–SAP, n = 7 for day 4 and n = 5 for day 6), *P < 0.05 by paired t-test; n.s., not significant.
Supplementary information
Video 1: 3-D view of Nmb-GFP neurons in RTN/pFRG region of CLARITY-processed P14 brainstem
Nmb-GFP neurons surround the lateral half of the facial nucleus, as shown in Figure 1d, e. (MP4 2683 kb)
Rights and permissions
About this article
Cite this article
Li, P., Janczewski, W., Yackle, K. et al. The peptidergic control circuit for sighing. Nature 530, 293–297 (2016). https://doi.org/10.1038/nature16964
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature16964
This article is cited by
-
A vagal reflex evoked by airway closure
Nature (2024)
-
SubSol-HIe is an AMPK-dependent hypoxia-responsive subnucleus of the nucleus tractus solitarius that coordinates the hypoxic ventilatory response and protects against apnoea in mice
Pflügers Archiv - European Journal of Physiology (2024)
-
Upregulation of breathing rate during running exercise by central locomotor circuits in mice
Nature Communications (2023)
-
Purinergic signaling mediates neuroglial interactions to modulate sighs
Nature Communications (2023)
-
Modulation of inspiratory burst duration and frequency by bombesin in vitro
Pflügers Archiv - European Journal of Physiology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.