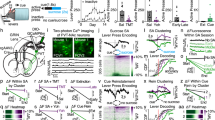
Abstract
Chronic opiate use induces opiate dependence, which is characterized by extremely unpleasant physical and emotional feelings after drug use is terminated. Both the rewarding effects of a drug and the desire to avoid withdrawal symptoms motivate continued drug use1,2,3, and the nucleus accumbens is important for orchestrating both processes4,5. While multiple inputs to the nucleus accumbens regulate reward6,7,8,9, little is known about the nucleus accumbens circuitry underlying withdrawal. Here we identify the paraventricular nucleus of the thalamus as a prominent input to the nucleus accumbens mediating the expression of opiate-withdrawal-induced physical signs and aversive memory. Activity in the paraventricular nucleus of the thalamus to nucleus accumbens pathway is necessary and sufficient to mediate behavioural aversion. Selectively silencing this pathway abolishes aversive symptoms in two different mouse models of opiate withdrawal. Chronic morphine exposure selectively potentiates excitatory transmission between the paraventricular nucleus of the thalamus and D2-receptor-expressing medium spiny neurons via synaptic insertion of GluA2-lacking AMPA receptors. Notably, in vivo optogenetic depotentiation restores normal transmission at these synapses and robustly suppresses morphine withdrawal symptoms. This links morphine-evoked pathway- and cell-type-specific plasticity in the paraventricular nucleus of the thalamus to nucleus accumbens circuit to opiate dependence, and suggests that reprogramming this circuit holds promise for treating opiate addiction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Koob, G. F. & Le Moal, M. Drug abuse: hedonic homeostatic dysregulation. Science 278, 52–58 (1997)
Vargas-Perez, H., Ting-A-Kee, R. A., Heinmiller, A., Sturgess, J. E. & van der Kooy, D. A test of the opponent-process theory of motivation using lesions that selectively block morphine reward. Eur. J. Neurosci. 25, 3713–3718 (2007)
Wikler, A. A theory of opioid dependence. NIDA Res. Monogr. 30, 174–178 (1980)
Harris, G. C. & Aston-Jones, G. Involvement of D2 dopamine receptors in the nucleus accumbens in the opiate withdrawal syndrome. Nature 371, 155–157 (1994)
Koob, G. F., Wall, T. L. & Bloom, F. E. Nucleus accumbens as a substrate for the aversive stimulus effects of opiate withdrawal. Psychopharmacology 98, 530–534 (1989)
Pascoli, V. et al. Contrasting forms of cocaine-evoked plasticity control components of relapse. Nature 509, 459–464 (2014)
Pascoli, V., Turiault, M. & Lüscher, C. Reversal of cocaine-evoked synaptic potentiation resets drug-induced adaptive behaviour. Nature 481, 71–75 (2012)
Britt, J. P. et al. Synaptic and behavioral profile of multiple glutamatergic inputs to the nucleus accumbens. Neuron 76, 790–803 (2012)
Stuber, G. D. et al. Excitatory transmission from the amygdala to nucleus accumbens facilitates reward seeking. Nature 475, 377–380 (2011)
Lim, B. K., Huang, K. W., Grueter, B. A., Rothwell, P. E. & Malenka, R. C. Anhedonia requires MC4R-mediated synaptic adaptations in nucleus accumbens. Nature 487, 183–189 (2012)
Wickersham, I. R., Finke, S., Conzelmann, K. K. & Callaway, E. M. Retrograde neuronal tracing with a deletion-mutant rabies virus. Nature Methods 4, 47–49 (2007)
Sesack, S. R. & Grace, A. A. Cortico-basal ganglia reward network: microcircuitry. Neuropsychopharmacology 35, 27–47 (2010)
Martin-Fardon, R. & Boutrel, B. Orexin/hypocretin (Orx/Hcrt) transmission and drug-seeking behavior: is the paraventricular nucleus of the thalamus (PVT) part of the drug seeking circuitry? Front. Behav. Neurosci. 6, 75 (2012)
Browning, J. R., Jansen, H. T. & Sorg, B. A. Inactivation of the paraventricular thalamus abolishes the expression of cocaine conditioned place preference in rats. Drug Alcohol Depend. 134, 387–390 (2014)
James, M. H. et al. Cocaine- and amphetamine-regulated transcript (CART) signaling within the paraventricular thalamus modulates cocaine-seeking behaviour. PLoS ONE 5, e12980 (2010)
Boyden, E. S., Zhang, F., Bamberg, E., Nagel, G. & Deisseroth, K. Millisecond-timescale, genetically targeted optical control of neural activity. Nature Neurosci. 8, 1263–1268 (2005)
Chow, B. Y. et al. High-performance genetically targetable optical neural silencing by light-driven proton pumps. Nature 463, 98–102 (2010)
Vanderschuren, L. J. et al. Morphine-induced long-term sensitization to the locomotor effects of morphine and amphetamine depends on the temporal pattern of the pretreatment regimen. Psychopharmacology 131, 115–122 (1997)
Bechara, A., Nader, K. & van der Kooy, D. Neurobiology of withdrawal motivation: evidence for two separate aversive effects produced in morphine-naive versus morphine-dependent rats by both naloxone and spontaneous withdrawal. Behav. Neurosci. 109, 91–105 (1995)
Stachniak, T. J., Ghosh, A. & Sternson, S. M. Chemogenetic synaptic silencing of neural circuits localizes a hypothalamus→midbrain pathway for feeding behavior. Neuron 82, 797–808 (2014)
Armbruster, B. N., Li, X., Pausch, M. H., Herlitze, S. & Roth, B. L. Evolving the lock to fit the key to create a family of G protein-coupled receptors potently activated by an inert ligand. Proc. Natl Acad. Sci. USA 104, 5163–5168 (2007)
Williams, J. T., Christie, M. J. & Manzoni, O. Cellular and synaptic adaptations mediating opioid dependence. Physiol. Rev. 81, 299–343 (2001)
Hyman, S. E., Malenka, R. C. & Nestler, E. J. Neural mechanisms of addiction: the role of reward-related learning and memory. Annu. Rev. Neurosci. 29, 565–598 (2006)
Kravitz, A. V., Tye, L. D. & Kreitzer, A. C. Distinct roles for direct and indirect pathway striatal neurons in reinforcement. Nature Neurosci. 15, 816–818 (2012)
Lobo, M. K. et al. Cell type-specific loss of BDNF signaling mimics optogenetic control of cocaine reward. Science 330, 385–390 (2010)
Schwartz, N. et al. Decreased motivation during chronic pain requires long-term depression in the nucleus accumbens. Science 345, 535–542 (2014)
Conrad, K. L. et al. Formation of accumbens GluR2-lacking AMPA receptors mediates incubation of cocaine craving. Nature 454, 118–121 (2008)
Lee, B. R. et al. Maturation of silent synapses in amygdala-accumbens projection contributes to incubation of cocaine craving. Nature Neurosci. 16, 1644–1651 (2013)
Creed, M., Pascoli, V. J. & Lüscher, C. Refining deep brain stimulation to emulate optogenetic treatment of synaptic pathology. Science 347, 659–664 (2015)
Stamatakis, A. M. & Stuber, G. D. Activation of lateral habenula inputs to the ventral midbrain promotes behavioral avoidance. Nature Neurosci. 15, 1105–1107 (2012)
Tye, K. M. et al. Amygdala circuitry mediating reversible and bidirectional control of anxiety. Nature 471, 358–362 (2011)
Yasoshima, Y., Scott, T. R. & Yamamoto, T. Differential activation of anterior and midline thalamic nuclei following retrieval of aversively motivated learning tasks. Neuroscience 146, 922–930 (2007)
Wolff, S. B. et al. Amygdala interneuron subtypes control fear learning through disinhibition. Nature 509, 453–458 (2014)
Ren, J. et al. Habenula “cholinergic” neurons co-release glutamate and acetylcholine and activate postsynaptic neurons via distinct transmission modes. Neuron 69, 445–452 (2011)
Petreanu, L., Huber, D., Sobczyk, A. & Svoboda, K. Channelrhodopsin-2-assisted circuit mapping of long-range callosal projections. Nature Neurosci. 10, 663–668 (2007)
Acknowledgements
We thank R. C. Malenka, K. Shen, L. Luo, T. R. Clandinin and members of the Chen laboratory for comments on the manuscript. We thank M. Asaad, J. Charalel and X. Sun for tracing and behaviour experiments. This work was supported by grants from The Whitehall Foundation, the Ajinomoto innovation alliance program, the Terman Scholarship and start-up funding from Stanford University. G.N. is supported by a training grant from the National Institute on Drug Abuse (5T32DA035165-02).
Author information
Authors and Affiliations
Contributions
X.K.C. conceived the study. Y.J.Z., G.N. and X.K.C. designed the experiments. Y.J.Z. and C.F.R.W. conducted experiments. Y.J.Z. and X.K.C. analysed data. G.N., Y.J.Z. and X.K.C. wrote the manuscript, and all authors participated in the revision of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Retrograde tracing with rabies virus labels brain areas innervating the NAc and naloxone-precipitated opiate withdrawal induces c-Fos expression in the PVT.
a, Representative images show the injection site in the medial shell of the NAc, and retrograde labelling with mCherry in the ventral hippocampus (vHipp), prelimbic cortex (PrL) and basolateral amygdala (BLA). b, Representative images show retrograde labelling with mCherry in the PVT, ranging from Bregma −0.4 to −2.2 mm. c, Distribution of the density of retrogradely labelled neurons in the PVT (n = 7). d, Representative images show c-Fos expression in the PVT induced by naloxone-precipitated opiate withdrawal. e, Distribution of the density of c-Fos expressing neurons in the PVT (n = 5). Area of the PVT at different anterior–posterior coordinates are determined on the basis of the brain atlas published by Allen Institute for Brain Science (available from http://mouse.brain-map.org/experiment/thumbnails/100142143?image_type=atlas). Scale bars, 500 μm. Mean ± s.e.m.
Extended Data Figure 2 Optogenetic targeting of glutamatergic transmission between the PVT and the NAc.
a, A confocal image shows ChR2–eYFP- (enhanced yellow fluorescent protein) expressing terminals from the PVT in the medial shell of the NAc. Inset, injection site in the PVT. Scale bars, 500 μm. b, Example traces (left) and quantification (right) of action potential firing evoked by light stimulation at different frequencies in ChR2-expressing PVT neurons (n = 7). Scale bar, 25 mV, 250 ms. c, Example traces (left) and quantification (right) of photo-evoked EPSCs recorded from the NAc before and after superfusion of CNQX (10 μM, n = 6). Scale bar, 50 pA, 25 ms. Wilcoxon signed-rank test. *P < 0.05. Mean ± s.e.m.
Extended Data Figure 3 Optogenetic activation of the PVT input evoked feed–forward inhibition onto MSNs in the NAc.
a, In situ hybridization with GAD1 (http://mouse.brain-map.org/gene/show/14191), GAD2 (http://mouse.brain-map.org/experiment/show/79591669) and vGlut2 (http://mouse.brain-map.org/experiment/show/73818754) in the PVT (©2014 Allen Institute for Brain Science). b, c, Superfusion of a GABAA receptor antagonist (picrotoxin, 100 μM) selectively blocked IPSCs (b) (n = 4), whereas superfusion of an AMPAR antagonist (CNQX, 10 μM) blocked both IPSCs and EPSCs (c) (n = 5). Scale bar, 400 pA, 30 ms. d, Example trace (left) and quantification (right) of the onset latency for photo-evoked IPSCs and EPSCs in the same neuron (n = 20). Scale bar, 400 pA, 10 ms. Wilcoxon signed-rank test. ***P < 0.001. e, Averaged amplitude of photo-evoked IPSCs in D1-MSNs (n = 11) and D2-MSNs (n = 14). Mann–Whitney U-test, P = 0.67. Mean ± s.e.m.
Extended Data Figure 4 Location of ChR2, ArchT and hM4Di expression in the PVT.
a, Schematics showing the PVT and its surrounding brain nucleus from anterior to posterior locations. 3V, third ventricle; MD, mediodorsal thalamus; CM, central middle thalamus; DG, dentate gyrus; Hb, habenula; IMD, intermediodorsal thalamus. b, Overlay of ChR2–YFP expression in 21 mice, including 13 mice in Fig. 1d and 8 mice in Fig. 4g, h. c, Overlay of ArchT–YFP expression in 18 mice, including 10 mice in Fig. 2d and 8 mice in Extended Data Fig. 7c. d, Overlay of hM4Di–mCherry expression in 16 mice, including 8 mice in Fig. 2g and 8 mice in Extended Data Fig. 7d. Intensity of red colour is proportional to the number of mice expressing virus in the marked area.
Extended Data Figure 5 Dose–response analysis of naloxone-precipitated withdrawal symptoms.
Concentration of naloxone was determined by 2 × 2 factorial design. Different doses of naloxone (0, 1, 5, 10 mg per kg body weight) were injected in chronic saline- (blue) or morphine- (red) treated mice. Behavioural signs of withdrawal including jump (a), rearing (b) and tremor (c) were recorded for 20 min immediately after naloxone injection. CPA (d) tests were performed 24 h after withdrawal. One-way ANOVA followed by Tukey’s test. Jumping: morphine + naloxone group F(3,36) = 9.93, P < 0.0001; rearing: saline + naloxone group F(3,36) = 7.07, P < 0.01; morphine + naloxone group F(3,36) = 22.98, P < 0.0001; tremor: saline + naloxone group F(3,29) = 3.74, P < 0.05; morphine + naloxone group F(3,36) = 40.48, P < 0.0001; CPA: saline + naloxone group F(3,29) = 2.67, P = 0.066; morphine + naloxone group F(3,36) = 9.93, P < 0.0001; P values for post-hoc Tukey’s test are indicated on each comparison pair. Mean ± s.e.m.
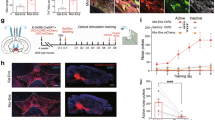
Extended Data Figure 6 Opiate withdrawal induced c–Fos expression in the PVTNAc projection neurons.
a, Example confocal image shows expression of c-Fos in a small number of PVTNAc projection neurons after injection of saline into chronic morphine treated mice. Left: scale bar, 100 μm. Right: magnified image shows the boxed area; scale bar, 50 μm. b, Quantification of CTB- (left) and c-Fos- (right) positive cells per square millimetre after injection of saline (white bar, n = 5) or naloxone (grey bar, n = 6) into chronic morphine-treated mice. c, Example confocal images show that spontaneous withdrawal from morphine (bottom) but not saline (top) treatment increases the expression of c-Fos in PVTNAc projection neurons. Left: scale bar, 100 μm. Right: magnified image shows the boxed area; scale bar, 50 μm. d, Quantification of CTB- (left) and c-Fos- (middle) positive cells per square millimetre and percentage of PVTNAc projection neurons (right) expressing c-Fos induced by spontaneous withdrawal from morphine (grey bar, n = 4) or saline (white bar, n = 4). Mann–Whitney U-test. *P < 0.05, **P < 0.01. Mean ± s.e.m.
Extended Data Figure 7 The PVT→NAc pathway is required for expression of behavioural aversion to footshock and LiCl injection.
a, Confocal images show robust expression of c-Fos (red) in the PVTNAc projection neurons (green) after footshock (top) and LiCl injection (bottom). Left: scale bar, 100 μm. Right: magnified image shows the boxed area; scale bar, 50 μm. b, Quantification of CTB- (left) and c-Fos- (middle) positive cells per square millimetre and percentage of PVTNAc projection neurons (right) expressing c-Fos induced by footshock (white bar, n = 4) or LiCl injection (grey bar, n = 4). c, Light stimulation suppressed the expression of footshock-induced CPA in ArchT- (n = 8) but not eGFP- (n = 8) expressing mice. d, Local infusion of CNO reduced the expression of LiCl-induced CPA in hM4Di- (n = 8) but not eGFP- (n = 8) expressing mice. Mann–Whitney U-test. *P < 0.05, **P < 0.01. Mean ± s.e.m.
Extended Data Figure 8 Effects of chronic morphine treatment and in vivo optogenetic LTD induction on the strength of AMPAR and NMDAR current in MSNs receiving input from the PVT or BLA.
a, b, In the PVT→NAc pathway, chronic morphine treatment specifically increases AMPAR but not NMDAR current on D2-MSNs (saline/morphine, n = 13/16 cells) but not D1-MSNs (saline/morphine, n = 14/14 cells). Two-way ANOVA (AMPAR: F(1,53) = 5.24, P < 0.05; NMDAR: F(1,53) = 0.04, P = 0.83) followed by Tukey’s test. **P < 0.01. c, d, In the BLA→NAc pathway, chronic morphine treatment increases both AMPAR current (c) and NMDAR current (d) on D1-MSNs (saline/morphine, n = 14/13 cells) but not D2-MSNs (saline/morphine, n = 13/12 cells). Two-way ANOVA (AMPAR: F(1,51) = 7.06, P < 0.05; NMDAR: F(1,51) = 0.35, P = 0.55) followed by Tukey’s test. *P < 0.05. e, Chronic morphine treatment has no effect on the AMPAR/NMDAR ratio in either D1-MSNs or D2-MSNs in the BLA→NAc pathway. Two-way ANOVA (F(1,51) = 7.06, P < 0.05). f, g, In the PVT→NAc pathway, in vivo optogenetic stimulation (4 ms, 1 Hz, 900 pulses) in morphine-treated mice specifically decreases AMPAR but not NMDAR current in D2-MSNs (morphine/morphine + 1 Hz, n = 16/17 cells) but not D1-MSNs (morphine/morphine + 1 Hz, n = 14/14 cells). Two-way ANOVA (AMPAR: F(1,57) = 4.24, P = 0.04; NMDAR: F(1,57) = 0.01, P = 0.92) followed by Tukey’s test. *P < 0.05. Mean ± s.e.m.
Extended Data Figure 9 Chronic morphine treatment and in vivo optogenetic LTD induction does not affect paired-pulse ratio of MSNs receiving PVT input.
a, b, Example traces (a) and quantification (b) of paired-pulse ratio of photo-evoked EPSCs in D1- and D2-MSNs. An escalating regimen of morphine treatment and in vivo optogenetic stimulation (4 ms, 1 Hz, 900 pulses) in morphine-treated mice had no obvious effect on the paired-pulse ratio of MSNs receiving PVT input (two-way ANOVA, F(2,35) = 0.02, P = 0.97). D1-MSNs: saline/morphine/morphine + 1 Hz, n = 9/7/6; D2-MSNs: saline/morphine/morphine + 1 Hz, n = 7/6/6. Scale bars, 200 pA, 50 ms. Mean ± s.e.m.
Rights and permissions
About this article
Cite this article
Zhu, Y., Wienecke, C., Nachtrab, G. et al. A thalamic input to the nucleus accumbens mediates opiate dependence. Nature 530, 219–222 (2016). https://doi.org/10.1038/nature16954
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature16954
This article is cited by
-
GPR55 is expressed in glutamate neurons and functionally modulates drug taking and seeking in rats and mice
Translational Psychiatry (2024)
-
A quadruple dissociation of reward-related behaviour in mice across excitatory inputs to the nucleus accumbens shell
Communications Biology (2023)
-
Calcitonin receptor signaling in nucleus accumbens D1R- and D2R-expressing medium spiny neurons bidirectionally alters opioid taking in male rats
Neuropsychopharmacology (2023)
-
A paraventricular thalamus to insular cortex glutamatergic projection gates “emotional” stress-induced binge eating in females
Neuropsychopharmacology (2023)
-
Locus coeruleus input-modulated reactivation of dentate gyrus opioid-withdrawal engrams promotes extinction
Neuropsychopharmacology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.