Abstract
The p53 pro-apoptotic tumour suppressor is mutated or functionally altered in most cancers. In epithelial tumours induced by ‘high-risk’ mucosal human papilloma viruses, including human cervical carcinoma and a growing number of head-and-neck cancers1, p53 is degraded by the viral oncoprotein E6 (ref. 2). In this process, E6 binds to a short leucine (L)-rich LxxLL consensus sequence within the cellular ubiquitin ligase E6AP3. Subsequently, the E6/E6AP heterodimer recruits and degrades p53 (ref. 4). Neither E6 nor E6AP are separately able to recruit p53 (refs 3, 5), and the precise mode of assembly of E6, E6AP and p53 is unknown. Here we solve the crystal structure of a ternary complex comprising full-length human papilloma virus type 16 (HPV-16) E6, the LxxLL motif of E6AP and the core domain of p53. The LxxLL motif of E6AP renders the conformation of E6 competent for interaction with p53 by structuring a p53-binding cleft on E6. Mutagenesis of critical positions at the E6–p53 interface disrupts p53 degradation. The E6-binding site of p53 is distal from previously described DNA- and protein-binding surfaces of the core domain. This suggests that, in principle, E6 may avoid competition with cellular factors by targeting both free and bound p53 molecules. The E6/E6AP/p53 complex represents a prototype of viral hijacking of both the ubiquitin-mediated protein degradation pathway and the p53 tumour suppressor pathway. The present structure provides a framework for the design of inhibitory therapeutic strategies against oncogenesis mediated by human papilloma virus.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Bosch, F. X. et al. Comprehensive control of human papillomavirus infections and related diseases. Vaccine 31 (Suppl. 7), H1–H31 (2013)
Scheffner, M., Werness, B. A., Huibregtse, J. M., Levine, A. J. & Howley, P. M. The E6 oncoprotein encoded by human papillomavirus types 16 and 18 promotes the degradation of p53. Cell 63, 1129–1136 (1990)
Huibregtse, J. M., Scheffner, M. & Howley, P. M. Localization of the E6-AP regions that direct human papillomavirus E6 binding, association with p53, and ubiquitination of associated proteins. Mol. Cell. Biol. 13, 4918–4927 (1993)
Scheffner, M., Nuber, U. & Huibregtse, J. M. Protein ubiquitination involving an E1–E2–E3 enzyme ubiquitin thioester cascade. Nature 373, 81–83 (1995)
Ansari, T., Brimer, N. & Vande Pol, S. B. Peptide interactions stabilize and restructure human papillomavirus type 16 E6 to interact with p53. J. Virol. 86, 11386–11391 (2012)
Davey, N. E., Travé, G. & Gibson, T. J. How viruses hijack cell regulation. Trends Biochem. Sci. 36, 159–169 (2011)
Vande Pol, S. B. & Klingelhutz, A. J. Papillomavirus E6 oncoproteins. Virology 445, 115–137 (2013)
Zanier, K. et al. Structural basis for hijacking of cellular LxxLL motifs by papillomavirus E6 oncoproteins. Science 339, 694–698 (2013)
Gu, J., Rubin, R. M. & Yuan, Z. M. A sequence element of p53 that determines its susceptibility to viral oncoprotein-targeted degradation. Oncogene 20, 3519–3527 (2001)
Li, X. & Coffino, P. High-risk human papillomavirus E6 protein has two distinct binding sites within p53, of which only one determines degradation. J. Virol. 70, 4509–4516 (1996)
Bernard, X. et al. Proteasomal degradation of p53 by human papillomavirus E6 oncoprotein relies on the structural integrity of p53 core domain. PLoS ONE 6, e25981 (2011)
Zanier, K. et al. Solution structure analysis of the HPV16 E6 oncoprotein reveals a self-association mechanism required for E6-mediated degradation of p53. Structure 20, 604–617 (2012)
Ronchi, V. P., Klein, J. M., Edwards, D. J. & Haas, A. L. The active form of E6-associated protein (E6AP)/UBE3A ubiquitin ligase is an oligomer. J. Biol. Chem. 289, 1033–1048 (2014)
Ristriani, T., Fournane, S., Orfanoudakis, G., Travé, G. & Masson, M. A single-codon mutation converts HPV16 E6 oncoprotein into a potential tumor suppressor, which induces p53-dependent senescence of HPV-positive HeLa cervical cancer cells. Oncogene 28, 762–772 (2009)
Fu, L. et al. Degradation of p53 by human Alphapapillomavirus E6 proteins shows a stronger correlation with phylogeny than oncogenicity. PLoS ONE 5, e12816 (2010)
Mesplède, T. et al. p53 degradation activity, expression, and subcellular localization of E6 proteins from 29 human papillomavirus genotypes. J. Virol. 86, 94–107 (2012)
Hiller, T., Poppelreuther, S., Stubenrauch, F. & Iftner, T. Comparative analysis of 19 genital human papillomavirus types with regard to p53 degradation, immortalization, phylogeny, and epidemiologic risk classification. Cancer Epidemiol. Biomark. Prev. 15, 1262–1267 (2006)
Foster, S. A., Demers, G. W., Etscheid, B. G. & Galloway, D. A. The ability of human papillomavirus E6 proteins to target p53 for degradation in vivo correlates with their ability to abrogate actinomycin D-induced growth arrest. J. Virol. 68, 5698–5705 (1994)
Cooper, B. et al. Requirement of E6AP and the features of human papillomavirus E6 necessary to support degradation of p53. Virology 306, 87–99 (2003)
Liu, Y. et al. Multiple functions of human papillomavirus type 16 E6 contribute to the immortalization of mammary epithelial cells. J. Virol. 73, 7297–7307 (1999)
Nakagawa, S. et al. Mutational analysis of human papillomavirus type 16 E6 protein: transforming function for human cells and degradation of p53 in vitro. Virology 212, 535–542 (1995)
Zanier, K. et al. The E6AP binding pocket of the HPV16 E6 oncoprotein provides a docking site for a small inhibitory peptide unrelated to E6AP, indicating druggability of E6. PLoS ONE 9, e112514 (2014)
Stutz, C. et al. Intracellular analysis of the interaction between the human papillomavirus type 16 E6 oncoprotein and inhibitory peptides. PLoS ONE 10, e0132339 (2015)
Hengstermann, A., Linares, L. K., Ciechanover, A., Whitaker, N. J. & Scheffner, M. Complete switch from Mdm2 to human papillomavirus E6-mediated degradation of p53 in cervical cancer cells. Proc. Natl Acad. Sci. USA 98, 1218–1223 (2001)
Lilyestrom, W., Klein, M. G., Zhang, R., Joachimiak, A. & Chen, X. S. Crystal structure of SV40 large T-antigen bound to p53: interplay between a viral oncoprotein and a cellular tumor suppressor. Genes Dev. 20, 2373–2382 (2006)
Scheffner, M., Takahashi, T., Huibregtse, J. M., Minna, J. D. & Howley, P. M. Interaction of the human papillomavirus type 16 E6 oncoprotein with wild-type and mutant human p53 proteins. J. Virol. 66, 5100–5105 (1992)
Cherry, J. J. et al. Structure based identification and characterization of flavonoids that disrupt human papillomavirus-16 E6 function. PLoS ONE 8, e84506 (2013)
Malecka, K. A. et al. Identification and characterization of small molecule human papillomavirus E6 inhibitors. ACS Chem. Biol. 9, 1603–1612 (2014)
Moon, A. F., Mueller, G. A., Zhong, X. & Pedersen, L. C. A synergistic approach to protein crystallization: combination of a fixed-arm carrier with surface entropy reduction. Protein Sci. 19, 901–913 (2010)
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997)
McCoy, A. J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007)
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D 66, 213–221 (2010)
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004)
Chen, V. B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D 66, 12–21 (2010)
DeLano, W. L. The PyMOL molecular graphics system (DeLano Scientific, 2002)
Cassonnet, P. et al. Benchmarking a luciferase complementation assay for detecting protein complexes. Nature Methods 8, 990–992 (2011)
Wang, Y., Rosengarth, A. & Luecke, H. Structure of the human p53 core domain in the absence of DNA. Acta Crystallogr. D 63, 276–281 (2007)
Acknowledgements
This work received institutional support from le Centre National de la Recherche Scientifique (CNRS), Université de Strasbourg, l’Institut National de la Santé et de la Recherche Médicale (INSERM) and Région Alsace. The work was supported by grants from Ligue contre le Cancer, National Institutes of Health (grant R01CA134737 to S.V.P.), l’Agence Nationale de la Recherche (ANR-13-JSV8-004-01), Instruct (ESFRI), the French Infrastructure for Integrated Structural Biology (FRISBI) and Fondation pour La Recherche Medicale (fellowship to F.X.R.F.). We thank P. Poussin-Courmontagne, E. Ennifar, V. Olieric and B. Kieffer for advice. The authors declare that the content is solely their responsibility and does not represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
D.M.Z., J.P., A.M., J.R.R., A.F., A.C.S. and K.Z. performed experiments; F.X.R., A.P., D.M.Z. and K.Z. performed structure determination; D.M.Z., F.X.R., J.P., A.M., A.P. and K.Z. analysed the data; D.M.Z., J.P. and K.Z. prepared figures; K.Z. and GT wrote the manuscript together with comments from all authors; M.M., S.V.P., A.P., GT and K.Z. supervised the work.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Constructs of the minimal HPV-16 E6/E6AP/p53 ternary complex.
Green: E6AP residues 403–414 (e6ap 1–12) fused to the C terminus of MBP (pink) via a AAA linker (red); gold: the HPV-16 E6 4C/4S construct12 (named E6 from hereon) comprising four cysteine to serine substitutions (cyan marks) that suppress aggregation mediated by disulfide cross-bridging; blue: p53 core domain (p53core).
Extended Data Figure 2 Assembly of the E6/MBP-e6ap heterodimer.
a, Dynamic light scattering (DLS) analysis of E6/MBP-e6ap samples. Histograms report the average hydrodynamic radii of the particles, whereas error bars indicate size polydispersity. Numbers above the histograms indicate molecular mass estimates assuming a spherical model. This analysis shows that binding to MBP-e6ap enhances the solubility of E6, which, in the unbound state, displays a solubility threshold of 50 μM. However, in the case of E6/MBP-e6ap samples (grey histograms), particle size increases with raising concentration. By contrast, introduction of the F47R mutation in E6 (green histograms) stabilizes particle size to values close to what is expected for a simple heterodimer (∼60 kDa). Therefore we conclude that, despite the increase in solubility, E6/MBP-e6ap still undergoes weak self-association via the E6N region hosting Phe47 (see ref. 12 for further discussion). b, Gel filtration analysis of E6/MBP-e6ap samples. The elution volumes for molecular size markers are reported on top of the figure. The expected elution volumes of a simple E6/MBP-e6ap heterodimer (1×, 60 kDa) and of a dimer of heterodimers (2×, 120 kDa) are indicated. Note the relative small shift in the elution volumes of the different samples compared with the differences in the hydrodynamic radii (a). This suggests that oligomers of the E6/MBP-e6ap heterodimer are rather weak and dissociate on the gel filtration column. See also Supplementary Methods.
Extended Data Figure 3 Interaction of p53core with preformed E6/MBP-e6ap heterodimer.
a, Isothermal titration calorimetry experiments were performed by titrating increasing amounts of p53core into E6/MBP-e6ap heterodimer samples adjusted to a concentration of 45 μM, which limits heterodimer oligomerization. Note that the F47R mutation in E6 abolishes binding to p53core. See also Extended Data Table 1. b, Top: comparison of gel filtration elution profiles of E6/MBP-e6ap/p53core (red dashed line) versus E6/MBP-e6ap samples (black line). Both samples were adjusted to a concentration of 250 μM before loading onto the gel filtration column. The expected elution volumes for p53core (24 kDa), monomeric E6/MBP-e6ap heterodimer (60 kDa) and E6/MBP-e6ap/p53core ternary complex (80 kDa) are indicated. Bottom: SDS–PAGE analysis of fractions comprising the elution peak of the ternary complex. Note the significant shift in the elution volumes of the main peak in the two chromatograms, indicating formation of a ternary complex. See also Supplementary Methods.
Extended Data Figure 4 Comparison of the structures of the two E6/MBP-e6ap/p53core heterotrimers (heterotrimers A and B) observed in the asymmetric unit.
Green: e6ap fused to the C terminus of MBP (pink) via the AAA linker (red); gold: HPV-16 E6; blue: p53core; spheres: zinc atoms. a, Different orientation of MBP in the two heterotrimers, which results from the different conformations of the two AAA linkers. b, Superposition of the E6/e6ap/p53 regions of heterotrimers A (green/gold/blue) and B (grey). The backbone r.m.s.d. for the E6/e6ap/p53 regions of the two heterotrimers was calculated by aligning backbone atoms of residues (1) 12–136 of HPV-16 E6, (2) 371–379 of e6ap and (3) 109–191 of p53core and found to correspond to 0.9 Å. Regions displaying significant differences are boxed. These regions are the ill-defined C-terminal region of the e6ap peptide (see also Extended Data Fig. 6) and the a4 helix of E6, which is not involved in the E6–p53 interface.
Extended Data Figure 5 The structures of the E6/e6ap and p53core subunits of the ternary complex are superimposable with previously solved structures of the E6/e6ap heterodimer and p53core.
Left: superposition of the previously solved E6/e6ap heterodimer (grey)8 onto the E6/e6ap subunit of the ternary complex (heterotrimer A, gold/green) determined here. Dashed lines highlight regions of conformational change, namely the N terminus of HPV-16 E6 and the C terminus of the e6ap peptide. Right: superposition of previously solved p53core in the unbound state (heterotrimer A, grey)37 onto the p53core subunit of the ternary complex bound (blue).
Extended Data Figure 6 Contributions of the e6ap C-terminal region to ternary complex interface.
a, Sequence of wild-type e6ap peptides used in the study. Green: wild-type e6ap(1–12) corresponding to the peptide used for crystallization of the ternary complex. Grey: wild-type e6ap(1–16) containing a four-amino-acid C-terminal extension. b, Electron density (2Fo − Fc) map (heterotrimer A) contoured at 1 s level for e6ap (green, left) and for selected E6 (orange) and p53 (blue) interface residues in the proximity of the C terminus of e6ap (right). Note the lack of electron density data for e6ap residues Glu10, Glu11 and Arg12. Two conformations for the side chains Glu10 and Arg12 of e6ap and Arg131 of E6 are proposed in the model. c, Isothermal titration calorimetry curves showing the interactions of p53core with pre-formed E6/e6ap complexes bearing either point mutations within e6ap C terminus (E10A, E11A and R12A) or the wild-type e6ap(1–16) peptide construct. Note that neither the e6ap point mutations nor the e6ap C-terminal boundary affect the interaction between the E6/e6ap heterodimer and p53core. See also Extended Data Table 1.
Extended Data Figure 7 Interactions mediated by hrm-HPV-conserved residues shaping the conformation of the N-terminal arm of HPV-16 E6.
Whereas Phe2 contributes to tethering of the N-terminal region to the core of the E6N domain, residues Pro5, Arg8 and Tyr54 are involved in a triple stacking interaction.
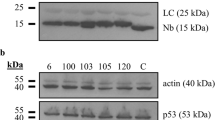
Extended Data Figure 8 Expression levels of HPV-16 E6, E6AP and p53 proteins in triple pull-down assays.
HEK293T were transfected with 1 μg of pSPICA-N1 plasmid (expressing E6AP fused to the C terminus of the G. princeps luciferase Gluc1 fragment), 1 μg pSPICA-N2 plasmid (expressing the E6 fused to the C terminus of the G. princeps luciferase Gluc2 fragment) and 1 μg of BioEase plasmid (expressing N-terminally biotinylated p53). Cell lysates were resolved by SDS–PAGE electrophoresis and E6, E6AP and p53 fusion proteins detected by western blotting. For gel source data, see Supplementary Fig. 1.
Supplementary information
Supplementary Information
This file contains Supplementary Methods. (PDF 94 kb)
Supplementary Figure 1
This file contains the source gel data for Figure 3 and Extended Data Figure 8. (PDF 18332 kb)
Source data
Rights and permissions
About this article
Cite this article
Martinez-Zapien, D., Ruiz, F., Poirson, J. et al. Structure of the E6/E6AP/p53 complex required for HPV-mediated degradation of p53. Nature 529, 541–545 (2016). https://doi.org/10.1038/nature16481
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature16481
This article is cited by
-
DCAF13 inhibits the p53 signaling pathway by promoting p53 ubiquitination modification in lung adenocarcinoma
Journal of Experimental & Clinical Cancer Research (2024)
-
The complexity of human papilloma virus in cancers: a narrative review
Infectious Agents and Cancer (2023)
-
Elucidating the clonal relationship of esophageal second primary tumors in patients with laryngeal squamous cell carcinoma
Infectious Agents and Cancer (2023)
-
MicroRNA-375 restrains the progression of lung squamous cell carcinoma by modulating the ERK pathway via UBE3A-mediated DUSP1 degradation
Cell Death Discovery (2023)
-
9-oxo-ODAs suppresses the proliferation of human cervical cancer cells through the inhibition of CDKs and HPV oncoproteins
Scientific Reports (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.