Abstract
Prevailing dogma holds that cell–cell communication through Notch ligands and receptors determines binary cell fate decisions during progenitor cell divisions, with differentiated lineages remaining fixed1. Mucociliary clearance2,3 in mammalian respiratory airways depends on secretory cells (club and goblet) and ciliated cells to produce and transport mucus. During development or repair, the closely related Jagged ligands (JAG1 and JAG2) induce Notch signalling to determine the fate of these lineages as they descend from a common proliferating progenitor4,5,6,7,8. In contrast to such situations in which cell fate decisions are made in rapidly dividing populations9,10, cells of the homeostatic adult airway epithelium are long-lived11,12,13, and little is known about the role of active Notch signalling under such conditions. To disrupt Jagged signalling acutely in adult mammals, here we generate antibody antagonists that selectively target each Jagged paralogue, and determine a crystal structure that explains selectivity. We show that acute Jagged blockade induces a rapid and near-complete loss of club cells, with a concomitant gain in ciliated cells, under homeostatic conditions without increased cell death or division. Fate analyses demonstrate a direct conversion of club cells to ciliated cells without proliferation, meeting a conservative definition of direct transdifferentiation14. Jagged inhibition also reversed goblet cell metaplasia in a preclinical asthma model, providing a therapeutic foundation15. Our discovery that Jagged antagonism relieves a blockade of cell-to-cell conversion unveils unexpected plasticity, and establishes a model for Notch regulation of transdifferentiation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Koch, U., Lehal, R. & Radtke, F. Stem cells living with a Notch. Development 140, 689–704 (2013)
Chilvers, M. A. & O’Callaghan, C. Local mucociliary defence mechanisms. Paediatr. Respir. Rev. 1, 27–34 (2000)
Roy, M. G. et al. Muc5b is required for airway defence. Nature 505, 412–416 (2014)
Zhang, S., Loch, A. J., Radtke, F., Egan, S. E. & Xu, K. Jagged1 is the major regulator of Notch-dependent cell fate in proximal airways. Dev. Dyn. 242, 678–686 (2013)
Morimoto, M., Nishinakamura, R., Saga, Y. & Kopan, R. Different assemblies of Notch receptors coordinate the distribution of the major bronchial Clara, ciliated and neuroendocrine cells. Development 139, 4365–4373 (2012)
Rock, J. R. et al. Notch-dependent differentiation of adult airway basal stem cells. Cell Stem Cell 8, 639–648 (2011)
Pardo-Saganta, A. et al. Injury induces direct lineage segregation of functionally distinct airway basal stem/progenitor cell subpopulations. Cell Stem Cell 16, 184–197 (2015)
Mori, M. et al. Notch3-Jagged signaling controls the pool of undifferentiated airway progenitors. Development 142, 258–267 (2015)
Kotton, D. N. & Morrisey, E. E. Lung regeneration: mechanisms, applications and emerging stem cell populations. Nature Med. 20, 822–832 (2014)
Hogan, B. L. M. L. M. M. L. M. et al. Repair and regeneration of the respiratory system: complexity, plasticity, and mechanisms of lung stem cell function. Cell Stem Cell 15, 123–138 (2014)
Rawlins, E. L. & Hogan, B. L. M. Ciliated epithelial cell lifespan in the mouse trachea and lung. Am. J. Physiol. Lung Cell. Mol. Physiol. 295, L231–L234 (2008)
Rock, J. R. & Hogan, B. L. M. Epithelial progenitor cells in lung development, maintenance, repair, and disease. Annu. Rev. Cell Dev. Biol. 27, 493–512 (2011)
Watson, J. K. et al. Clonal dynamics reveal two distinct populations of basal cells in slow-turnover airway epithelium. Cell Rep . 12, 90–101 (2015)
Rawlins, E. L. & Hogan, B. L. M. Epithelial stem cells of the lung: privileged few or opportunities for many? Development 133, 2455–2465 (2006)
Martin, C., Frija-Masson, J. & Burgel, P. R. Targeting mucus hypersecretion: New therapeutic opportunities for COPD? Drugs 74, 1073–1089 (2014)
Luca, V. C. et al. Structural basis for Notch1 engagement of Delta-like 4. Science 347, 847–853 (2015)
Cordle, J. et al. A conserved face of the Jagged/Serrate DSL domain is involved in Notch trans-activation and cis-inhibition. Nature Struct. Mol. Biol . 15, 849–857 (2008)
Kim, C. F. B. et al. Identification of bronchioalveolar stem cells in normal lung and lung cancer. Cell 121, 823–835 (2005)
Guha, A. et al. Neuroepithelial body microenvironment is a niche for a distinct subset of Clara-like precursors in the developing airways. Proc. Natl Acad. Sci. USA 109, 12592–12597 (2012)
Wu, Y. et al. Therapeutic antibody targeting of individual Notch receptors. Nature 464, 1052–1057 (2010)
Madisen, L. et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nature Neurosci. 13, 133–140 (2010)
Misaghi, S. et al. Polyclonal hyper-IgE mouse model reveals mechanistic insights into antibody class switch recombination. Proc. Natl Acad. Sci. USA 110, 15770–15775 (2013)
Chen, G. et al. SPDEF is required for mouse pulmonary goblet cell differentiation and regulates a network of genes associated with mucus production. J. Clin. Invest. 119, 2914–2924 (2009)
Pardo-Saganta, A., Law, B. M., Gonzalez-Celeiro, M., Vinarsky, V. & Rajagopal, J. Ciliated cells of pseudostratified airway epithelium do not become mucous cells after ovalbumin challenge. Am. J. Respir. Cell Mol. Biol. 48, 364–373 (2013)
Shin, Y. S., Takeda, K. & Gelfand, E. W. Understanding asthma using animal models. Allergy Asthma Immunol. Res. 1, 10–18 (2009)
Nials, A. T. & Uddin, S. Mouse models of allergic asthma: acute and chronic allergen challenge. Dis. Model. Mech . 1, 213–220 (2008)
Xing, Y., Li, A., Borok, Z., Li, C. & Minoo, P. NOTCH1 is required for regeneration of Clara cells during repair of airway injury. Stem Cells 30, 946–955 (2012)
Zheng, D., Yin, L. & Chen, J. Evidence for Scgb1a1+ cells in the generation of p63+ cells in the damaged lung parenchyma. Am. J. Respir. Cell Mol. Biol. 50, 595–604 (2014)
Rawlins, E. L. et al. The role of Scgb1a1+ Clara cells in the long-term maintenance and repair of lung airway, but not alveolar, epithelium. Cell Stem Cell 4, 525–534 (2009)
Pardo-Saganta, A. et al. Parent stem cells can serve as niches for their daughter cells. Nature 523, 597–601 (2015)
Lee, C. V., Sidhu, S. S. & Fuh, G. Bivalent antibody phage display mimics natural immunoglobulin. J. Immunol. Methods 284, 119–132 (2004)
Liang, W. C. et al. Function blocking antibodies to neuropilin-1 generated from a designed human synthetic antibody phage library. J. Mol. Biol. 366, 815–829 (2007)
Huntzicker, E. G. et al. Differential effects of targeting Notch receptors in a mouse model of liver cancer. Hepatology 61, 942–952 (2015)
Vonrhein, C. et al. Data processing and analysis with the autoPROC toolbox. Acta Crystallogr. D 67, 293–302 (2011)
Collaborative Computational Project, Number 4. The CCP4 suite: Programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994)
Adams, P. D. et al. PHENIX: A comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D 66, 213–221 (2010)
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010)
Bricogne G. et al. BUSTER version 2.11.2 (2011)
Laskowski, R. A., Moss, D. S. & Thornton, J. M. Main-chain bond lengths and bond angles in protein structures. J. Mol. Biol. 231, 1049–1067 (1993)
Chen, V. B. et al. MolProbity: All-atom structure validation for macromolecular crystallography. Acta Crystallogr. D 66, 12–21 (2010)
Schrödinger, L. The PyMOL Molecular Graphics System version 1.2r3pre edn
Ertürk, A., Lafkas, D. & Chalouni, C. Imaging cleared intact biological systems at a cellular level by 3DISCO. J. Vis. Exp . 89, 51382 (2014)
You, Y. & Brody, S. L. Culture and differentiation of mouse tracheal epithelial cells. Methods Mol. Biol. 945, 123–143 (2012)
Veta, M. et al. Automatic nuclei segmentation in H&E stained breast cancer histopathology images. PLoS ONE 8, e70221 (2013)
Acknowledgements
The authors thank E. Jackson and R. Pattni for work generating the Scgb1a1-ERT2GNE mice; L.Nguyen, L. Orellana, P. Grigg and the Genentech Transgenic Technology Laboratories and Research Support Facility for technical assistance with mouse strains and colonies; F. Chu, L. Rangell, S. Chalasani, C. Jones III and C. Espiritu for cell staining; A. Ertürk, C. Chalouni, S. Gierke, M. Gonzalez-Edick and the Genentech Center for Advanced Light Microscopy (CALM) for imaging; C. K. Poon for cytokine measurements; S. P. Tsai and M. Dostalek for pharmacokinetic analyses; T. Hagenbeek for help with the immune cell studies. Use of the Stanford Synchrotron Radiation Lightsource SSRL 12-12 at Stanford Linear Accelerator Center National Accelerator Laboratory is supported by the US Department of Energy (DOE), DOE Office of Biological and Environmental Research, National Institutes of Health, and National Institute of General Medical Sciences. The contents of this publication are the responsibility of the authors and do not necessarily represent the views of NIH or NIGMS.
Author information
Authors and Affiliations
Contributions
D.L. performed experiments and analysed data of Figs 2, 3, 4 and Extended Data Fig. 4, 5, 6, 7, 8, 9, 10, A.S. performed reporter assays for Fig. 1 and experiments for Extended Data Fig. 2c and mouse studies for Extended Data Fig. 4b, G.d.L.B. purified antibody fragments and crystallized the JAG1–Fab complex, Y. Chen and S.S.S performed affinity maturation and characterization of antibodies, C.C., S.W. and Y.W. generated the phage display antibodies and performed the in vitro binding experiments and affinity maturation, M.R. performed all electron microscopy studies, C.S. performed the cilia functionality studies, M.R.-G. and S.W. designed the targeting vector and supervised the generation of the Scgb1a1-CreERT2GNE mouse line, M.Z. and X.Wu performed the ovalbumin studies, J.E.-A. performed all quantifications of immunofluorescence staining, H.M. performed qPCR and analysis of whole lungs, Y.Chinn and J.Q.H. assisted with anti-JAG1 development and expressed and purified JAG1 protein; W.P.L. helped design and supervise the ovalbumin study, C.A. analysed tissue sections from the ovalbumin study, J.E. contributed to the design of qPCR and ovalbumin studies as well as contributing intellectually, J.P. solved and analysed the structure, and made the structural figures in Fig. 1 and Extended Data Fig. 3, J.B.L. analysed the histology of all lung and skin samples except samples from the ovalbumin study. C.W.S. supervised the experiments and wrote the paper with D.L.
Corresponding author
Ethics declarations
Competing interests
D.L., A.S., C.C., G.d.L.B., Y.Chen S.S.S., M.R., M.Z., J.E.-A., H.M., W.P.L., Y.Chinn, J.Q.H., C.A., J.E., Y.W., J.P., J.B.L. and C.W.S. are or were employed by Genentech Inc., which has commercial interests in some of the molecules described.
Extended data figures and tables
Extended Data Figure 1 Surface plasmon resonance affinity measurements of anti-JAG1.b70 and anti-JAG2.b33 binding.
a, Surface plasmon resonance (SPR) was used to determine anti-JAG1.b70 and anti-JAG2.b33 binding affinities to purified human (h) or mouse (m) JAG1 and JAG2 antigens. Representative curves from one assay run with three technical replicates are shown. At least two additional assays have been performed with binding to human JAG1 and JAG2 antigens, yielding consistent results. b, SPR binding constants. For human JAG1, human and mouse JAG2, human DLL1, and human and mouse DLL4, antibodies were coated onto a CM5 biosensor chip and the ligand was subsequently added for binding assessment. *By contrast, because mouse JAG1 and DLL1 showed some background binding to the empty flow cell, the antigens were coated directly onto the CM5 biosensor chip, and purified antibodies in Fab fragment format were subsequently added for binding assessment; steady-state measurements were used for the low-affinity binding of anti-JAG2.b33 to mouse JAG1. Data are mean ± s.d. of three technical replicates. See Methods for details.
Extended Data Figure 2 Notch ligand proteins used to characterize blocking antibodies, crystallographic data, and in vitro verification of lack of significant cross-reactivity of anti-JAG2.b33.
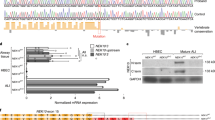
a, Protein reagents used to characterize anti-JAG1 and anti-JAG2 binding. Purified human and mouse JAG1, JAG2, DLL1 and DLL4 extracellular fragments (2–5 μg) used for antibody characterization studies were analysed by SDS–PAGE under non-reducing (NR) and reducing (R) conditions. b, Table of crystallographic data collection and refinement statistics. c, Immunoblot analysis of NOTCH2 intracellular domain (NICD2) in U87 glioblastoma cells to assess selectivity of JAG1 inhibition. JAG1 immobilized on beads was used to induce NOTCH2 signalling in U87 cells, which endogenously express high levels of NOTCH2, in the presence of the indicated reagents. NOTCH2 signalling was assessed using an antibody that preferentially recognizes the γ-secretase-cleaved (active) form of NICD2 (ref. 33). As a control, a γ-secretase inhibitor (DAPT, 5 μΜ in DMSO) inhibited signalling relative to control treatment (DMSO alone), evidenced by a clear decrease in NICD2 levels. Likewise, anti-JAG1.b70 (25 μg ml−1) completely blocked NOTCH2 signalling; by contrast, a high concentration of anti-JAG2.b33 (25 μg ml−1) did not detectably decrease NICD2 levels, consistent with our other data that this antibody does not inhibit JAG1 signalling despite low-affinity binding to JAG1. Asterisk indicates a nonspecific band. A full scan of the blot may be found in Supplementary Fig. 1.
Extended Data Figure 3 Anti-JAG1.b70 human–mouse cross-reactivity and inhibitory mechanism.
a, b, Amino acid sequence alignment of human and mouse JAG1 (a) and human JAG1 and JAG2 (b) showing the DSL through EGF3 domains. Asterisk (*) indicates identical amino acids; colon (:) denotes conservative difference; full stop (.) denotes semi-conservative difference; and blank space () denotes non-conservative difference. c, The anti-JAG1.b70 epitope on human JAG1 is highlighted pink, with residues unique to mouse JAG1 highlighted in green. Similarly, residues differing between JAG1 and JAG2 positions are highlighted yellow. d, The NOTCH1–DLL4 crystal structure (PDB 4XL1; ref. 16) was superimposed onto our JAG–anti-JAG1-Fab coordinates. The high structural similarity between DLL4 and JAG1 is evident within the DSL and EGF1 domains (root mean squared deviation (r.m.s.d.) < 1.1 Å). In this view, the DLL4 C2 domain and the anti-JAG1 Fab were omitted for clarity. e, A ~90° view relative to d, with the anti-JAG1 Fab shown in space-filling representation. Depicting the anti-JAG1.b70 Fab bound to JAG1 indicates that the antibody light chain (LC; cyan) would clash with Notch1 (arrow), thus supporting an inhibitory mechanism based on steric occlusion of receptor binding.
Extended Data Figure 4 JAG blocking antibodies inhibit JAG-induced Notch signalling in vivo.
a, Immunofluorescence staining of bronchiolar epithelium for CC10 (green) and FOXJ1 (red) from mice dosed every 3 days over 8 days with the indicated antibodies. Nuclei were counterstained with DAPI. b, Haematoxylin and eosin staining of skin from mice treated with control antibody, anti-JAG1.b70, anti-JAG2.b33 or anti-JAG1.b70 plus anti-JAG2.b33. JAG2 but not JAG1 inhibition induced a loss of mature sebocytes in sebaceous glands (arrows in control and anti-JAG1.b70 skin, left two panels) (n = 3 for each group). c, Immunofluorescence staining of bronchiolar epithelium for CC10 (green) and acetylated-α-tubulin (red) from mice treated with intranasal administration of control or anti-JAG1.b70 plus anti-JAG2.b33 antibodies for 5 days. Nuclei were labelled with DAPI (n = 3 for each group). d, Immunofluorescence staining for the acetylated-α-tubulin (red), the neuroepithelial-cell-specific marker CGRP (pink) and CC10 (green) from mice treated with anti-JAG1.b70 plus anti-JAG2.b33. Right panel shows a merged image and includes DAPI labelling. Club cells adjacent to neuroepithelial cells escape the JAG-blockade-induced transdifferentiation. e, Expression analysis of whole lungs from mice treated with the indicated antibodies (n = 3 for each group). Genes analysed included the Notch signalling targets Hes1, Hey1 and Hey2 as well as the club-cell-specific genes Scgb1a1, Muc5b, Muc5ac and the ciliated-cell-specific gene Foxj1. Both fold change and P values are plotted. Vertical and red horizontal dashed lines delineate twofold changes and P < 0.05, respectively. Anti-JAG1.b70 significantly increased expression of Foxj1 and reduced expression of Scgb1a1 by at least twofold. By contrast, no significant changes were observed when treating with anti-JAG2.b33 alone. The combination of anti-JAG1.b70 plus anti-JAG2.b33 significantly reduced expression of all Notch target genes analysed as well as club-cell-specific genes while increasing expression of Foxj1. Thus, the magnitude and type of gene expression changes revealed inhibition of Notch signalling and cell fate conversion in a manner that mirrored antibody induced cell fate changes assessed by immunofluorescence and other methods. Scale bars, 10 μm (a, c), 100 μm (b),
Extended Data Figure 5 Determination of the functionally relevant ligand-receptor pair in the adult airway.
a, Immunofluorescence staining for CC10 (green) and acetylated-α-tubulin (red) of mice treated with isotype control antibody, the NOTCH1 or NOTCH2 blocking antibodies anti-NRR1 or anti-NRR2, or the combination of anti-NRR1 plus anti-NRR2 (n = 3 for each group). NOTCH2 inhibition alone markedly reduced club cells numbers, with a corresponding increase in ciliated cell numbers, whereas NOTCH1 inhibition alone had little or no effect; dual blockade of both receptors induced the strongest phenotype. Results indicate that NOTCH2 is the dominant receptor controlling this phenotype, with NOTCH1 perhaps playing a secondary and functionally redundant role. b, c, Immunohistochemical staining of airways for JAG1 and NOTCH2 after control treatment (b) or JAG blockade (c) as in a revealed that JAG1 protein was most prominently expressed in ciliated cells, although a weak signal was observed throughout the epithelium. The JAG1 signal was strongly detected throughout the ciliated epithelium after JAG blockade, suggesting that the ciliated cells are the relevant signal-sending cell type. NOTCH2 protein staining showed a complementary pattern, with expression clearly localized to club cells and little or no signal detectable in the ciliated epithelium after JAG inhibition. d, Consistent with its secondary role relative to NOTCH2, NOTCH1 protein expression was very weak in the epithelial layer (top), with strong NOTCH1 staining on blood vessels from the same tissue sections serving as a positive control (bottom). e, Fluorescent RNAscope in situ hybridization to detect mRNA expression of Jag1, Jag2, Notch1 and Notch2 in mouse bronchiolar epithelium. Specific detection of each of the mouse NOTCH receptor and ligand mRNAs is shown in green, whereas mouse Foxj1 and Scgb1a1 mRNA, to mark ciliated and club cells, are shown in red and white, respectively. The signals using this method appear as coloured puncta. Samples were counterstained with DAPI to reveal nuclei. Consistent with their functioning as the primary ligand–receptor pair controlling cell fate, Jag1 and Notch2 were more highly expressed than Notch1 and Jag2, which was only weakly detectable (n = 4 for each probe). Jag1 and Jag2 signals appear in the same cells as Foxj1, indicating co-expression in ciliated cells (arrows, two left panels). By contrast, Notch1 and Notch2 signals appear in the same cells as Scgb1a1, indicating co-expression in club cells (arrows, two right panels). These results thus confirm the immunohistochemistry findings and extend the expression results to include Jag2, for which a reliable immunohistochemistry method is lacking. Scale bars, 20 μm (a), 10 μm (b–e).
Extended Data Figure 6 Lack of proliferation and intermediate cells are seen during transdifferentiation.
a, Representative immunofluorescence images of sections used for quantification shown in Fig. 2f, of the percentage of airway cells stained for the proliferation marker KI67 (white) (n = 5 mice per group). b, Representative immunofluorescence images (BrdU, green) of sections used for the quantification shown in Fig. 2h, of percentages of BrdU-positive airway cells (n = 5 mice per group). c, d, Immunofluorescence staining for CC10 (green), and acetylated-α-tubulin (red, c) or FOXJ1 (red, d) of bronchiolar epithelium from mice 5 days after treatment with anti-JAG1.b70 plus anti-JAG2.b33. e, Quantification of the percentage of intermediate cells over the number of CC10-positive cells, that appeared positive for both CC10 and FOXJ1 by immunofluorescence at day 5 of control treatment or JAG blockade (n = 4, mean ± s.d.; unpaired t-test, *P < 0.05). A significant percentage of club cells remaining after JAG blockade (17.0 ± 6.8%) expressed both CC10 and FOXJ1, a master transcription factor dictating ciliated differentiation. Such CC10+/FOXJ1+ cells were also detected in control lungs, but at significantly lower percentages (0.46 ± 0.09%). f, Immunogold transmitted electron microscopy for high-resolution detection of CC10 (18-nm gold particles, arrowheads) and acetylated-α-tubulin (12-nm gold particles, arrows) in cells from the bronchiolar epithelium. Images of control cells (left) consistently showed CC10 expression restricted to secretory vesicles of apparent club cells, adjacent to and distinct from ciliated cells that expressed acetylated-α-tubulin in the basal bodies and cilia, confirming the specificity of immunostaining. Treatment with anti-JAG1.b70 plus anti-JAG2.b33 yielded a fraction of cells that displayed both acetylated-α-tubulin at basal bodies and CC10 in the cytoplasm (right), consistent with a phenotypic intermediate expected during a club-to-ciliated cell conversion. Scale bars, 20 μm (a, b), 10 μm (c, d) and 1 μm (f).
Extended Data Figure 7 Lineage tracing shows that club cells transdifferentiate into ciliated cells.
a, Targeting construct to generate Scgb1a1-CreERT2GNE knock-in mice. PGK-neo and HSV-tk cassettes were used for positive and negative selection, respectively. See Methods for details. b, Whole-mount imaging of the right caudal lobe of Scgb1a1-CreERT2GNE/Rosa26-lsl-tdTomato mice after mice were treated as in Fig. 3a. Images shown are maximum z-projections of optical sections obtained with an ultramicroscope (n = 3 for each group). Scale bar, 1 mm. The overall signal from the club cell lineage trace appears approximately equal and well distributed throughout the lobe, even after conversion to ciliated cells (bottom), confirming the lack of any notable cell loss, including apoptosis, in transdifferentiating club cells. c, Lineage tracing as in Fig. 3b, except using FOXJ1 instead of acetylated-α-tubulin as the ciliated cell marker. Scale bar, 10 μm. d, Lineage tracing results demonstrating that inhibition of NOTCH1 plus NOTCH2 induces club-to-ciliated cell transdifferentiation. Scale bar, 10 μm. After treating mice with anti-NRR1 and anti-NRR2 blocking antibodies, immunofluorescence staining of the bronchiolar epithelium was performed as in c and Fig. 3b. After NOTCH1 plus NOTCH2 inhibition, the tdTomato-positive cells, marking the club cell lineage, express acetylated-α-tubulin (left) and FOXJ1 (right), and have thus assumed a ciliated cell identity.
Extended Data Figure 8 Club cells slowly reappear after antibody washout with expansion localized at the brochoalveolar duct junctions.
a, Re-establishment of normal club and ciliated cell patterning after transdifferentiation. Immunofluorescence staining for CC10 (green) and acetylated-α-tubulin (red) of mice treated with a single dose of isotype control antibody or anti-JAG1.b70 plus anti-JAG2.b33. Bronchiolar epithelia were analysed 1, 3, 6, 10 and 13 weeks after treatment, as indicated, to determine the time needed for club cells to reappear (n = 3 for each time point). The first signs of recovery were evident after 6 weeks, with increased but incomplete recovery observed after 13 weeks. Reappearing rows of club cells seemed to originate from brochoalveolar duct junctions (BAJs), resulting in a gradient of reestablishment from smaller to larger airways. b, Whole-mount imaging of the right caudal lobe of Scgb1a1-CreERT2GNE/Rosa26-lsl-tdTomato mice. After induction of lineage tracing with four doses of tamoxifen (200 mg kg−1), mice were treated with a single dose of isotype control antibody (left) or anti-JAG1.b70 plus anti-JAG2.b33 (right), 1 week after the last tamoxifen injection. Lungs were analysed 13 weeks after treatment. Images shown are maximum z-projections of optical sections obtained with an Ultramicroscope (n = 4 for each group). In lungs from mice treated with anti-JAG1.b70 plus anti-JAG2.b33, clusters of lineage-traced cells were observed at the BAJs (arrows in bottom right panel); such clusters are absent in control lungs. c, d, Immunofluorescence staining of bronchiolar epithelium for the lineage-tracing marker tdTomato (pink), CC10 (green) and FOXJ1 (red, c) or acetylated-α-tubulin (red, d), from same mice as in b. Whereas mostly single cells and small clones of lineage-traced cells are found at the BAJs of control lungs, large clones are seen in lungs after JAG blockade, confirming the pattern of reestablishment of club cells after treatment (a). Nuclei were labelled with DAPI. Scale bars, 20 μm (a), 1 mm (b, top), 0.5 mm (b, bottom) and 10 μm (c, d).
Extended Data Figure 9 NOTCH blockade inhibits goblet cell metaplasia in vivo.
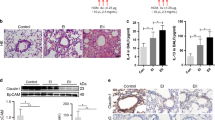
a, Mice were sensitized during a 35-day period after an i.p. injection of ovalbumin or vehicle (non-sensitized control) and then challenged with aerosolized ovalbumin for 7 consecutive days to induce inflammation and goblet cell metaplasia. Mice were also treated, at 1 and 4 days after challenge, with isotype control antibody, anti-JAG1.b70, anti-JAG2.b33 or anti-JAG1.b70 plus anti-JAG2.b33, as indicated (n = 6 mice per group, mean ± s.d.). b, Alcian blue/PAS staining of lung sections from mice treated with anti-JAG1.b70, anti-JAG2.b33 or the combination, on days 36 and 39, indicated as dose 1 and 2 in a. c, Quantification of goblet cell area (n = 6 for each group, mean ± s.d.; unpaired t-test, ***P < 0.001, **P < 0.01). d, Inflammation index as assessed by haematoxylin and eosin staining of lung sections. e, Alcian blue/PAS staining of lung sections from mice treated with anti-NRR1, anti-NRR2, or the combination, on days 36 and 39, indicated as dose 1 and 2 in a. f, Quantification of goblet cell area (n = 6 for each group, mean ± s.d.; unpaired t-test, ***P < 0.001). g, Inflammation index as assessed by haematoxylin and eosin staining of lung sections. All scale bars, 20 μm.
Extended Data Figure 10 Characterization of the immune response during the ovalbumin challenge.
a, Total numbers of immune cell populations found in the bronchoalveolar lavage fluid (BALF) of mice from the prevention goblet cell metaplasia study (Extended Data Fig. 9a–d). A significant reduction in both lymphocytes and macrophages is observed (n = 7 for each group, mean ± s.d.; unpaired t-test, *P < 0.05, **P < 0.01), but not in neutrophils and eosinophils, which are the most relevant cells that drive the metaplasia phenotype. b, Total numbers of immune cell populations found in the BALF of mice from the intervention goblet cell metaplasia study in Fig. 4. Although dual JAG blockade as well as blockade of JAG1 alone reverse goblet cell metaplasia (Fig. 4), a significant reduction in neutrophils was observed only after dual JAG blockade; no changes in other cell types, including eosinophils, were observed (n = 7 for each group, mean ± s.d.; unpaired t-test, **P < 0.01). c, Analysis of cytokine levels in blood serum (left) and BALF (right) of mice from the prevention study summarized in Extended Data Fig. 9a–d. Both antibody treatments resulted in a significant increase in the levels of IL4 in the BALF, although this increase was modest and not sustained to the later time point (n = 7 for each group; mean ± s.d.; unpaired t-test, *P < 0.05). d, Analysis of cytokine levels in blood serum (left) and BALF (right) of mice from the intervention study summarized in Fig. 4. Neither antibody treatment resulted in altered cytokine levels at this time point (n = 7 for each group; mean ± s.d., unpaired t-test). These results support an epithelial-cell-specific mechanism in which prevention and reversal of goblet cell metaplasia reflect direct effects of JAG inhibition on lung cell fate.
Supplementary information
Supplementary Figure 1
This file contains the image of the entire Western blot film used to compile Extended Data Figure 2c. (PDF 592 kb)
Supplementary Data
This file contains a spreadsheet showing the raw data used for quantifications in Fig. 2, 3 and Extended Data Fig. 6. (XLSX 16 kb)
Control for cilia motility
The video shows single tracheal cells from Scgb1a1-CreERT2GNE /Rosa26-lsl-tdTomato mice induced with four doses of 200mg kg-1 tamoxifen and treated with a single dose of control antibody for six days, one week after the last Tamoxifen injection. CC10-traced cells appear white. At least two ciliated cells (control, not labeled white and, thus, not converted from club cells following JAG blockade) with motile cilia are visible (right side). A tdTomato-positive (white) club cell (no cilia) is visible on the left. (MOV 5498 kb)
Cilia on transdifferentiated cells are motile
The video shows single tracheal cells from Scgb1a1-CreERT2GNE /Rosa26-lsl-tdTomato mice induced with four doses of 200mg kg-1 Tamoxifen and treated with a single dose of with anti-JAG1.b70 plus anti-JAG2.b33 for six days, one week after the last Tamoxifen injection. At least two ciliated cells (control, not labeled white and, thus, not converted from club cells following JAG blockade) with motile cilia are visible (middle). A tdTomato-positive cell (white), marking a daughter cell derived from the club cell lineage, has beating cilia (middle), supporting the notion that an induced ciliated cell functions normally, similar to the control (unlabelled) ciliated cells. (MOV 5967 kb)
Rights and permissions
About this article
Cite this article
Lafkas, D., Shelton, A., Chiu, C. et al. Therapeutic antibodies reveal Notch control of transdifferentiation in the adult lung. Nature 528, 127–131 (2015). https://doi.org/10.1038/nature15715
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature15715
This article is cited by
-
Notch signaling in thyrocytes is essential for adult thyroid function and mammalian homeostasis
Nature Metabolism (2023)
-
Antibody blockade of Jagged1 attenuates choroidal neovascularization
Nature Communications (2023)
-
A topographic atlas defines developmental origins of cell heterogeneity in the human embryonic lung
Nature Cell Biology (2023)
-
Stem cell-based therapy for pulmonary fibrosis
Stem Cell Research & Therapy (2022)
-
Roles of airway basal stem cells in lung homeostasis and regenerative medicine
Respiratory Research (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.