Abstract
Deep brain stimulation (DBS) has improved the prospects for many individuals with diseases affecting motor control, and recently it has shown promise for improving cognitive function as well. Several studies in individuals with Alzheimer disease and in amnesic rats have demonstrated that DBS targeted to the fimbria–fornix1,2,3, the region that appears to regulate hippocampal activity, can mitigate defects in hippocampus-dependent memory3,4,5. Despite these promising results, DBS has not been tested for its ability to improve cognition in any childhood intellectual disability disorder. Such disorders are a pressing concern: they affect as much as 3% of the population and involve hundreds of different genes. We proposed that stimulating the neural circuits that underlie learning and memory might provide a more promising route to treating these otherwise intractable disorders than seeking to adjust levels of one molecule at a time. We therefore studied the effects of forniceal DBS in a well-characterized mouse model of Rett syndrome (RTT), which is a leading cause of intellectual disability in females. Caused by mutations that impair the function of MeCP2 (ref. 6), RTT appears by the second year of life in humans, causing profound impairment in cognitive, motor and social skills, along with an array of neurological features7. RTT mice, which reproduce the broad phenotype of this disorder, also show clear deficits in hippocampus-dependent learning and memory and hippocampal synaptic plasticity8,9,10,11. Here we show that forniceal DBS in RTT mice rescues contextual fear memory as well as spatial learning and memory. In parallel, forniceal DBS restores in vivo hippocampal long-term potentiation and hippocampal neurogenesis. These results indicate that forniceal DBS might mitigate cognitive dysfunction in RTT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Laxton, A. W. et al. A phase I trial of deep brain stimulation of memory circuits in Alzheimer's disease. Ann. Neurol. 68, 521–534 (2010)
Hamani, C. et al. Memory enhancement induced by hypothalamic/fornix deep brain stimulation. Ann. Neurol. 63, 119–123 (2008)
Shirvalkar, P. R., Rapp, P. R. & Shapiro, M. L. Bidirectional changes to hippocampal theta-gamma comodulation predict memory for recent spatial episodes. Proc. Natl Acad. Sci. USA 107, 7054–7059 (2010)
Phillips, R. G. & LeDoux, J. E. Lesions of the fornix but not the entorhinal or perirhinal cortex interfere with contextual fear conditioning. J. Neurosci. 15, 5308–5315 (1995)
Maren, S. & Fanselow, M. S. Electrolytic lesions of the fimbria/fornix, dorsal hippocampus, or entorhinal cortex produce anterograde deficits in contextual fear conditioning in rats. Neurobiol. Learn. Mem. 67, 142–149 (1997)
Amir, R. E. et al. Rett syndrome is caused by mutations in X-linked MECP2, encoding methyl-CpG-binding protein 2. Nature Genet. 23, 185–188 (1999)
Chahrour, M. & Zoghbi, H. Y. The story of Rett syndrome: from clinic to neurobiology. Neuron 56, 422–437 (2007)
Guy, J., Gan, J., Selfridge, J., Cobb, S. & Bird, A. Reversal of neurological defects in a mouse model of Rett syndrome. Science 315, 1143–1147 (2007)
Chao, H. T. et al. Dysfunction in GABA signalling mediates autism-like stereotypies and Rett syndrome phenotypes. Nature 468, 263–269 (2010)
Moretti, P. et al. Learning and memory and synaptic plasticity are impaired in a mouse model of Rett syndrome. J. Neurosci. 26, 319–327 (2006)
Samaco, R. C. et al. Female Mecp2+/− mice display robust behavioral deficits on two different genetic backgrounds providing a framework for pre-clinical studies. Hum. Mol. Genet. 22, 96–109 (2013)
Freund, H. J. et al. Cognitive functions in a patient with Parkinson-dementia syndrome undergoing deep brain stimulation. Arch. Neurol. 66, 781–785 (2009)
Whittle, N. et al. Deep brain stimulation, histone deacetylase inhibitors and glutamatergic drugs rescue resistance to fear extinction in a genetic mouse model. Neuropharmacology 64, 414–423 (2013)
Suthana, N. et al. Memory enhancement and deep-brain stimulation of the entorhinal area. N. Engl. J. Med. 366, 502–510 (2012)
Gary-Bobo, E. & Bonvallet, M. Commissural projection to the amygdala through the fimbria fornix system in the cat. Exp. Brain Res. 27, 61–70 (1977)
Morris, R. G., Garrud, P., Rawlins, J. N. & O’Keefe, J. Place navigation impaired in rats with hippocampal lesions. Nature 297, 681–683 (1982)
Jones, M. W. et al. A requirement for the immediate early gene Zif268 in the expression of late LTP and long-term memories. Nature Neurosci. 4, 289–296 (2001)
van Praag, H., Kempermann, G. & Gage, F. H. Running increases cell proliferation and neurogenesis in the adult mouse dentate gyrus. Nature Neurosci. 2, 266–270 (1999)
Shors, T. J. et al. Neurogenesis in the adult is involved in the formation of trace memories. Nature 410, 372–376 (2001)
Stuchlik, A. Dynamic learning and memory, synaptic plasticity and neurogenesis: an update. Front. Behav. Neurosci. 8, 106 (2014)
Toda, H., Hamani, C., Fawcett, A. P., Hutchison, W. D. & Lozano, A. M. The regulation of adult rodent hippocampal neurogenesis by deep brain stimulation. J. Neurosurg. 108, 132–138 (2008)
Encinas, J. M., Hamani, C., Lozano, A. M. & Enikolopov, G. Neurogenic hippocampal targets of deep brain stimulation. J. Comp. Neurol. 519, 6–20 (2011)
Stone, S. S. et al. Stimulation of entorhinal cortex promotes adult neurogenesis and facilitates spatial memory. J. Neurosci. 31, 13469–13484 (2011)
Wenk, G. L., Naidu, S., Casanova, M. F., Kitt, C. A. & Moser, H. Altered neurochemical markers in Rett’s syndrome. Neurology 41, 1753–1756 (1991)
Wichmann, T. & Delong, M. R. Deep brain stimulation for neurologic and neuropsychiatric disorders. Neuron 52, 197–204 (2006)
Cif, L. et al. Antero-ventral internal pallidum stimulation improves behavioral disorders in Lesch-Nyhan disease. Mov. Disord. 22, 2126–2129 (2007)
Deon, L. L., Kalichman, M. A., Booth, C. L., Slavin, K. V. & Gaebler-Spira, D. J. Pallidal deep-brain stimulation associated with complete remission of self-injurious behaviors in a patient with Lesch–Nyhan syndrome: a case report. J. Child Neurol. 27, 117–120 (2012)
Franzini, A., Broggi, G., Cordella, R., Dones, I. & Messina, G. Deep-brain stimulation for aggressive and disruptive behavior. World Neurosurg. 80, S29.e11–s29.e14 (2013)
Malenka, R. C. & Bear, M. F. LTP and LTD: an embarrassment of riches. Neuron 44, 5–21 (2004)
Ramocki, M. B. & Zoghbi, H. Y. Failure of neuronal homeostasis results in common neuropsychiatric phenotypes. Nature 455, 912–918 (2008)
Paxinos, G. & Franklin, K. B. J. The Mouse Brain in Stereotaxic Coordinates (Academic Press, 2001)
Laxton, A. W., Lipsman, N. & Lozano, A. M. Deep brain stimulation for cognitive disorders. Handb. Clin. Neurol. 116, 307–311 (2013)
Maren, S., De Oca, B. & Fanselow, M. S. Sex differences in hippocampal long-term potentiation (LTP) and Pavlovian fear conditioning in rats: positive correlation between LTP and contextual learning. Brain Res. 661, 25–34 (1994)
Corcoran, K. A. & Maren, S. Hippocampal inactivation disrupts contextual retrieval of fear memory after extinction. J. Neurosci. 21, 1720–1726 (2001)
Moy, S. S. et al. Mouse behavioral tasks relevant to autism: phenotypes of 10 inbred strains. Behav. Brain Res. 176, 4–20 (2007)
Huang, H. S. et al. Behavioral deficits in an Angelman syndrome model: effects of genetic background and age. Behav. Brain Res. 243, 79–90 (2013)
Bouwknecht, J. A. & Paylor, R. Behavioral and physiological mouse assays for anxiety: a survey in nine mouse strains. Behav. Brain Res. 136, 489–501 (2002)
Shahbazian, M. et al. Mice with truncated MeCP2 recapitulate many Rett syndrome features and display hyperacetylation of histone H3. Neuron 35, 243–254 (2002)
Nadler, J. J. et al. Automated apparatus for quantitation of social approach behaviors in mice. Genes Brain Behav. 3, 303–314 (2004)
Marsicano, G. et al. The endogenous cannabinoid system controls extinction of aversive memories. Nature 418, 530–534 (2002)
Davis, S., Bliss, T. V., Dutrieux, G., Laroche, S. & Errington, M. L. Induction and duration of long-term potentiation in the hippocampus of the freely moving mouse. J. Neurosci. Methods 75, 75–80 (1997)
Tang, J. & Dani, J. A. Dopamine enables in vivo synaptic plasticity associated with the addictive drug nicotine. Neuron 63, 673–682 (2009)
Malleret, G. et al. Inducible and reversible enhancement of learning, memory, and long-term potentiation by genetic inhibition of calcineurin. Cell 104, 675–686 (2001)
Nokia, M. S., Anderson, M. L. & Shors, T. J. Chemotherapy disrupts learning, neurogenesis and theta activity in the adult brain. Eur. J. Neurosci. 36, 3521–3530 (2012)
Nokia, M. S., Sisti, H. M., Choksi, M. R. & Shors, T. J. Learning to learn: theta oscillations predict new learning, which enhances related learning and neurogenesis. PLoS ONE 7, e31375 (2012)
Jafari-Sabet, M. NMDA receptor blockers prevents the facilitatory effects of post-training intra-dorsal hippocampal NMDA and physostigmine on memory retention of passive avoidance learning in rats. Behav. Brain Res. 169, 120–127 (2006)
Jiao, R., Yang, C., Zhang, Y., Xu, M. & Yang, X. Cholinergic mechanism involved in the nociceptive modulation of dentate gyrus. Biochem. Biophys. Res. Commun. 379, 975–979 (2009)
Acknowledgements
We thank M. Xue, M. C. Weston and V. Brandt for comments on the manuscript, members of the Zoghbi laboratory for helpful discussions, and C. M. Spencer, C. T. Wotjak, F. Wei and D. Yu for technical suggestions. This work was supported by the W. M. Keck Foundation (H.Y.Z. and J.T.), the Cockrell Family Foundation, the Rett Syndrome Research Trust, Carl. C. Anderson, Sr. and Marie Jo Anderson Charitable Foundation, R01NS057819 (H.Y.Z.), and the Howard Hughes Medical Institute (H.Y.Z.), DP5OD009134 (R.C.S), R25 N070694 (A.J.P.) and in part by the Neuroconnectivity Core, Mouse Neurobehavioral Core, and Neurovisualization Core of IDDRC at Baylor College of Medicine (U54 HD083092 from the Eunice Kennedy Shriver National Institute of Child Health & Human Development), and the C06RR029965 grant from the National Center for Research Resources.
Author information
Authors and Affiliations
Contributions
J.T. and H.Y.Z. designed the experiments. S.H., B.T., Z.W., Y.S., H.T., Y.G., K.U. and J.T. performed the research. S.H., B.T., K.U., H.Y.Z. and J.T. analysed and interpreted the data. R.C.S., A.J.P. and D.J.C. provided comments on the manuscript. S.H., H.Y.Z. and J.T. wrote and edited the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 2 Fear memory in RTT mice and wild-type control animals.
All mice were trained with tone–foot-shock pairings on day 0. Memory retention was tested 3 h, 1 day, 3 day, and 7 day after training. a, b, Impaired fear memory in RTT mice (n = 20) compared to wild-type (WT) littermates (n = 20). These animals were implanted with electrodes but did not receive DBS or sham treatment. A significant main effect of genotype was observed (two-way repeated-measures ANOVA followed by Tukey’s post hoc test: context, F1,38 = 15.32, P < 0.001; cue, F1,38 = 20.70, P < 0.001). *P < 0.05; **P < 0.01; ***P < 0.001 versus wild type. c, d, Cued fear memory in RTT mice (n = 20) and wild-type littermates (n = 20) that were implanted with electrodes but without DBS or sham treatment. During the retention test, freezing in the tone phase (T) was significantly more than in the no tone phase (NT) across all the test time points in both wild-type (c) and RTT mice (d). e–h, Retrieval of cue fear memory in DBS- or sham-treated RTT and wild-type mice. During the cued memory test, all four groups of animals actively responded to the tone presentation (WT-sham, n = 21; WT-DBS, n = 21; RTT-sham, n = 14; RTT-DBS, n = 17). There was a significant increase of freezing time in the tone phase (T) compared to the no-tone phase (NT) at each of the test time points over all the groups. *P < 0.05, **P < 0.01, ***P < 0.001 (two-tailed paired t-test). All data are presented as mean ± s.e.m.
Extended Data Figure 3 Increased handling, but not forniceal DBS, increased locomotor activity and decreased the anxiety level in RTT and wild-type mice.
a, There was no difference among the four DBS/sham-treated groups in the total distance travelled in the open-field test (WT-sham, n = 20; WT-DBS, n = 20; RTT-sham, n = 17; RTT-DBS, n = 18; genotype, F1,71 = 1.13, P = 0.292; treatment, F1,71 = 0.13, P = 0.724; genotype × treatment, F1,71 = 0.063, P = 0.803). RTT and wild-type mice that received DBS/sham treatment travelled longer distances than RTT (n = 20) and wild-type (n = 20) animals that were implanted with electrodes but did not experience DBS/sham procedures, respectively. b, During the open-field test, there was no difference in the centre:total distance ratio among the four DBS groups (genotype, F1,71 = 1.22, P = 0.273; treatment, F1,71 = 0.0079, P = 0.93; genotype × treatment, F1,71 = 0.081, P = 0.777). Both RTT and wild-type mice that received DBS/sham treatment travelled more in the centre area compared to implanted RTT and wild-type animals that did not recieve DBS/sham procedures. c, In the light/dark test there was no difference in the amount of time spent in the light compartment among the four chronically treated groups (n = 12 per group; two-way ANOVA: genotype, F1,44 = 1.83, P = 0.183; treatment, F1,44 = 0.057, P = 0.813; genotype × treatment, F1,44 = 0.33, P = 0.567). Both RTT and wild-type mice that received DBS/sham treatment spent more time in the light compartment than implanted RTT (n = 15) and wild-type (n = 14) animals that did not receive DBS/sham procedures. *P < 0.05,0 **P < 0.01, ***P < 0.001 (two-tailed t-test). All data are presented as mean ± s.e.m.
Extended Data Figure 4 Forniceal DBS did not alter the pain threshold, motor function or social behaviour in RTT or wild-type mice.
a, There was no group difference in foot shock threshold intensities to evoke flinch, vocalization or jumping (WT-sham, n = 14; WT-DBS, n = 14; RTT-sham, n = 11; RTT-DBS, n = 12; two-way ANOVA, no significant main effect of genotype, treatment, or genotype × treatment interaction, P > 0.05). b, In a rotarod test (n = 12 mice per group), latency to fall increased over trials but there was no difference among the four groups (two-way repeated measures ANOVA: group, F3,44 = 1.68, P = 0.184; trial, F7,308 = 34.26, P < 0.001; group × trial interaction, F21,308 = 1.22, P = 0.230). c, RTT mice showed decreased latency to fall in the wire-hang test compared to wild-type animals, but there was no difference between DBS- and sham-treated groups for either RTT or wild-type mice (n = 12 per group; two-way ANOVA: genotype, F1,44 = 10.41, P = 0.002; treatment, F1,44 = 0.33, P = 0.566; genotype × treatment interaction, F1,44 = 0.75, P = 0.392). d, RTT mice showed a decreased latency to fall in the dowel test compared to wild-type animals, but there was no difference between DBS- and sham-treated groups for either genotype (n = 12 per group; genotype, F1,44 = 23.63, P < 0.001; treatment, F1,44 = 0.0018, P = 0.966; genotype × treatment interaction, F1,44 = 0.83, P = 0.367). e, f, In the three chamber test, all four groups of animals (n = 12 per group) showed a clear preference for the partner mice compared to the object (e). Two-way ANOVA revealed a significant genotype main effect of the interaction time with the partner mice (F1,44 = 4.56, P = 0.038), indicating altered social behaviour in RTT mice (P = 0.063, RTT-sham versus WT-sham, Tukey’s post hoc). However, DBS did not change the interaction time with the partners (treatment, F1,44 = 0.28, P = 0.597; genotype × treatment interaction, F1,44 = 0.31, P = 0.579) or the object (treatment, F1,44 = 2.64, P = 0.111; genotype × treatment interaction, F1,44 = 0.015, P = 0.905) (f). **P < 0.01, ***P < 0.001 (Tukey’s post hoc in c, d; two-tailed paired t-test in e). All data are presented as mean ± s.e.m.
Extended Data Figure 5 Forniceal DBS did not alter the body weight, visual or sensorimotor skills in RTT or wild-type mice.
a, All four groups (n = 12 mice per group) showed changes in body weight over time. Two-way repeated measure ANOVA revealed a significant main effect of group (F3,44 = 6.73, P < 0.001) and age (F4,176 = 89.32, P < 0.001). Tukey’s post hoc showed that sham-treated RTT mice were significantly heavier than sham-treated wild-type mice (P = 0.015), but there was no difference in body weight between sham-treated and DBS-treated wild-type mice (P = 0.861) or between sham-treated and DBS-treated RTT mice (P = 0.099). b, Comparison of body weight at the age of 23 weeks among the four groups (two-way ANOVA: genotype, F1,44 = 10.06, P = 0.003; treatment: F1,44 = 1.93, P = 0.172). c–e, Swimming test in the water maze task with a flagged platform (n = 18 mice per group). Sham-treated RTT mice did not have different escape latencies than sham-treated wild-type controls (c, two-way repeated-measures ANOVA: genotype, F1,34 = 1.73, P = 0.197; genotype × treatment interaction, F1,34 = 0.133, P = 0.718). DBS did not change the escape latencies in either wild-type controls (d; treatment, F1,34 = 0.44, P = 0.513; treatment × day interaction, F1,34 = 1.24, P = 0.273) or RTT mice (e, treatment, F1,34 = 2.36, P = 0.134; treatment × day interaction, F1,34 = 0.41, P = 0.524). *P < 0.05; n.s., not significant (Tukey’s post hoc). All data are presented as mean ± s.e.m.
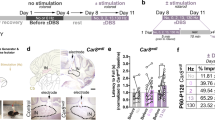
Extended Data Figure 6 Effect of forniceal DBS on hippocampal electrophysiological signatures.
a, Representative traces of LFPs recorded in the dentate gyrus 1 day before and 3 weeks after DBS/sham treatment. There were no electrographic seizure spikes in any of the four groups of mice after DBS/sham treatment. Scale bars: 10 s, 1 mV. b, Input–output (I/O) curves of the evoked responses of the perforant path recorded in the dentate gyrus in DBS/sham-treated mice. For each of the four groups, I/O curves were generated 1 day before and 3 weeks after forniceal DBS. All data points were normalized to the maximum value of the population spike amplitude before DBS/sham and the abscissa represents the seven increments used in each mouse. The I/O relationship was not altered by DBS in sham-treated wild-type mice (WT-sham; n = 5, F1,4 = 0.062, P = 0.818), DBS-treated wild-type mice (WT-DBS; n = 4, F1,3 = 0.036, P = 0.861), or sham-treated RTT mice (RTT-sham; n = 5, F1,4 = 0.018, P = 0.901). DBS reduced the amplitude of the evoked population spikes from the baseline test in DBS-treated RTT mice (RTT-DBS; n = 5, F1,4 = 6.73, P = 0.060). *P < 0.05 (Tukey’s post hoc). All data are presented as mean ± s.e.m.
Extended Data Figure 7 Unilateral forniceal DBS induces neuronal activity and stimulates neurogenesis bilaterally in the dentate gyrus.
a, Representative images showing that expression of the Fos gene was increased following forniceal DBS in wild-type and RTT mice compared to their sham controls, respectively (percentage of ipsilateral c-Fos-positive cells over the dentate granule cells: WT-sham, 0.26 ± 0.04%; WT-DBS, 34.52 ± 4.62%; RTT-sham, 0.30 ± 0.05%; RTT-DBS, 32.55 ± 3.74%). b, Representative images showing that there were more BrdU+ (green), DCX+ (red), and merged (yellow) cells in the dentate gyrus in forniceal DBS-treated wild-type and RTT mice than in their respective sham controls. Scale bar, 100 µm. Con, contralateral; Ips, ipsilateral.
Extended Data Figure 8 The cholinergic antagonist atropine did not alter forniceal DBS-induced enhancement of fear memory.
a, Placement of guide cannula and recording electrode into the dorsal hippocampus. b, Hippocampal infusion of 1.0 µg atropine did not change the amplitudes of the evoked potentials of the FFx recorded in the dentate gyrus in both RTT and wild-type mice. There was no difference of the population spike amplitudes before or after atropine infusion in both RTT mice (n = 5; one-way ANOVA, F9,36 = 0.69, P = 0.715) and wild-type controls (n = 3; F9,18 = 0.99, P = 0.485). c, Representative hippocampal EEG traces before and after vehicle (V) or atropine (A) infusion. Scale bars: 0.5 s, 0.2 mV. d, RTT mice (n = 17) showed less spontaneous hippocampal theta activity than wild-type animals (n = 20) (**P < 0.01, two-tailed t-test). e, Hippocampal infusion of atropine, but not vehicle, reduced hippocampal theta oscillation in both RTT and wild-type mice compared to their pre-infusion baselines (WT-V, n = 9; WT-A, n = 11; RTT-V, n = 8; RTT-A, n = 9; *P < 0.05, two-tailed paired t-test; n.s., not significant). f, Hippocampal microinfusion of atropine before fear conditioning training did not alter fear memory in forniceal DBS treated RTT mice or wild-type controls. Mice in all four groups (WT-V, n = 10; WT-A, n = 11; RTT-V, n = 12; RTT-A, n = 13) experienced 2 weeks of forniceal DBS that was finished 3 weeks before fear conditioning training. Atropine or vehicle was bilaterally infused into the dorsal hippocampus before training. Memory retention was tested 24 h after training. Two-way ANOVA revealed a significant main effect of genotype (F1,42 = 10.27, P = 0.003), but there was no difference between atropine- and vehicle-treated mice (treatment, F1,42 = 0.34, P = 0.562; genotype × treatment interaction, F1,42 = 0.069, P = 0.794). Atropine did not change cued fear memory, either: two-way ANOVA revealed no difference between genotypes (F1,42 = 2.99, P = 0.091) or between atropine- and vehicle-treated mice (treatment, F1,42 = 0.046, P = 0.831; genotype × treatment interaction, F1,42 = 0.154, P = 0.697). *P < 0.05; n.s., not significant (Tukey’s post hoc). g, Intra-hippocampal atropine infusion alone did not change the basal level of freezing in the contextual test environment in either wild-type or RTT mice. There was no difference between vehicle- (n = 9) or atropine-treated (n = 6) mice (P > 0.05, two-tailed t-test). h, Schematic representation of the dorsal hippocampus at seven rostral-caudal planes (according to ref. 31) for the microinfusion sites in DBS-treatment experiments. The numbers on the left represent the posterior coordinate from the bregma. All data are presented as mean ± s.e.m.
Rights and permissions
About this article
Cite this article
Hao, S., Tang, B., Wu, Z. et al. Forniceal deep brain stimulation rescues hippocampal memory in Rett syndrome mice. Nature 526, 430–434 (2015). https://doi.org/10.1038/nature15694
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature15694
This article is cited by
-
Deep brain stimulation creates informational lesion through membrane depolarization in mouse hippocampus
Nature Communications (2022)
-
Does the Application of Deep Brain Stimulation to Modulate Memory and Neural Circuity in AD Hold Substantial Promise?
Neuroscience Bulletin (2022)
-
The role of GABAergic signalling in neurodevelopmental disorders
Nature Reviews Neuroscience (2021)
-
Presymptomatic training mitigates functional deficits in a mouse model of Rett syndrome
Nature (2021)
-
The effect of fornix deep brain stimulation in brain diseases
Cellular and Molecular Life Sciences (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.