Abstract
The most abundant mRNA post-transcriptional modification is N6-methyladenosine (m6A), which has broad roles in RNA biology1,2,3,4,5. In mammalian cells, the asymmetric distribution of m6A along mRNAs results in relatively less methylation in the 5′ untranslated region (5′UTR) compared to other regions6,7. However, whether and how 5′UTR methylation is regulated is poorly understood. Despite the crucial role of the 5′UTR in translation initiation, very little is known about whether m6A modification influences mRNA translation. Here we show that in response to heat shock stress, certain adenosines within the 5′UTR of newly transcribed mRNAs are preferentially methylated. We find that the dynamic 5′UTR methylation is a result of stress-induced nuclear localization of YTHDF2, a well-characterized m6A ‘reader’. Upon heat shock stress, the nuclear YTHDF2 preserves 5′UTR methylation of stress-induced transcripts by limiting the m6A ‘eraser’ FTO from demethylation. Remarkably, the increased 5′UTR methylation in the form of m6A promotes cap-independent translation initiation, providing a mechanism for selective mRNA translation under heat shock stress. Using Hsp70 mRNA as an example, we demonstrate that a single m6A modification site in the 5′UTR enables translation initiation independent of the 5′ end N7-methylguanosine cap. The elucidation of the dynamic features of 5′UTR methylation and its critical role in cap-independent translation not only expands the breadth of physiological roles of m6A, but also uncovers a previously unappreciated translational control mechanism in heat shock response.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Meyer, K. D. & Jaffrey, S. R. The dynamic epitranscriptome: N6-methyladenosine and gene expression control. Nature Rev. Mol. Cell Biol. 15, 313–326 (2014)
Fu, Y., Dominissini, D., Rechavi, G. & He, C. Gene expression regulation mediated through reversible m6A RNA methylation. Nature Rev. Genet. 15, 293–306 (2014)
Wang, X. et al. N6-methyladenosine-dependent regulation of messenger RNA stability. Nature 505, 117–120 (2014)
Liu, N. et al. N6-methyladenosine-dependent RNA structural switches regulate RNA-protein interactions. Nature 518, 560–564 (2015)
Wang, X. et al. N6-methyladenosine modulates messenger RNA translation efficiency. Cell 161, 1388–1399 (2015)
Meyer, K. D. et al. Comprehensive analysis of mRNA methylation reveals enrichment in 3′ UTRs and near stop codons. Cell 149, 1635–1646 (2012)
Dominissini, D. et al. Topology of the human and mouse m6A RNA methylomes revealed by m6A-seq. Nature 485, 201–206 (2012)
Jia, G. et al. N6-methyladenosine in nuclear RNA is a major substrate of the obesity-associated FTO. Nature Chem. Biol. 7, 885–887 (2011)
Zheng, G. et al. ALKBH5 is a mammalian RNA demethylase that impacts RNA metabolism and mouse fertility. Mol. Cell 49, 18–29 (2013)
Qian, S. B., McDonough, H., Boellmann, F., Cyr, D. M. & Patterson, C. CHIP-mediated stress recovery by sequential ubiquitination of substrates and Hsp70. Nature 440, 551–555 (2006)
Schwartz, S. et al. Perturbation of m6A writers reveals two distinct classes of mRNA methylation at internal and 5′ sites. Cell Rep. 8, 284–296 (2014)
Schwartz, S. et al. High-resolution mapping reveals a conserved, widespread, dynamic mRNA methylation program in yeast meiosis. Cell 155, 1409–1421 (2013)
Anckar, J. & Sistonen, L. Regulation of HSF1 function in the heat stress response: implications in aging and disease. Annu. Rev. Biochem. 80, 1089–1115 (2011)
Mendillo, M. L. et al. HSF1 drives a transcriptional program distinct from heat shock to support highly malignant human cancers. Cell 150, 549–562 (2012)
Jackson, R. J., Hellen, C. U. & Pestova, T. V. The mechanism of eukaryotic translation initiation and principles of its regulation. Nature Rev. Mol. Cell Biol. 11, 113–127 (2010)
Hinnebusch, A. G. The scanning mechanism of eukaryotic translation initiation. Annu. Rev. Biochem. 83, 779–812 (2014)
Spriggs, K. A., Bushell, M. & Willis, A. E. Translational regulation of gene expression during conditions of cell stress. Mol. Cell 40, 228–237 (2010)
Panniers, R. Translational control during heat shock. Biochimie 76, 737–747 (1994)
Richter, K., Haslbeck, M. & Buchner, J. The heat shock response: life on the verge of death. Mol. Cell 40, 253–266 (2010)
McGarry, T. J. & Lindquist, S. The preferential translation of Drosophila hsp70 mRNA requires sequences in the untranslated leader. Cell 42, 903–911 (1985)
Klemenz, R., Hultmark, D. & Gehring, W. J. Selective translation of heat shock mRNA in Drosophila melanogaster depends on sequence information in the leader. EMBO J. 4, 2053–2060 (1985)
Rubtsova, M. P. et al. Distinctive properties of the 5′-untranslated region of human hsp70 mRNA. J. Biol. Chem. 278, 22350–22356 (2003)
Sun, J., Conn, C. S., Han, Y., Yeung, V. & Qian, S. B. PI3K–mTORC1 attenuates stress response by inhibiting cap-independent Hsp70 translation. J. Biol. Chem. 286, 6791–6800 (2011)
Zhang, X. et al. Translational control of the cytosolic stress response by mitochondrial ribosomal protein L18. Nature Struct. Mol. Biol. 22, 404–410 (2015)
Harcourt, E. M., Ehrenschwender, T., Batista, P. J., Chang, H. Y. & Kool, E. T. Identification of a selective polymerase enables detection of N6-methyladenosine in RNA. J. Am. Chem. Soc. 135, 19079–19082 (2013)
Kershaw, C. J. & O’Keefe, R. T. Splint ligation of RNA with T4 DNA ligase. Methods Mol. Biol. 941, 257–269 (2012)
Stark, M. R., Pleiss, J. A., Deras, M., Scaringe, S. A. & Rader, S. D. An RNA ligase-mediated method for the efficient creation of large, synthetic RNAs. RNA 12, 2014–2019 (2006)
Pelletier, J. & Sonenberg, N. Internal initiation of translation of eukaryotic mRNA directed by a sequence derived from poliovirus RNA. Nature 334, 320–325 (1988)
Hellen, C. U. & Sarnow, P. Internal ribosome entry sites in eukaryotic mRNA molecules. Genes Dev. 15, 1593–1612 (2001)
Kunkel, T. A. & Erie, D. A. DNA mismatch repair. Annu. Rev. Biochem. 74, 681–710 (2005)
Maroney, P. A., Chamnongpol, S., Souret, F. & Nilsen, T. W. Direct detection of small RNAs using splinted ligation. Nature Protocols 3, 279–287 (2008)
Trapnell, C., Pachter, L. & Salzberg, S. L. TopHat: discovering splice junctions with RNA-seq. Bioinformatics 25, 1105–1111 (2009)
Bailey, T. L. et al. MEME SUITE: tools for motif discovery and searching. Nucleic Acids Res. 37, W202–W208 (2009)
Acknowledgements
We would like to thank Qian laboratory members for helpful discussions and Cornell University Life Sciences Core Laboratory Center for performing deep sequencing. This work was supported by grants from the US National Institutes of Health DP2 OD006449 and R01AG042400 (to S.-B.Q.) and NIDA DA037150 (to S.R.J.) and the US Department of Defense (W81XWH-14-1-0068) (to S.-B.Q.).
Author information
Authors and Affiliations
Contributions
J.Z. and S.-B.Q. conceived the project. J.Z. performed most experiments. J.W. analysed the sequencing data. X.G. performed Ribo-seq. X.Z. assisted heat shock assays. S.R.J. helped with original FTO ideas. S.-B.Q. wrote the manuscript. All authors discussed the results and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
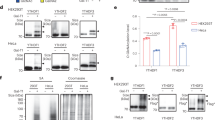
Extended Data Figure 1 Subcellular localization of the m6A machinery in cells before and after heat shock stress.
a, MEF cells before or 2 h after heat shock (HS; 42 °C, 1 h) were immunostanied with indicated antibodies. DAPI was used for nuclear staining. b, MEF (left panel) and HeLa cells (right panel) were subject to heat shock stress (42 °C, 1 h) followed by recovery at 37 °C for various times. Anti-YTHDF2 immunostaining was counterstained by DAPI. Images are representative of at least 50 cells. Bar, 10 µm.
Extended Data Figure 2 mRNA stability and induction in response to heat shock stress.
a, Effects of heat shock stress on mRNA stability. MEF cells without heat shock stress (No HS), immediately after heat shock stress (42 °C, 1 h) (Post HS 0 h), or 2 h recovery at 37 °C (Post HS 2 h) were subject to further incubation in the presence of 5 µg ml−1 ActD. At the indicated times, mRNA levels were determined by qPCR. Error bars, mean ± s.e.m.; n = 3 biological replicates. b, MEF cells were collected at indicated times after heat shock stress (42 °C, 1 h) followed by RNA extraction and real-time PCR. Relative levels of indicated transcripts are normalized to β-actin. Error bars, mean ± s.e.m.; n = 3 biological replicates. c, HSF1 wild-type (WT) and knockout (KO) cells were subject to heat shock stress (42 °C, 1 h) followed by recovery at 37 °C for various times. Real-time PCR was conducted to quantify transcripts encoding Hsp70 and YTHDF2. Relative levels of transcripts are normalized to β-actin. Error bars, mean ± s.e.m.; *P < 0.05, **P < 0.01, unpaired two-tailed t-test; n = 3 biological replicates.
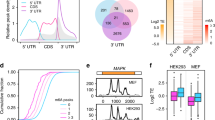
Extended Data Figure 3 Characterization of m6A sites in MEF cells with or without heat shock stress.
a, b, m6A profiling was conducted on MEF cells before (a) or 2 h after (b) heat shock (42 °C, 1 h). Left, pie chart presenting fractions of m6A peaks in different transcript segments. Right, sequence logo representing the consensus motif relative to m6A. CDS, coding sequence region.
Extended Data Figure 4 m6A profiling of HSPA8 in MEF cells with or without heat shock stress.
An example of constitutively expressed transcript HSPA8 in MEF cells with or without heat shock stress. Coverage of m6A immunoprecipitation and control reads (input) are indicated in red and grey, respectively. The transcript architecture is shown below the x axis.
Extended Data Figure 5 Dynamic m6A modification of HSPA1A by YTHDF2 and FTO.
An example of stress-induced transcript HSPA1A in post-stressed MEF cells with either YTHDF2 or FTO knockdown. Coverage of m6A immunoprecipitation and control reads (input) are indicated in red and blue, respectively. The transcript architecture is shown below the x axis.
Extended Data Figure 6 Direct competition between YTHDF2 and FTO in m6A binding.
a, Synthesized mRNA with m6A was incubated with FTO (2 µg) in the presence of an increasing amount of YTHDF2 (0, 0.5, 1 and 2 µg), followed by RNA pull-down and immunoblotting. b, Synthesized mRNA with m6A was incubated with FTO (1 µg in top panel and 2 µg in bottom panel) in the absence of presence of YTHDF2 (4 µg), followed by m6A dot blotting.
Extended Data Figure 7 YTHDF2 knockdown does not affect Hsp70 transcription after stress.
MEF cells with or without YTHDF2 knockdown were subject to heat shock stress (42 °C, 1 h) followed by recovery at 37 °C for various times. Real-time PCR was conducted to quantify Hsp70 mRNA levels. Error bars, mean ± s.e.m.; n = 3 biological replicates. sh-Scram, scrambled shRNA.
Extended Data Figure 8 FTO knockdown promotes Hsp70 synthesis.
a, m6A blotting of purified HSPA1A in MEF with or without FTO knockdown. Messenger RNAs synthesized by in vitro transcription in the absence or presence of m6A were used as control. RNA staining is shown as loading control. Representative of two biological replicates. b, MEF cells with or without FTO knockdown were collected at indicated times after heat shock stress (42 °C, 1 h) followed by immunoblotting using antibodies indicated. N, no heat shock. Representative of three biological replicates.
Extended Data Figure 9 m6A modification promotes cap-independent translation.
a, Fluc reporter mRNAs with or without 5′UTR was synthesized in the absence or presence of m6A. The transfected MEFs were incubation in the presence of 5 µg ml−1 ActD. At the indicated times, mRNA levels were determined by qPCR. Error bars, mean ± s.e.m.; n = 3 biological replicates. b, Fluc reporter mRNAs with or without Hsp70 5′UTR was synthesized in the absence of presence of m6A, followed by addition of a non-functional cap analogue ApppG. Fluc activity in transfected MEF cells was recorded using real-time luminometry. c, Constructs expressing Fluc reporter bearing 5′UTR from Hsc70 or Hsp105 are depicted on the top. Fluc activities in transfected MEF cells were quantified and normalized to the control containing normal A. Error bars, mean ± s.e.m.; *P < 0.05, unpaired two-tailed t-test; n = 3 biological replicates.
Extended Data Figure 10 Site-specific detection of m6A modification on HSPA1A.
a, Sequences of HSPA1A template and the DNA primer used for site-specific detection. Synthesized mRNAs containing a single m6A site (red) or A (blue) are used as positive and negative controls, respectively. The red shading in the HSPA1A sequence indicates predicted m6A sites. Autoradiogram shows primer extension of controls (left panel) and endogenous HSPA1A (right panel). b, Fluc mRNAs with or without m6A incorporation were incubated in the rabbit reticulocyte lysate system (RRL) at 30 °C for up to 60 min. Messenger RNA levels were determined by qPCR. Error bars, mean ± s.e.m.; n = 3 biological replicates.
Supplementary information
Supplementary Information
This file contains the original scan of the blots. (PDF 5312 kb)
Supplementary Data
This file contains Supplementary Table 1. (XLSX 1996 kb)
Supplementary Data
This file contains Supplementary Table 2. (XLSX 535 kb)
Rights and permissions
About this article
Cite this article
Zhou, J., Wan, J., Gao, X. et al. Dynamic m6A mRNA methylation directs translational control of heat shock response. Nature 526, 591–594 (2015). https://doi.org/10.1038/nature15377
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature15377
This article is cited by
-
The role of the methyltransferase METTL3 in prostate cancer: a potential therapeutic target
BMC Cancer (2024)
-
The relationship between the network of non-coding RNAs-molecular targets and N6-methyladenosine modification in tumors of urinary system
Cell Death & Disease (2024)
-
RNA modification-mediated mRNA translation regulation in liver cancer: mechanisms and clinical perspectives
Nature Reviews Gastroenterology & Hepatology (2024)
-
RNA m6A methylation and regulatory proteins in pulmonary arterial hypertension
Hypertension Research (2024)
-
Lactylation-driven FTO targets CDK2 to aggravate microvascular anomalies in diabetic retinopathy
EMBO Molecular Medicine (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.