Abstract
The Mesozoic era (252–66 million years ago), known as the domain of dinosaurs, witnessed a remarkable ecomorphological diversity of early mammals. The key mammalian characteristics originated during this period and were prerequisite for their evolutionary success after extinction of the non-avian dinosaurs 66 million years ago. Many ecomorphotypes familiar to modern mammal fauna evolved independently early in mammalian evolutionary history. Here we report a 125-million-year-old eutriconodontan mammal from Spain with extraordinary preservation of skin and pelage that extends the record of key mammalian integumentary features into the Mesozoic era. The new mammalian specimen exhibits such typical mammalian features as pelage, mane, pinna, and a variety of skin structures: keratinous dermal scutes, protospines composed of hair-like tubules, and compound follicles with primary and secondary hairs. The skin structures of this new Mesozoic mammal encompass the same combination of integumentary features as those evolved independently in other crown Mammalia, with similarly broad structural variations as in extant mammals. Soft tissues in the thorax and abdomen (alveolar lungs and liver) suggest the presence of a muscular diaphragm. The eutriconodont has molariform tooth replacement, ossified Meckel’s cartilage of the middle ear, and specialized xenarthrous articulations of posterior dorsal vertebrae, convergent with extant xenarthran mammals, which strengthened the vertebral column for locomotion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Alibardi, L. Perspectives on hair evolution based on some comparative studies on vertebrate cornification. J. Exp. Zool. 318B, 325–343 (2012)
Ji, Q. et al. The earliest known eutherian mammal. Nature 416, 816–822 (2002)
Ji, Q. et al. A swimming mammaliaform from the Middle Jurassic and ecomorphological diversification of early mammals. Science 311, 1123–1127 (2006)
Meng, Q.-J. et al. An arboreal docodont from the Jurassic and mammaliaform ecological diversification. Science 347, 764–768 (2015)
Meng, J. et al. A Mesozoic gliding mammal from northeastern China. Nature 444, 889–893 (2006)
Vullo, R. et al. Mammalian hairs in Early Cretaceous amber. Naturwissenschaften 97, 683–687 (2010)
Zhou, C.-F. et al. A Jurassic mammaliaform and the earliest mammalian evolutionary adaptations. Nature 500, 163–167 (2013)
Meng, J. & Wyss, A. R. Multituberculate and other mammal hair recovered from Palaeogene excreta. Nature 385, 712–714 (1997)
Chernova, O. F. Evolutionary aspects of hair polymorphism. Biol. Bull. 33, 43–52 (2006)
Storch, G. Eurotamandua joresi, ein Myrmecophagide aus dem Eozän der “Grube Messel” bei Darmstadt (Mammalia, Xenarthra). Senckenbergiana Lethaea 61, 247–289 (1981)
Ting, S.-Y. A Paleocene edentate from Nanxiong Basin, Guangdong. Palaeontol. Sin. New Series C 17, 85–118 (1987)
Rose, K. D. & Emry, R. J. in Mammal Phylogeny: Placentals (eds Szalay, F. S., Novacek, M. J. & McKenna, M. C. ) 81–102 (Springer, 1993)
Luo, Z.-X. & Wible, J. R. A Late Jurassic digging mammal and early mammalian diversification. Science 308, 103–107 (2005)
Linnaeus, C. Systema Naturæ per Regna Tria Naturæ, Secundum Classes, Ordines, Genera, Species, cum Characteribus, Differentiis, Synonymis, Locis Vol. 1 (Regnum Animale) editio decima, reformata (Laurentius Salvius, 1758)
Kermack, K. A. et al. The lower jaw of Morganucodon. Zool. J. Linn. Soc. 53, 87–175 (1973)
Chow, M. & Rich, T. H. V. A new triconodontan (Mammalia) from the Jurassic of China. J. Vertebr. Paleontol. 3, 226–231 (1984)
Fregenal-Martínez, M. A. & Meléndez, N. in Lake Basins through Space and Time (eds Gierlowski-Kordesch, E. H. & Kelts, K. ) AAPG Studies in Geology Vol. 46, 303–314 (2000)
Buscalioni, A. D. & Fregenal-Martínez, M. A. A holistic approach to the palaeoecology of Las Hoyas Konservat-Lagerstätte (La Huérgina Formation, Lower Cretaceous, Iberian Ranges, Spain). J. Iber. Geol. 36, 297–326 (2010)
Rougier, G. W. et al. An Early Cretaceous mammal from the Kuwajima Formation (Tetori Group), Japan, and a reassessment of triconodont phylogeny. Ann. Carnegie Mus. 76, 73–115 (2007)
Gao, C.-L. et al. A new mammal skull from the Lower Cretaceous of China with implications for the evolution of obtuse-angled molars and ‘amphilestid’ eutriconodonts. Proc. R. Soc. B 277, 237–246 (2010)
Ji, Q. et al. A Chinese triconodont mammal and mosaic evolution of the mammalian skeleton. Nature 398, 326–330 (1999)
Luo, Z.-X. et al. A new eutriconodont mammal and evolutionary development in early mammals. Nature 446, 288–293 (2007)
Meng, J. et al. Transitional mammalian middle ear from a new Cretaceous Jehol eutriconodont. Nature 472, 181–185 (2011)
Jenkins, F. A. & Schaff, C. R. The Early Cretaceous mammal Gobiconodon (Mammalia, Triconodonta) from the Cloverly Formation in Montana. J. Vertebr. Paleontol. 8, 1–24 (1988)
Kielan-Jaworowska, Z. & Dashzeveg, D. Early Cretaceous amphilestid (“triconodont”) mammal from Mongolia. Acta Palaeontol. Pol. 43, 413–438 (1998)
Li, C.-K. et al. A new species of Gobiconodon (Triconodonta, Mammalia) and its implication for the age of Jehol Biota. Chin. Sci. Bull. 48, 1129–1134 (2003)
Hu, Y.-M. Postcranial Morphology of Repenomamus (Eutriconodonta, Mammalia): Implications for the Higher-Level Phylogeny of Mammals. Dissertation, City Univ. of New York (2006)
Luo, Z.-X. Developmental patterns in Mesozoic evolution of mammal ears. Annu. Rev. Ecol. Syst. 42, 355–380 (2011)
Kielan-Jaworowska, Z. et al. Mammals from the Age of Dinosaurs. Origins, Evolution, and Structure (Columbia Univ. Press, 2004)
Gaudin, T. J. The morphology of xenarthrous vertebrae (Mammalia: Xenarthra). Fieldiana Geol. New Series 41, 1–38 (1999)
Lessertisseur, J. & Saban, R. in Traité de Zoologie Tome 16, Fascicule 1 (Mammifères, Téguments et Squelette) (ed. Grassé, P. P. ) 709–1078 (Masson et Cie, 1967)
Kusuhashi, N. et al. New triconodontids (Mammalia) from the Lower Cretaceous Shahai and Fuxin formations, northeastern China. Geobios 42, 765–781 (2009)
Gaetano, L. C. & Rougier, G. W. New materials of Argentoconodon fariasorum (Mammaliaformes, Triconodontidae) from the Jurassic of Argentina and its bearing on triconodont phylogeny. J. Vertebr. Paleontol. 31, 829–843 (2011)
Cuenca-Bescós, G. & Canudo, J. I. A new gobiconodontid mammal from the Early Cretaceous of Spain and its palaeogeographic implications. Acta Palaeontol. Pol. 48, 575–582 (2003)
Sweetman, S. C. A gobiconodontid (Mammalia, Eutriconodonta) from the Early Cretaceous (Barremian) Wessex Formation of the Isle of Wight, southern Britain. Palaeontology 49, 889–897 (2006)
Lovell, J. E. & Getty, R. The hair follicle, epidermis, dermis, and skin glands of the dog. Am. J. Vet. Res. 18, 873–885 (1957)
Whiteley, H. J. Giant compound hair follicles in the skin of the rabbit. Nature 181, 850 (1958)
Evans, H. E. & de Lahunta, A. Miller’s Anatomy of the Dog (Elsevier Saunders, 2012)
Spencer, B. & Sweet, G. The structure and development of the hairs of monotreme and marsupials. Q. J. Microsc. Sci. 36, 549–588 (1899)
Hausman, L. A. The cortical fusi of mammalian hair shafts. Am. Nat. 66, 461–470 (1932)
Rudnicka, L. et al. in Atlas of Trichoscopy: Dermoscopy in Hair and Scalp Disease (eds Rudnicka, L., Olszewska, M. & Rakowska, A. ) 11–46 (Springer, 2012)
Vincent, J. F. V. & Owers, P. Mechanical design of hedgehog spines and porcupine quills. J. Zool. 210, 55–75 (1986)
Montandon, S. A. et al. Two waves of anisotropic growth generate enlarged follicles in the spiny mouse. EvoDevo 5, 33 (2014)
Weibel, E. R. et al. Design of peripheral airways for efficient gas exchange. Respir. Physiol. Neurobiol. 148, 3–21 (2005)
Dal Sasso, C. & Signore, M. Exceptional soft tissue preservation in a theropod dinosaur from Italy. Nature 392, 383–387 (1998)
Bramble, D. M. & Jenkins, F. A. Mammalian locomotor-respiratory integration: implications for diaphragmatic and pulmonary design. Science 262, 235–240 (1993)
Campione, N. E. & Evans, D. C. A universal scaling relationship between body mass and proximal limb bone dimensions in quadrupedal terrestrial tetrapods. BMC Biol. 10, 60 (2012)
Foster, J. R. Preliminary body mass estimates for mammalian genera of the Morrison Formation (Upper Jurassic, North America). PaleoBios 28, 114–122 (2009)
Kirk, E. C. et al. Intrinsic hand proportions of eucharchontans and other mammals: Implications for the locomotor behavior of plesiadapiforms. J. Hum. Evol. 55, 278–299 (2008)
Stanley, W. T. et al. A new hero emerges: another exceptional mammalian spine and its potential adaptive significance. Biol. Lett. 9, 20130486 (2013)
Acknowledgements
Research funds were provided by Spanish MINECO, Project CGL-2013-42643 P and Junta de Comunidades de Castilla-La Mancha. We thank J. L. Sañudo for finding the specimen, M. Llandres and O. Dülfer for preparation, D. Kranz for artwork, O. Sanisidro for the lifelike reconstruction of S. xenarthrosus, G. Oleschinski for photography, M. Furió and A. Valera for SEM, K. Jäger for three-dimensional reconstructions, and T. McCann for improving the English. R. L. Cifelli is thanked for review and B. Mähler for discussion on actuo-taphonomical experiments on rat carcasses.
Author information
Authors and Affiliations
Contributions
A.D.B. designed the Las Hoyas research project; J.M.-L., R.V., H.M.-A., and A.D.B. participated in the fieldwork; T.M., J.M.-L., R.V., H.M.-A., Z.-X.L., and A.D.B. organized and conducted the research (preparation, computed tomography scan, light microscopy, and SEM imaging) and analysed data; Z.-X.L. performed phylogenetic analyses; and T.M., J.M.-L., R.V., Z.-X.L., and A.D.B. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
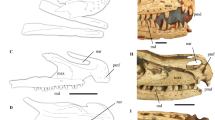
Extended Data Figure 1 New gobiconodontid S. xenarthrosus, holotype transferred slab MCCMLH30000A.
Skull exposed in ventral aspect. Inset: left calcaneus in dorsal view on the same slab somewhat apart from the skeleton. Arrow points to skin patch preserving hair bundles between dorsal vertebrae 11 and 14.
Extended Data Figure 2 Dentition and xenarthrous vertebrae of Spinolestes.
a, SEM images (stereo pair) of right lower mandible and right upper maxilla with molariforms. b, SEM image of anterior portion of skull and mandibles with dentition. c, SEM image of penultimate (m3) and ultimate (m4) right lower molariforms in lingual view. Abbreviations: C/c, upper/lower canine; I/i, incisor; M/m, molariform; P/p, premolar; D/d, deciduous tooth; R/r, replacing tooth. d, SEM images (stereo pair) of dorsal vertebrae D 12 to D 19 in lateral aspect with ribs.
Extended Data Figure 3 Phylogenetic relationships of Spinolestes and patterns of mammalian integumentary structure in early mammalian evolution.
a, Position of Spinolestes within Eutriconodonta (Bremer index/bootstrap values on the key clades of this study). b, Evolution of mammalian integumentary structures (simplified tree). Data sets and full analyses are presented in Supplementary Information.
Extended Data Figure 4 Skin structures of S. xenarthrosus.
a, SEM image of skin surface showing compound hair follicles (FO), epidermal cells (keratinocytes), and pores (P). b, Schematic drawings of scale-like skin folds (SCF) with compound hair follicles (FO) of the dog with primary (PH) and secondary (SH) hairs of three ontogenetic stages (redrawn from ref. 38). c, Skin surface of Spinolestes with scale-like wrinkles. d, Schematic diagram of scale-like wrinkled skin folds with hair follicles of a dog (redrawn from ref. 38). e, SEM image of epidermal cells (keratinocytes) of a hair follicle. f, SEM images of skin surface with polygonal epidermal cells (keratinocytes) and pores. g, Detail of the keratinocytes.
Extended Data Figure 5 SEM images and interpretative line drawings of cuticular scale patterns.
a–c, Primary (a, b) and secondary (b, c) hairs.
Extended Data Figure 6 Hair structures of S. xenarthrosus.
a, c, d, Patch of skin located between dorsal vertebrae 11 and 14 (arrow in Fig. 1 and Extended Data Fig. 1) on slab MCCMLH30000A under translucent light. b, SEM image of an orifice of a compound follicle (FO) with primary hair (PH) and three broken secondary hairs (SH). e, SEM image of a fraying hair of S. xenarthrosus showing fusiform cortical cells (FCC). f, Schematic diagram of human hair bulb (B) with cuticular scales (CU) and fusi (F) of the hair shaft (HS). g, Schematic diagram of a human head hair with fusiform cortical cells (FCC) and fusi. f, g, Redrawn from ref. 40. Abbreviations: C, cuticula; HB, hair bulb.
Extended Data Figure 7 External pinna of S. xenarthrosus.
a, b, Comparison with pinna and scalp of decaying rat (Rattus norvegicus): a, on transferred slab MCCMLH30000A; b, on counter slab MCCMLH30000B on original limestone rock matrix. c, d, Pinna and scalp of decaying rat (Rattus norvegicus) for comparison. c, Head of decaying rat after 274 days in water at room temperature. The skull is detached from the scalp and has fallen to the bottom. Scalp and right pinna are still intact and connected to the torso of the floating carcass. d, Detail of c with well-preserved right pinna in original position at the scalp. The mandible is displaced and loosely hanging down from the skin.
Extended Data Figure 8 Keratinous dermal scutes (SC) of S. xenarthrosus.
a, Oval dermal scutes dorsally of dorsal vertebra 20. b, Oval dermal scute dorsally of left ischium (ISC-L). c, Oval dermal scute from dorsal region with tubules (T) merging to homogeneous keratinous matrix.
Extended Data Figure 9 Visceral cavities of S. xenarthrosus with internal organs.
a, Detail of the visceral cavities of MCCMLH30000B with separation of the anterior thoracic cavity (TC) containing the lungs (LU), and the posterior abdominal cavity (AC) containing the liver (LI). Dashed line represents the diaphragm. SE, sternal elements. b, Lung tissue in the anterior part of the thoracic cavity. c, Peripheral conducting and acinar airways of the lungs with typical dichotomous structure of the bronchial tree. d, Peripheral conducting and acinar airways of the lungs from transferred slab MCCMLH30000A under translucent light. e, f, Details of the peripheral conducting airway dichotomies (DI) and acinar structures (ACI) of the lungs.
Supplementary information
Supplementary Information
This file contains Supplementary Text and Data, see contents page for details. (PDF 2988 kb)
CT reconstruction of right maxilla of Spinolestes xenarthrosus with heavily worn DM3 and DM4 still in place labially of replacing M3 and M4.
CT reconstruction of right maxilla of Spinolestes xenarthrosus with heavily worn DM3 and DM4 still in place labially of replacing M3 and M4. (MP4 12294 kb)
CT reconstruction of right mandible of Spinolestes xenarthrosus with wide Meckel´s groove (ossified Meckel´s cartilage fallen off).
CT reconstruction of right mandible of Spinolestes xenarthrosus with wide Meckel´s groove (ossified Meckel´s cartilage fallen off). (MP4 12361 kb)
PowerPoint slides
Rights and permissions
About this article
Cite this article
Martin, T., Marugán-Lobón, J., Vullo, R. et al. A Cretaceous eutriconodont and integument evolution in early mammals. Nature 526, 380–384 (2015). https://doi.org/10.1038/nature14905
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature14905
This article is cited by
-
Middle ear innovation in Early Cretaceous eutherian mammals
Nature Communications (2023)
-
On the way from Asia to America: eutriconodontan mammals from the Early Cretaceous of Yakutia, Russia
The Science of Nature (2023)
-
Petrosal morphology of the Early Cretaceous triconodontid Astroconodon from the Cloverly Formation (Montana, USA)
Journal of Mammalian Evolution (2023)
-
Cretaceous amniote integuments recorded through a taphonomic process unique to resins
Scientific Reports (2020)
-
Helminth eggs from early cretaceous faeces
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.