Abstract
Atmospheric methane is the second most important greenhouse gas after carbon dioxide, and is responsible for about 20% of the global warming effect since pre-industrial times1,2. Rice paddies are the largest anthropogenic methane source and produce 7–17% of atmospheric methane2,3. Warm waterlogged soil and exuded nutrients from rice roots provide ideal conditions for methanogenesis in paddies with annual methane emissions of 25–100-million tonnes3,4. This scenario will be exacerbated by an expansion in rice cultivation needed to meet the escalating demand for food in the coming decades4. There is an urgent need to establish sustainable technologies for increasing rice production while reducing methane fluxes from rice paddies. However, ongoing efforts for methane mitigation in rice paddies are mainly based on farming practices and measures that are difficult to implement5. Despite proposed strategies to increase rice productivity and reduce methane emissions4,6, no high-starch low-methane-emission rice has been developed. Here we show that the addition of a single transcription factor gene, barley SUSIBA2 (refs 7, 8), conferred a shift of carbon flux to SUSIBA2 rice, favouring the allocation of photosynthates to aboveground biomass over allocation to roots. The altered allocation resulted in an increased biomass and starch content in the seeds and stems, and suppressed methanogenesis, possibly through a reduction in root exudates. Three-year field trials in China demonstrated that the cultivation of SUSIBA2 rice was associated with a significant reduction in methane emissions and a decrease in rhizospheric methanogen levels. SUSIBA2 rice offers a sustainable means of providing increased starch content for food production while reducing greenhouse gas emissions from rice cultivation. Approaches to increase rice productivity and reduce methane emissions as seen in SUSIBA2 rice may be particularly beneficial in a future climate with rising temperatures resulting in increased methane emissions from paddies9,10.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kirschke, S. et al. Three decades of global methane sources and sinks. Nature Geosci. 6, 813–823 (2013)
Bridgham, S. D., Hinsby, C.-Q., Jason, K. K. & Zhuang, Q. Methane emissions from wetlands: biogeochemical, microbial, and modeling perspectives from local to global scales. Glob. Change Biol. 19, 1325–1346 (2013)
Liu, Y. & Whitman, W. B. Metabolic, phylogenetic, and ecological diversity of the methanogenic archaea. Ann. NY Acad. Sci. 1125, 171–189 (2008)
Sass, R. L. & Cicerone, R. J. Photosynthate allocations in rice plants: Food or atmospheric methane. Proc. Natl Acad. Sci. USA 99, 11993–11995 (2002)
Qiu, J. China cuts methane emissions from rice fields. Nature http://www.nature.com/news/2009/090818/full/news.2009.833.html (2009)
Denier van der Gon, H. A. et al. Optimizing grain yields reduces CH4 emission from rice paddy fields. Proc. Natl Acad. Sci. USA 99, 12021–12024 (2002)
Sun, C. et al. A novel WRKY transcription factor, SUSIBA2, participates in sugar signaling in barley by binding to the sugar-responsive elements of the iso1 promoter. Plant Cell 15, 2076–2092 (2003)
Sun, C., Höglund, A.-S., Olsson, H., Mangelsen, E. & Jansson, C. Antisense oligodeoxynucleotide inhibition as a potent strategy in plant biology: identification of SUSIBA2 as a transcriptional activator in plant sugar signaling. Plant J. 44, 128–138 (2005)
Yvon-Durocher, G. et al. Methane fluxes show consistent temperature dependence across microbial to ecosystem scales. Nature 507, 488–491 (2014)
Hoehler, T. M. & Alperin, M. J. Methane minimalism. Nature 507, 436–437 (2014)
Rushton, P. J., Somssich, I. E., Ringler, P. & Shen, Q. J. WRKY transcription factors. Trends Plant Sci. 15, 247–258 (2010)
Ashby, K. D., Casey, T. A., Rasmussen, M. A. & Petrich, J. W. Steady-state and time-resolved spectroscopy of F420 extracted from methanogen cells and its utility as a marker for fecal contamination. J. Agric. Food Chem. 49, 1123–1127 (2001)
Liu, P., Yang, Y., Lü, Z. & Lu, Y. Response of a rice paddy soil methanogen to syntrophic growth as revealed by transcriptional analyses. Appl. Environ. Microbiol. 80, 4668–4676 (2014)
Angel, R., Claus, P. & Conrad, R. Methanogenic archaea are globally ubiquitous in aerated soils and become active under wet anoxic conditions. ISME J. 6, 847–862 (2012)
Edwards, J. et al. Structure, variation, and assembly of the root-associated microbiomes of rice. Proc. Natl Acad. Sci. USA 112, E911–E920 (2015)
Conrad, R., Klose, M., Lu, Y. & Chidthaisong, A. Methanogenic pathway and archaeal communities in three different anoxic soils amended with rice straw and maize straw. Frontiers Microbiol. 3 http://dx.doi.org/10.3389/fmicb.2012.00004 (2012)
Peng, J., Lü, Z., Rui, J. & Lu, Y. Dynamics of the methanogenic archaeal community during plant residue decomposition in an anoxic rice field soil. Appl. Environ. Microbiol. 74, 2894–2901 (2008)
Zhang, M.-Z. et al. Molecular insights into how a deficiency of amylose affects carbon allocation-carbohydrate and oil analysis and gene expression profiling in the seeds of a rice waxy mutant. BMC Plant Biol. 12, 230 (2012)
Nalawade, S., Nalawade, S., Liu, C., Jansson, C. & Sun, C. Development of an efficient tissue culture after crossing (TCC) system for transgenic improvement of barley as a bioenergy crop. Appl. Energy 91, 405–411 (2012)
Sun, C., Sathish, P., Ahlandsberg, S., Deiber, A. & Jansson, C. The two genes encoding starch-branching enzymes IIa and IIb are differentially expressed in barley. Plant Physiol. 118, 37–49 (1998)
Hiei, Y., Ohta, S., Komari, T. & Kumashiro, T. Efficient transformation of rice (Oryza sativa L.) mediated by Agrobacterium and sequence analysis of the boundaries of the T-DNA. Plant J. 6, 271–282 (1994)
Jain, M., Nijhawan, A., Tyagi, A. K. & Khurana, J. P. Validation of housekeeping genes as internal control for studying gene expression in rice by quantitative real-time PCR. Biochem. Biophys. Res. Commun. 345, 646–651 (2006)
Caldana, C., Scheible, W. R., Mueller-Roeber, B. & Ruzicic, S. A quantitative RT–PCR platform for high-throughput expression profiling of 2500 rice transcription factors. Plant Methods 3, 7 (2007)
Yu, Y., Lee, C., Kim, J. & Hwang, S. Group-specific primer and probe sets to detect methanogenic communities using quantitative real-time polymerase chain reaction. Biotechnol. Bioeng. 89, 670–679 (2005)
Westerholm, M. et al. Quantification of syntrophic acetate-oxidizing microbial communities in biogas processes. Environ. Microbiol. Rep. 3, 500–505 (2011)
Narihiro, T. & Sekiguchi, Y. Oligonucleotide primers, probes and molecular methods for the environmental monitoring of methanogenic archaea. Microb. Biotechnol. 4, 585–602 (2011)
Harrison, R. M., Yamulki, S., Goulding, K. W. T. & Webster, C. P. Effect of fertilizer application on NO and N2O fluxes from agricultural fields. J. Geophys. Res. 100, 25923–25931 (1995)
Westerholm, M., Hansson, M. & Schnürer, A. Improved biogas production from whole stillage by co-digestion with cattle manure. Bioresour. Technol. 114, 314–319 (2012)
Yang, S., Peng, S., Xu, J., Luo, Y. & Li, D. Methane and nitrous oxide emissions from paddy field as affected by water-saving irrigation. Phys. Chem. Earth 53–54, 30–37 (2012)
Acknowledgements
Special thanks to S. Stymne. We would also like to thank B. Müller, X. Feng, M. Erikson, L. Sun, S. Isaksson, J. Ascue and S. Mayer for their help in determining concentrations of methane and methanogens, and B. Ingemarsson for discussions concerning the work layout. This work was funded by the following organisations and foundations: The Swedish Research Council for Environment, Agricultural Sciences and Spatial Planning (Formas) for Project No 219-2014-1172; the joint Formas/Sida-funded programme (Project No 220-2009-2069) on sustainable development in developing countries; the SLU Lärosätesansökan Programme (TC4F) for Team 4 supported by Vinnova; the Swedish Research Council for Environment, Agricultural Sciences and Spatial Planning (Formas) under the Strategic Research Area for the TCBB Programme; National Natural Science Foundation of China (projects no 30771298 and no 31370389); the SLU programme BarleyFunFood; the Carl Trygger Foundation for Project No CTS 11: 450; funding in part by the US Department of Energy Contract DE-AC05-76RL01830 with the Pacific Northwest National Laboratory.
Author information
Authors and Affiliations
Contributions
J.S., Z.C., Q.G., Y.W. and D.Z. performed measurements of methane emissions from paddies; J.S. also performed western blot and zymogram analyses, methanogen quantification and starch determination. C.H. was responsible for plasmid constructions, rice transformation, Southern blot analysis and phenotypic trait characterization. X.Y. carried out gene expression analysis, starch determination, sugar induction experiments and phenotypic trait characterization. Y.J. performed plasmid validation, insertion site identification, methanogen quantification, measurements of methane emissions in phytotrons and sugar induction experiments, electrophoretic mobility shift assay (EMSA), qPCR and light microscopy. C.J. was involved in the initiation, layout and discussions concerning the work and manuscript revision. F.W. was involved in the planning of rice transformation and field trial settings. A.S. revised the manuscript and helped with methane and methanogen determinations. C.S. initiated and coordinated the work, designed the experiments, performed some experiments, and drafted and revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Validation of an expression cassette containing barley SBEIIb promoter and barley SUSIBA2 (HvSBEIIb p:HvSUSIBA2) in a binary vector and rice genome.
a, Construction of an expression cassette containing HvSBEIIb p:HvSUSIBA2 in a binary vector was performed as described in the Methods. b, Validation of the construct in the rice genome was performed by PCR-based cloning using primers and HindIII-adaptor ligation, followed by sequencing. Two insertion sites were identified in rice chromosome 2, from nucleotides 57443 to 57444 and 57447 to 57448 (GenBank accession number AP006069), respectively.
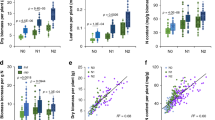
Extended Data Figure 2 Methane emissions of SUSIBA2 rice compared with Nipponbare (Nipp) in phytotrons.
a, Methane emission of Nipp, SUSIBA2-77 and SUSIBA2-80 rice (15 daf). Six independent plants (n = 6) from each rice line were used for measurements. b, Methane emission of Nipp and SUSIBA2-77 (28 daf) at 30 min (left panel) or 60 min (right panel) after plants were covered. A linear relationship over time for the measured methane concentrations at 30 and 60 min was found, as presented by similar methane fluxes determined from the different time points. Three plants (n = 3) from each rice line were used for time point measurements. A statistically significant reduction of methane emission in SUSIBA2 rice is indicated (one-way ANOVA, **P ≤ 0.01 or *P ≤ 0.05, error bars show s.d.).
Extended Data Figure 3 Diurnal and seasonal methane emissions from SUSIBA2-77 and SUSIBA-80 rice compared with Nipponbare (Nipp) in autumn 2014 at three sites in China.
Methane emissions of six independent plants (n = 6) from three time points during the day (morning 8.00 a.m., noon 12.00 p.m. and afternoon 4.00 p.m.) on different dates are presented. Key rice development stages for the corresponding dates are indicated. Time points for sampling soil and roots for methanogen analysis are indicated by red arrows. a, Methane emission from Fuzhou. b, Methane emission from Guangzhou. c, Methane emission from Nanning. Reported statistically significant reduction of methane emission in SUSIBA2 rice is indicated (one-way ANOVA, **P ≤ 0.01 or *P ≤ 0.05, error bars show s.d.).
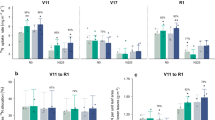
Extended Data Figure 4 qPCR quantification of rhizospheric methanogenic communities associated with SUSIBA2-77 rice and Nipp in phytotrons.
Soil and root samples from three independent positions, positions 1–3 (n = 3), in the underground vicinity close to the root tip and proximal regions of three independent plants (n = 3) for Nipp and SUSIBA2-77 rice were collected and analysed. Technical triplicates per position were applied. Six pairs of primers (Supplementary Table 1) were used to quantify total archaea (ARC) and methanogens (MET), and the orders Methanobacteriales (MBT), Methanomicrobiales (MMB) and Methanocellales and two families Methanosaetaceae (Mst) and Methanosarcinaceae (Msc) of the order Methanosarcinales, respectively. Results from soil and root samples are shown in a and b, respectively. Existing numbers of all methanogenic groups and total archaea were significantly reduced in the SUSIBA2 rhizosphere compared to that of Nipp (one-way ANOVA, **P ≤ 0.01 or *P ≤ 0.05, error bars show s.d.).
Extended Data Figure 5 qPCR quantification of rhizospheric methanogens associated with SUSIBA2 rice and Nipponbare (Nipp) from rice paddies.
Soil and root samples (a mixture) from three positions (n = 3) close to the root tip and proximal regions of three independent plants (n = 3) for Nipp, SUSIBA2-77 and SUSIBA2-80 rice, respectively, were collected. The sampling time and sites are indicated in Extended Data Fig. 3. Technical triplicates per position were applied. Six pairs of primers (Supplementary Table 1) were used to quantify total archaea (ARC) and methanogens (MET), and the orders Methanobacteriales (MBT), Methanomicrobiales (MMB) and Methanocellales and two families Methanosaetaceae (Mst) and Methanosarcinaceae (Msc) of the order Methanosarcinales, respectively. Primers (Supplementary Table 1) specific to Methanocella were also used for quantification. a, Methanogenic communities in samples from Fuzhou. b, Methanogenic communities in samples from Nanning. c, Methanocella in samples from Nanning. All methanogenic groups and total archaea were significantly reduced in the SUSIBA2 rice rhizosphere compared with Nipp (one-way ANOVA, **P ≤ 0.01 or *P ≤ 0.05, error bars show s.d.).
Extended Data Figure 6 Binding activity of HvSUSIBA2 to SURE sequences in the rice ISA1 promoter.
a, Three SURE sequences in the rice ISA1 promoter (GenBank accession number AB093426) were used and a negative (‘A stretch’) control was included. b, Barley SUSIBA2 protein (HvSUSIBA2) was overexpressed from E. coli and used for electrophoretic mobility shift assay (EMSA).
Extended Data Figure 7 HvSBEIIb promoter activity analysis in rice seedlings.
a, HvSBEIIb p:GUS was introduced in Nipponbare (Nipp). GUS activity was stained in different tissues of transformant and Nipp lines. The GUS activity was found in transformant stems and induced by sucrose (Suc) in leaves. a, Nipp leaf. b, Transformant leaf. c, Nipp stem. d, Transformant stem. e, Nipp root. f, Transformant root. g, Nipp leaf induced by 100 mM sucrose. h, Transformant leaf induced by 100 mM sucrose. Scale bars, 2 mm.
Extended Data Figure 8 Transcriptomic analysis of genes related to sugar metabolism in late tillering plants of SUSIBA2 rice and Nipponbare (Nipp).
Relative expression levels of 23 genes together with a housekeeping gene, TIP41-like, were analysed by qPCR and compared between Nipp and SUSIBA2-77 and SUSIBA2-80 rice in leaves, stems and roots at late tillering stage and in seeds at 7 daf. Three plants were used (n = 3) and technical triplicates were performed for each rice line. SUT3 and SUS5/7 transcripts were not detected in all samples (not shown) and HvSUSIBA2 expression was similar in SUSIBA2-77 and SUSIBA2-80 rice, as presented in Fig. 3b. One-way ANOVA was used for statistical analysis (*P ≤ 0.05 or **P ≤ 0.01, error bars show s.d.). ND, not detected.
Extended Data Figure 9 Transcriptomic analysis of 23 genes related to sugar metabolism in developing rice seeds of SUSIBA2 rice and Nipponbare (Nipp).
Relative expression levels of 23 genes together with a housekeeping gene, TIP41-like, were analysed by qPCR and compared between SUSIBA2-77 rice and Nipp at 5, 7, 9, 11 and 14 daf. Seeds from three independent plants were used (n = 3) and technical triplicates were performed. SUT3 and SUS5/7 transcripts were not detected in all samples (not shown) and HvSUSIBA2 expression is presented in Fig. 3b. One-way ANOVA was used for statistical analysis (*P ≤ 0.05 or **P ≤ 0.01, error bars show s.d.).
Extended Data Figure 10 Sugar-induction in Nipponbare (Nipp) and SUSIBA2 rice.
Rice leaves of Nipp and SUSIBA2-77 were depleted for sucrose in the dark before induction with 100 mM sucrose. Sugar induction was carried out in the dark for 24 h. An enhanced expression of HvSUSIBA2-regulated genes (9 of the 12 genes except SUT5, SUS1 and GBSSI that were not detected in either rice cultivar under dark conditions) in SUSIBA2-77 was observed compared with Nipp. One-way ANOVA was used for statistical analysis (*P ≤ 0.05 or **P ≤ 0.01, error bars show s.d.). The experiment was repeated at least three times (n = 3).
Supplementary information
Supplementary Information
This file contains Supplementary Table 1. (PDF 137 kb)
Supplementary Information
This file contains Supplementary Table 2. (XLSX 263 kb)
Supplementary Data
This file contains the sequences of the construct and insertion sites in Extended Data Fig. 1. (PDF 214 kb)
Source data
Rights and permissions
About this article
Cite this article
Su, J., Hu, C., Yan, X. et al. Expression of barley SUSIBA2 transcription factor yields high-starch low-methane rice. Nature 523, 602–606 (2015). https://doi.org/10.1038/nature14673
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature14673
This article is cited by
-
Genome-wide identification of the basic leucine zipper transcription factor genes related to starch synthesis in wheat (Triticum aestivum L.)
Plant Growth Regulation (2024)
-
Variations in the archaeal community and associated methanogenesis in peat profiles of three typical peatland types in China
Environmental Microbiome (2023)
-
Improved bioenergy value of residual rice straw by increased lipid levels from upregulation of fatty acid biosynthesis
Biotechnology for Biofuels and Bioproducts (2023)
-
Greenhouse gas emissions and mitigation in rice agriculture
Nature Reviews Earth & Environment (2023)
-
Loss-of-function gs3 allele decreases methane emissions and increases grain yield in rice
Nature Climate Change (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.