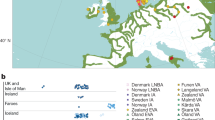
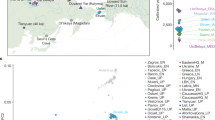
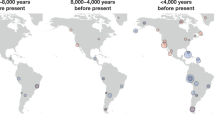
Abstract
The Bronze Age of Eurasia (around 3000–1000 BC) was a period of major cultural changes. However, there is debate about whether these changes resulted from the circulation of ideas or from human migrations, potentially also facilitating the spread of languages and certain phenotypic traits. We investigated this by using new, improved methods to sequence low-coverage genomes from 101 ancient humans from across Eurasia. We show that the Bronze Age was a highly dynamic period involving large-scale population migrations and replacements, responsible for shaping major parts of present-day demographic structure in both Europe and Asia. Our findings are consistent with the hypothesized spread of Indo-European languages during the Early Bronze Age. We also demonstrate that light skin pigmentation in Europeans was already present at high frequency in the Bronze Age, but not lactose tolerance, indicating a more recent onset of positive selection on lactose tolerance than previously thought.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
Primary accessions
European Nucleotide Archive
Data deposits
DNA sequence alignments are available from the European Nucleotide Archive (http://www.ebi.ac.uk/ena) under accession number PRJEB9021.
References
Fu, Q. et al. Genome sequence of a 45,000-year-old modern human from western Siberia. Nature 514, 445–449 (2014)
Seguin-Orlando, A. et al. Genomic structure in Europeans dating back at least 36,200 years. Science 346, 1113–1118 (2014)
Rasmussen, M. et al. An Aboriginal Australian genome reveals separate human dispersals into Asia. Science 334, 94–98 (2011)
Raghavan, M. et al. Upper Palaeolithic Siberian genome reveals dual ancestry of Native Americans. Nature 505, 87–91 (2014)
Raghavan, M. et al. The genetic prehistory of the New World Arctic. Science 345, 1255832 (2014)
Rasmussen, M. et al. The genome of a Late Pleistocene human from a Clovis burial site in western Montana. Nature 506, 225–229 (2014)
Bramanti, B. et al. Genetic discontinuity between local hunter-gatherers and Central Europe’s first farmers. Science 326, 137–140 (2009)
Malmström, H. et al. Ancient DNA reveals lack of continuity between Neolithic hunter-gatherers and contemporary Scandinavians. Curr. Biol. 19, 1758–1762 (2009)
Skoglund, P. et al. Origins and genetic legacy of Neolithic farmers and hunter-gatherers in Europe. Science 336, 466–469 (2012)
Lazaridis, I. et al. Ancient human genomes suggest three ancestral populations for present-day Europeans. Nature 513, 409–413 (2014)
Haak, W. et al. Ancient DNA from European early Neolithic farmers reveals their Near Eastern affinities. PLoS Biol. 8, e1000536 (2010)
Gamba, C. et al. Genome flux and stasis in a five millennium transect of European prehistory. Nature Commun. 5, 5257 (2014)
Kristiansen, K. in The World System and the Earth System. Global Socioenvironmental Change and Sustainability Since the Neolithic (eds Hornborg, B. & Crumley, C.) (Left Coast Press, 2007)
Shishlina, N. Reconstruction of the Bronze Age of the Caspian Steppes. Life Styles and Life Ways of Pastoral Nomads. Vol. 1876 (Archaeopress, 2008)
Anthony, D. The Horse, The Wheel and Language. How Bronze-Age Riders from the Eurasian Steppes Shaped the Modern World (Princeton Univ. Press, 2007)
Harrison, R. & Heyd, V. The Transformation of Europe in the third millennium BC: the example of ‘Le Petit-Chasseur I + III’ (Sion, Valais, Switzerland). Praehistorische Zeitschrift. 82, 129–214 (2007)
Vandkilde, H. Culture and Change in the Central European Prehistory, 6th to 1st millennium BC (Aarhus Univ. Press, 2007)
Kristiansen, K. & Larsson, T. The Rise of Bronze Age Society. Travels, Transmissions and Transformations (Cambridge Univ. Press, 2005)
Hanks, B. K., Epimakhov, A. V. & Renfrew, A. C. Towards a refined chronology for the Bronze Age of the southern Urals, Russia. Antiquity 81, 353–367 (2007)
Kuznetsov, P. F. The emergence of Bronze Age chariots in Eastern Europe. Antiquity 80, 638–645 (2006)
Koryakova, L. & Epimakhov, A. V. The Urals and Western Siberia in the Bronze and Iron Ages (Cambridge Univ. Press, 2007)
Rasmussen, M. et al. Ancient human genome sequence of an extinct Palaeo-Eskimo. Nature 463, 757–762 (2010)
Carpenter, M. L. et al. Pulling out the 1%: whole-genome capture for the targeted enrichment of ancient DNA sequencing libraries. Am. J. Hum. Genet. 93, 852–864 (2013)
Barros Damgaard, P. d. et al. Improving access to endogenous DNA in ancient bones and teeth. Preprint at bioRxivhttp://dx.doi.org/10.1101/014985 (2015)
Adler, C. J., Haak, W., Donlon, D., Cooper, A. & The Genographic Consortium Survival and recovery of DNA from ancient teeth and bones. J. Archaeol. Sci. 38, 956–964 (2011)
Orlando, L. et al. True single-molecule DNA sequencing of a Pleistocene horse bone. Genome Res. 21, 1705–1719 (2011)
Olalde, I. & Lalueza-Fox, C. Modern humans’ paleogenomics and the new evidences on the European prehistory. Science and Technology of Archaeological Research 1, http://dx.doi.org/10.1179/2054892315Y.0000000002 (2015)
Grigoriev, S. Ancient Indo-Europeans (Charoid, 2002)
Bendezu-Sarmiento, J. De l’Âge du Bronze et lÂge du Fer au Kazakkstan, gestes funéraires et paramètres biologiques. Identités culturelles des population Andronovo et Saka (De Boccard, 2007)
Kozintsev, A. G., Gromov, A. V. & Moiseyev, V. G. Collateral relatives of American Indians among the Bronze Age populations of Siberia? Am. J. Phys. Anthropol. 108, 193–204 (1999)
Kristiansen, K. in Becoming European. The transformation of third millennium Northern and Western Europe (eds Prescott, C. & Glørstad, H.) (Oxbow Books, 2012)
Haak, W. et al. Massive migration from the steppe was a source for Indo-European languages in Europe. Nature http://dx.doi.org/10.1038/nature14317 (this issue)
Olalde, I. et al. Derived immune and ancestral pigmentation alleles in a 7,000-year-old Mesolithic European. Nature 507, 225–228 (2014)
Itan, Y., Powell, A., Beaumont, M. A., Burger, J. & Thomas, M. G. The origins of lactase persistence in Europe. PLoS Computational Biol. 5, e1000491 (2009)
Mallory, J. In Search of the Indo-Europeans. Language, Archaeology and Myth (Thames & Hudson, 1987)
Renfrew, A. C. Archaeology and Language. The Puzzle of Indo-European Origins (Penguin, 1987)
Mallory, J. & Mair, V. The Tarim Mummies. Ancient China and the Mystery of the Earliest People from the West (Thames & Hudson, 2000)
Keyser, C. et al. Ancient DNA provides new insights into the history of south Siberian Kurgan people. Hum. Genet. 126, 395–410 (2009)
Meyer, M. & Kircher, M. Illumina sequencing library preparation for highly multiplexed target capture and sequencing. Cold Spring Harb. Protocols (2010)
Orlando, L. et al. Recalibrating Equus evolution using the genome sequence of an early Middle Pleistocene horse. Nature 499, 74–78 (2013)
Malaspinas, A.-S. et al. Two ancient human genomes reveal Polynesian ancestry among the indigenous Botocudos of Brazil. Curr. Biol. 24, R1035–R1037 (2014)
Willerslev, E. & Cooper, A. Ancient DNA. Proc. Royal Soc. B 272, 3–16 (2005)
Briggs, A. W. et al. Patterns of damage in genomic DNA sequences from a Neandertal. Proc. Natl Acad. Sci. USA 104, 14616–14621 (2007)
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25, 1754–1760 (2009)
Schubert, M. et al. Improving ancient DNA read mapping against modern reference genomes. BMC Genomics 13, 178 (2012)
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009)
Quinlan, A. R. & Hall, I. M. BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26, 841–842 (2010)
Jónsson, H., Ginolhac, A., Schubert, M., Johnson, P. & Orlando, L. mapDamage2.0: fast approximate Bayesian estimates of ancient DNA damage parameters. Bioinformatics (2013)
Fu, Q. et al. DNA analysis of an early modern human from Tianyuan Cave, China. Proc. Natl Acad. Sci. USA 110, 2223–2227 (2013)
Korneliussen, T. S., Albrechtsen, A. & Nielsen, R. ANGSD: analysis of next generation sequencing data. BMC Bioinformatics 15, (2014)
Patterson, N., Price, A. L. & Reich, D. Population structure and Eigenanalysis. PLoS Genet. 2, e190 (2006)
Alexander, D. H., Novembre, J. & Lange, K. Fast model-based estimation of ancestry in unrelated individuals. Genome Res. 19, 1655–1664 (2009)
Patterson, N. et al. Ancient admixture in human history. Genetics 192, 1065–1093 (2012)
Weir, B. S. & Hill, W. Estimating F-statistics. Annu. Rev. Genet. 36, 721–750 (2002)
Nyström, V. et al. Microsatellite genotyping reveals end-Pleistocene decline in mammoth autosomal genetic variation. Mol. Ecol. 21, 3391–3402 (2012)
Browning, S. R. & Browning, B. L. Rapid and accurate haplotype phasing and missing-data inference for whole-genome association studies by use of localized haplotype clustering. Am. J. Hum. Genet. 81, 1084–1097 (2007)
Acknowledgements
We thank K. Magnussen, L. A. Petersen, C. D. Mortensen and A. Seguin-Orlando at the Danish National Sequencing Centre for help with the sequencing. We thank C. G. Zacho for technical assistance. The project was funded by The European Research Council (FP/2007-2013, grant no. 269442, The Rise), The University of Copenhagen (KU2016 programme), Marie Curie Actions of the European Union (FP7/2007-2013, grant no. 300554), The Villum Foundation (Young Investigator Programme, grant no. 10120), Frederik Paulsen, The Miller Institute, University of California, Berkeley, The Lundbeck Foundation, and The Danish National Research Foundation.
Author information
Authors and Affiliations
Contributions
E.W. and K.K. initiated and led the study. M.E.A., J.S., L.V., H.S., P.B.D., A.M., M.R., L.S. performed the DNA laboratory work. M.Si., S.R., M.E.A., A.-S.M., P.B.D., A.M. analysed the genetic data. K.-G.S., T.A., N.L., L.H., J.B., P.D.C., P.D., P.R.D., A.E., A.V.E., K.F., M.F., G.G., T.G., A.G., S.G., T.H., R.J., J.K., V.K., A.K., V.K., A.K., I.L., C.L., A.M., G.M., I.M., M.M., R.M., V.M., D.Po., G.P., L.P., D.Pr., L.P., M.Sa., N.S., V.Sm., V.Sz., V.I.S., G.T., S.V.T., L.V., M.V., L.Y., V.Z. collected the samples and/or provided input to the archaeological interpretations. T.H. and D.C. conducted radiocarbon dating. T.S.-P., L.O., S.B., R.N. provided input to the genetic analyses. E.W., K.K., M.E.A., M.Si., K.-G.S. wrote the paper with input from all co-authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Principal component analysis of ancient genomes.
a, b, Principal component analysis of ancient individuals projected onto contemporary individuals from non-African populations (a), Europe, West Asia and the Caucasus (b). Grey labels represent population codes indicating coordinates for individuals (small) and median of the population (large). Coloured labels indicate positions for ancient individuals (small) and median for ancient groups (large). Ancient individuals within a group are connected to the respective median position by coloured lines.
Extended Data Figure 2 Pairwise outgroup f3 statistics.
Panels depict pairwise plots of outgroup f3 statistics of the form f3(Ju’hoan North;Population1, Population2), showing the correlation of the amount of shared genetic drift for a pair of ancient groups (Population1) with all modern populations (Population2) in the Human Origins data set (panel A). Closely related ancient groups are expected to show highly correlated statistics. a, Sintashta/Corded Ware. b, Yamnaya/Afanasievo. c, Sintashta/Andronovo. d, Okunevo/Mal’ta. Coloured circles indicate modern populations; error bars indicate ± 1 standard error from the block jackknife.
Extended Data Figure 3 Yamnaya ancestry mirrors Mal’ta ancestry in present-day Europeans and Caucasians.
Panels show pairwise plots of D-statistics D(Outgroup, Ancient)(Bedouin, Modern), contrasting Mal’ta (MA1) and Hunter-gatherers (a), and MA1 and Yamnaya (b). Coloured labels indicate modern populations, with lines corresponding to ± 1 standard error of the respective D-statistic from block jacknife. Text away from the diagonal line indicates an ancient group with relative increase in allele sharing with the respective modern populations.
Extended Data Figure 4 Genetic differentiation between ancient and modern groups in Human Origins data set.
Panels show FST between pairs of modern and ancient groups (coloured lines) for subsets of ancient groups, with results for the remaining groups in the background (grey). Top, early Europeans. Middle, Bronze Age Europeans and steppe/Caucasus. Bottom, Bronze Age Asians. Results based on Human Origins data set (panel A).
Extended Data Figure 5 Genetic differentiation between ancient and modern groups in 1000 Genomes data set.
Matrix of pairwise FST values between modern and ancient groups in the 1000 Genomes data set (panel B).
Extended Data Figure 6 Distribution of uniparental lineages in Bronze Age Eurasians.
a, b, Barplots showing the relative frequency of Y chromosome (a) and mitochondrial DNA lineages (b) in different Bronze Age groups. Top row shows overall frequencies for all individuals combined.
Extended Data Figure 7 Derived allele frequencies for lactase persistence in modern and ancient groups.
Derived allele frequency of rs4988235 in the LCT gene inferred from imputation of ancient individuals. Numbers indicate the total number of chromosomes for each group.
Supplementary information
Supplementary Information
This file contains Supplementary Information sections 1-6. Section 1: An introduction to the sampled cultures and their dating. Section 2: Brief description of the samples (including Supplementary Tables 1-3). Section 3: Laboratory work and sample selection (including Supplementary Tables 4-5, and Supplementary Figure 1). Section 4: Radiocarbon dating. Section 5: Bioinformatics and DNA authentication. Section 6: Population genomics (including Supplementary Table 9 and Supplementary Figures 2-6). (PDF 4331 kb)
Supplementary Table 6
This table contains sequencing summary statistics. (XLSX 20 kb)
Supplementary Table 7
This table contains an overview of aDNA damage statistics. (XLS 44 kb)
Supplementary Table 8
This table contains results of DNA contamination tests. (XLSX 18 kb)
Supplementary Table 10
This table contains D-test for all combinations D(Outgroup,Ancient1)(Ancient2)(Ancient3); 1000 Genomes dataset. (XLSX 1915 kb)
Supplementary Table 11
This table contains “Outgroup” f3-statistics for all combinations of ancient and modern groups; Human Origins dataset. (XLSX 748 kb)
Supplementary Table 12
This table contains all-pair “admixture” f3-statistics; 1000 Genomes dataset. (XLSX 3921 kb)
Supplementary Table 13
This table contains derived allele frequencies of 104 SNP catalogue for putative selection; 1000 Genomes dataset. (XLSX 63 kb)
Supplementary Table 14
This table contains an overview of mtDNA haplogroups and identified variants. (XLS 97 kb)
Rights and permissions
About this article
Cite this article
Allentoft, M., Sikora, M., Sjögren, KG. et al. Population genomics of Bronze Age Eurasia. Nature 522, 167–172 (2015). https://doi.org/10.1038/nature14507
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature14507
This article is cited by
-
The Allen Ancient DNA Resource (AADR) a curated compendium of ancient human genomes
Scientific Data (2024)
-
Population genomics of post-glacial western Eurasia
Nature (2024)
-
aMeta: an accurate and memory-efficient ancient metagenomic profiling workflow
Genome Biology (2023)
-
Genetic history of East-Central Europe in the first millennium CE
Genome Biology (2023)
-
correctKin: an optimized method to infer relatedness up to the 4th degree from low-coverage ancient human genomes
Genome Biology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.