Abstract
Hypothalamic pro-opiomelanocortin (POMC) neurons promote satiety. Cannabinoid receptor 1 (CB1R) is critical for the central regulation of food intake. Here we test whether CB1R-controlled feeding in sated mice is paralleled by decreased activity of POMC neurons. We show that chemical promotion of CB1R activity increases feeding, and notably, CB1R activation also promotes neuronal activity of POMC cells. This paradoxical increase in POMC activity was crucial for CB1R-induced feeding, because designer-receptors-exclusively-activated-by-designer-drugs (DREADD)-mediated inhibition of POMC neurons diminishes, whereas DREADD-mediated activation of POMC neurons enhances CB1R-driven feeding. The Pomc gene encodes both the anorexigenic peptide α-melanocyte-stimulating hormone, and the opioid peptide β-endorphin. CB1R activation selectively increases β-endorphin but not α-melanocyte-stimulating hormone release in the hypothalamus, and systemic or hypothalamic administration of the opioid receptor antagonist naloxone blocks acute CB1R-induced feeding. These processes involve mitochondrial adaptations that, when blocked, abolish CB1R-induced cellular responses and feeding. Together, these results uncover a previously unsuspected role of POMC neurons in the promotion of feeding by cannabinoids.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
04 March 2015
Minor changes were made to citations in the Methods.
References
Dietrich, M. O. & Horvath, T. L. Hypothalamic control of energy balance: insights into the role of synaptic plasticity. Trends Neurosci. 36, 65–73 (2013)
Aponte, Y., Atasoy, D. & Sternson, S. M. AGRP neurons are sufficient to orchestrate feeding behavior rapidly and without training. Nature Neurosci. 14, 351–355 (2011)
Krashes, M. J. et al. Rapid, reversible activation of AgRP neurons drives feeding behavior in mice. J. Clin. Invest. 121, 1424–1428 (2011)
Yang, Y., Atasoy, D., Su, H. H. & Sternson, S. M. Hunger states switch a flip-flop memory circuit via a synaptic AMPK-dependent positive feedback loop. Cell 146, 992–1003 (2011)
DiPatrizio, N. V. & Piomelli, D. The thrifty lipids: endocannabinoids and the neural control of energy conservation. Trends Neurosci. 35, 403–411 (2012)
Bermudez-Silva, F. J., Cardinal, P. & Cota, D. The role of the endocannabinoid system in the neuroendocrine regulation of energy balance. J. Psychopharmacol. 26, 114–124 (2012)
Sinnayah, P. et al. Feeding induced by cannabinoids is mediated independently of the melanocortin system. PLoS ONE 3, e2202 (2008)
Bakkali-Kassemi, L. et al. Effects of cannabinoids on neuropeptide Y and β-endorphin expression in the rat hypothalamic arcuate nucleus. Br. J. Nutr. 105, 654–660 (2011)
Ho, J., Cox, J. M. & Wagner, E. J. Cannabinoid-induced hyperphagia: correlation with inhibition of proopiomelanocortin neurons? Physiol. Behav. 92, 507–519 (2007)
Hentges, S. T., Low, M. J. & Williams, J. T. Differential regulation of synaptic inputs by constitutively released endocannabinoids and exogenous cannabinoids. J. Neurosci. 25, 9746–9751 (2005)
Bellocchio, L. et al. Bimodal control of stimulated food intake by the endocannabinoid system. Nature Neurosci. 13, 281–283 (2010)
Hentges, S. T. Synaptic regulation of proopiomelanocortin neurons can occur distal to the arcuate nucleus. J. Neurophysiol. 97, 3298–3304 (2007)
Zhan, C. et al. Acute and long-term suppression of feeding behavior by POMC neurons in the brainstem and hypothalamus, respectively. J. Neurosci. 33, 3624–3632 (2013)
Dores, R. M. & Baron, A. J. Evolution of POMC: origin, phylogeny, posttranslational processing, and the melanocortins. Ann. NY Acad. Sci. 1220, 34–48 (2011)
Dube, M. G., Horvath, T. L., Leranth, C., Kalra, P. S. & Kalra, S. P. Naloxone reduces the feeding evoked by intracerebroventricular galanin injection. Physiol. Behav. 56, 811–813 (1994)
Kalra, S. P. & Horvath, T. L. Neuroendocrine interactions between galanin, opioids, and neuropeptide Y in the control of reproduction and appetite. Ann. NY Acad. Sci. 863, 236–240 (1998)
Mountjoy, K. G., Mortrud, M. T., Low, M. J., Simerly, R. B. & Cone, R. D. Localization of the melanocortin-4 receptor (MC4-R) in neuroendocrine and autonomic control circuits in the brain. Mol. Endocrinol. 8, 1298–1308 (1994)
Balthasar, N. et al. Divergence of melanocortin pathways in the control of food intake and energy expenditure. Cell 123, 493–505 (2005)
Leibowitz, S. F. & Hor, L. Endorphinergic and alpha-noradrenergic systems in the paraventricular nucleus: effects on eating behavior. Peptides 3, 421–428 (1982)
Leibowitz, S. F. Brain neurotransmitters and appetite regulation. Psychopharmacol. Bull. 21, 412–418 (1985)
Morozov, Y. M. et al. Antibodies to cannabinoid type 1 receptor co-react with stomatin-like protein 2 in mouse brain mitochondria. Eur. J. Neurosci. 38, 2341–2348 (2013)
Morozov, Y. M., Horvath, T. L. & Rakic, P. A tale of two methods: Identifying neuronal CB1 receptors. Mol. Metabol. 3, 338 (2014)
Bénard, G. et al. Mitochondrial CB(1) receptors regulate neuronal energy metabolism. Nature Neurosci. 15, 558–564 (2012)
Hebert-Chatelain, E. et al. Studying mitochondrial CB1 receptors: Yes we can. Molecular Metabol. 3, 339 (2014)
Hebert-Chatelain, E. et al. Cannabinoid control of brain bioenergetics: Exploring the subcellular localization of the CB1 receptor. Mol. Metabol. 3, 495–504 (2014)
Schneeberger, M. et al. Mitofusin 2 in POMC neurons connects ER stress with leptin resistance and energy imbalance. Cell 155, 172–187 (2013)
Nasrallah, C. M. & Horvath, T. L. Mitochondrial dynamics in the central regulation of metabolism. Nature Rev. Endocrinol. 10, 650–658 (2014)
Diano, S. et al. Peroxisome proliferation-associated control of reactive oxygen species sets melanocortin tone and feeding in diet-induced obesity. Nature Med. 17, 1121–1127 (2011)
Negre-Salvayre, A. et al. A role for uncoupling protein-2 as a regulator of mitochondrial hydrogen peroxide generation. FASEB 11, 809–815 (1997)
Echtay, K. S. et al. Superoxide activates mitochondrial uncoupling proteins. Nature 415, 96–99 (2002)
Andrews, Z. B. et al. UCP2 mediates ghrelin’s action on NPY/AgRP neurons by lowering free radicals. Nature 454, 846–851 (2008)
Diano, S. & Horvath, T. L. Mitochondrial uncoupling protein 2 (UCP2) in glucose and lipid metabolism. Trends Mol. Med. 18, 52–58 (2012)
Huszar, D. et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 88, 131–141 (1997)
Fan, W., Boston, B. A., Kesterson, R. A., Hruby, V. J. & Cone, R. D. Role of melanocortinergic neurons in feeding and the agouti obesity syndrome. Nature 385, 165–168 (1997)
Cone, R. D. Anatomy and regulation of the central melanocortin system. Nature Neurosci. 8, 571–578 (2005)
Elias, C. F. et al. Leptin differentially regulates NPY and POMC neurons projecting to the lateral hypothalamic area. Neuron 23, 775–786 (1999)
Cowley, M. A. et al. Leptin activates anorexigenic POMC neurons through a neural network in the arcuate nucleus. Nature 411, 480–484 (2001)
Horvath, T. L., Naftolin, F., Kalra, S. P. & Leranth, C. Neuropeptide-Y innervation of beta-endorphin-containing cells in the rat mediobasal hypothalamus: a light and electron microscopic double immunostaining analysis. Endocrinology 131, 2461–2467 (1992)
Soria-Gómez, E. et al. The endocannabinoid system controls food intake via olfactory processes. Nature Neurosci. 17, 407–415 (2014)
Greenberg, I., Kuehnle, J., Mendelson, J. H. & Bernstein, J. G. Effects of marihuana use on body weight and caloric intake in humans. Psychopharmacology 49, 79–84 (1976)
Foltin, R. W., Brady, J. V. & Fischman, M. W. Behavioral analysis of marijuana effects on food intake in humans. Pharmacol. Biochem. Behav. 25, 577–582 (1986)
Coppola, A. et al. A central thermogenic-like mechanism in feeding regulation: an interplay between arcuate nucleus T3 and UCP2. Cell Metab. 5, 21–33 (2007)
Parton, L. E. et al. Glucose sensing by POMC neurons regulates glucose homeostasis and is impaired in obesity. Nature 449, 228–232 (2007)
Horvath, T. L. et al. Brain uncoupling protein 2: uncoupled neuronal mitochondria predict thermal synapses in homeostatic centers. J. Neurosci. 19, 10417–10427 (1999)
Andrews, Z. B. et al. Ghrelin promotes and protects nigrostriatal dopamine function via a UCP2-dependent mitochondrial mechanism. J. Neurosci. 29, 14057–14065 (2009)
Landry, M., Vila-Porcile, E., Hokfelt, T. & Calas, A. Differential routing of coexisting neuropeptides in vasopressin neurons. Eur. J. Neurosci. 17, 579–589 (2003)
van den Pol, A. N. Neuropeptide transmission in brain circuits. Neuron 76, 98–115 (2012)
Pinto, S. et al. Rapid rewiring of arcuate nucleus feeding circuits by leptin. Science 304, 110–115 (2004)
Zimmer, A., Zimmer, A. M., Hohmann, A. G., Herkenham, M. & Bonner, T. I. Increased mortality, hypoactivity, and hypoalgesia in cannabinoid CB1 receptor knockout mice. Proc. Natl Acad. Sci. USA 96, 5780–5785 (1999)
Zhang, C. Y. et al. Uncoupling protein-2 negatively regulates insulin secretion and is a major link between obesity, beta cell dysfunction, and type 2 diabetes. Cell 105, 745–755 (2001)
Dietrich, M. O., Liu, Z. W. & Horvath, T. L. Mitochondrial dynamics controlled by mitofusins regulate Agrp neuronal activity and diet-induced obesity. Cell 155, 188–199 (2013)
Dietrich, M. O. et al. AgRP neurons regulate development of dopamine neuronal plasticity and nonfood-associated behaviors. Nature Neurosci. 15, 1108–1110 (2012)
Cetin, A., Komai, S., Eliava, M., Seeburg, P. H. & Osten, P. Stereotaxic gene delivery in the rodent brain. Nature Protocols 1, 3166–3173 (2006)
Moreira, P. I. et al. Mitochondria from distinct tissues are differently affected by 17β-estradiol and tamoxifen. J. Steroid Biochem. Mol. Biol. 123, 8–16 (2011)
Acknowledgements
The authors thank M. Shanabrough and J. Bober for technical support and R. Jakab for assisting with the illustrations. This work was supported by the US National Institutes of Health (DP1 DK098058, R01 DK097566, R01 AG040236 and P01 NS062686), the American Diabetes Association, The Klarmann Family Foundation, the Helmholtz Society (ICEMED) and the Deutsche Forschungsgemeinschaft SFB 1052/1 (Obesity Mechanisms).
Author information
Authors and Affiliations
Contributions
M.K., S.D. and T.L.H. developed the conceptual framework of this study. M.K., M.O.D., X.-B.G., S.D. and T.L.H. interpreted results. M.K. performed experiments and analysed results. Experimental contributions: L.V. contributed to Figs 4h–j, 5d and Extended Data Figs 1b, 5e and 6a, b; J.G.K. contributed to Figs 2e, f, 3i, 5a, b and Extended Data Fig. 2g; J.D.K. contributed to Figs 3b–d, 5e–g and Extended Data Figs 5c and 6c; F.H. contributed to Figs 4a, 5c and Extended Data Fig. 5a, b, d; S.E.S. contributed to Fig. 3a; C.M.C., C.R.V. and J.K.E. provided key animal models; Y.M.M. and P.R. contributed to Fig. 3b and Extended Data Fig. 1c; P.R., I.B. and M.A.C. provided materials, animals and equipment; K.S.-B. contributed to Figs 3f and 4d–g; X.-B.G. contributed to Figs 1C, Da–c and 3j. M.K. and T.L.H. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
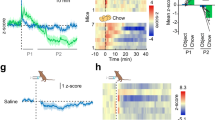
Extended Data Figure 1 Characterization of CB1R-dependent food intake.
a, Bimodal effects of different ACEA doses on food intake in fed mice (vehicle, n = 23 mice, 100 ± 16.3%; ACEA (in mg kg−1 body weight, intraperitoneal): 0.1, n = 8, 104.5 ± 46.6%; 0.5, n = 3, 190.8 ± 40.4%; 1.0, n = 19, 196.7 ± 30%; 2.5, n = 16, 87.1 ± 18%; 5.0, n = 11, 59.2 ± 15.5%; P < 0.01 versus vehicle, one-way ANOVA, followed by Dunnett’s multiple comparisons test; six independent experiments with litters from different parents). b, Neutral dose of ACEA on feeding (5 mg kg−1 body weight, intraperitoneal) did not alter locomotor activity of fed mice (n = 3 mice/group; P > 0.05). c, Impaired feeding response to ACEA (1 mg kg−1 body weight, intraperitoneal) in CB1R-heterozygote mice (Cnr1+/−, n = 6 mice, 1 h: 0.04 ± 0.01 g, 2 h: 0.07 ± 0.01 g) and CB1R-deficient mice (Cnr1−/−, 1 h: n = 6, 0.02 ± 0.01 g, 2 h: n = 4, 0.03 ± 0.01 g) mice, when compared to CB1R wild-type mice (Cnr1+/+, 1 h: n = 12, 0.13 ± 0.01 g, 2 h: n = 4, 0.18 ± 0.04 g; P < 0.01, P < 0.001 versus wild-type; two independent experiments). d, Central, local ACEA injection into the ARC induced food intake (vehicle, n = 4 mice, 1 h: 0.05 ± 0.03 g, 2 h: 0.12 ± 0.01 g; ACEA, n = 4, 1 h: 0.25 ± 0.03 g; 2 h: 0.43 ± 0.05 g; P < 0.01, P < 0.001). e, Verification of correct ARC cannula placement by HOECHST (blue) injection (representative image (two different magnifications) of four independent experiments). f, Hyperphagic CB1R activation (1 mg kg−1 body weight ACEA, intraperitoneal) was abolished by central, local ARC RIMO-mediated CB1R blockade (vehicle plus vehicle, n = 8 mice, 0.05 ± 0.01 g; vehicle plus ACEA, n = 8, 0.15 ± 0.02 g; RIMO plus vehicle, n = 8, 0.09 ± 0.02 g; RIMO plus ACEA, n = 8, 0.09 ± 0.02 g; P < 0.05, #P < 0.05 for interaction between RIMO and ACEA, two-way ANOVA, followed by Šidák’s multiple comparisons test; two independent experiments). g, Hyperphagic CB1R activation (1 mg kg−1 body weight WIN, intraperitoneal) was reduced by local ARC RIMO-mediated CB1R blockade (vehicle plus WIN, n = 8 mice, 0.21 ± 0.03 g; RIMO+WIN, n = 8, 0.1 ± 0.02 g; P < 0.01). h, RIMO-induced hypophagic blockade of CB1R in fasted mice (vehicle, n = 10 mice, 1 h: 0.76 ± 0.07 g, 2 h: 1.18 ± 0.07 g; RIMO, n = 11 mice, 1 h: 0.42 ± 0.05 g, 2 h: 0.75 ± 0.08 g; P < 0.01, P < 0.001; two independent experiments). Values (biological replicates) denote mean ± s.e.m. If not otherwise stated, P values (unpaired comparisons) by two-tailed Student’s t-test. Scale bars, 25 μm.
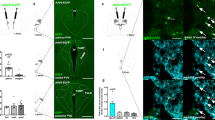
Extended Data Figure 2 DREADD-mediated regulation of POMC neurons.
a, Selective DREADD expression specified by local ARC mCherry fluorescence. b, POMC neurons (green) contain mCherry-labelled DREADD (red, arrowheads). c, CNO-activated inhibitory DREADD reduced ARC cFOS immunolabelled neurons in fed mice (arrowheads). Representative images of four independent experiments (a–c). d, e, CNO-activated inhibitory DREADD blocked ACEA-induced POMC activation (cFOS; vehicle plus ACEA, n = 6 mice, 60.4 ± 3.6%; CNO plus ACEA, n = 5, 32.3 ± 2.5%; P < 0.001). f, CNO-activated POMC-specific inhibitory DREADD did not acutely affect feeding but enhanced it after 8 h (vehicle, n = 17 mice, 0.42 ± 0.04 g; CNO, n = 16, 0.58 ± 0.04 g; 24 h after injection: vehicle, n = 5 mice, 2.57 ± 0.07 g; CNO, n = 5, 3.37 ± 0.18 g; P < 0.01 versus vehicle; three independent experiments). g, CNO-activated POMC-specific stimulating DREADD did not acutely affect feeding but reduced it after 8 h (vehicle, n = 6 mice, 0.58 ± 0.05 g; CNO, n = 6, 0.34 ± 0.05 g; P < 0.01 versus vehicle; 24 h after injection: vehicle, 3.96 ± 0.15 g; CNO, 3.65 ± 0.21 g; P > 0.05 versus vehicle). Values (biological replicates) denote mean ± s.e.m. If not otherwise stated, P values (unpaired comparisons) by two-tailed Student’s t-test. Scale bars, 100 μm (a), 25 μm (b) and 50 μm (c, d).
Extended Data Figure 3 Hyperphagic CB1R activation selectively increased PVN β-endorphin.
a–d, i, PVN α-MSH remained unchanged after hyperphagic CB1R activation (PVN unilateral analysis; vehicle, n = 6 values (technical replicates)/6 sections/3 mice (biological replicates); 60 min ACEA, n = 10/10/5; 90 min ACEA, n = 6/6/3; values, see Extended Data Table 1a). e–h, j, In contrast, hyperphagic ACEA increased PVN β-endorphin 60 and 90 min after application (PVN unilateral analysis; vehicle, n = 13 values/13 sections/6 mice; 60 min ACEA, n = 4/4/4; 90 min ACEA, n = 14/14/7; values, see Extended Data Table 1b. P < 0.001, P < 0.05 versus vehicle, one-way ANOVA, followed by Dunnett’s multiple comparisons test, two independent experiments using litters from different parents). Error bars indicate mean ± s.e.m. Scale bars, 25 μm.
Extended Data Figure 4 Bimodal character of ARC CB1R-driven β-endorphin increase.
a, Compared to vehicle (bilateral PVN analysis; n = 22 values (technical replicates)/11 sections/4 mice (biological replicates), hyperphagic doses (1 mg kg−1 body weight, respectively) of WIN (n = 24/12/4) or ACEA (n = 18/9/3) induced PVN β-endorphin immunoreactivity. Neutral dose (5 mg kg−1 BW) of ACEA (n = 18/9/3) on feeding showed no effects (see Extended Data Table 2 for all values). P < 0.05, P < 0.01, P < 0.001 versus vehicle, one-way ANOVA, followed by Dunnett's multiple comparisons test. b, Representative binary images of four independent experiments showing β-endorphin immunoreactivity after thresholding (image segmentation) using ImageJ software (see Methods). c, Compared to vehicle (unilateral PVN analysis; n = 4 mice (biological replicates), 2–3 sections (technical replicates) per mouse), central, hyperphagic local ARC injection of ACEA (n = 5 mice, 3 sections per mouse) increased PVN β-endorphin immunoreactivity (see Extended Data Table 3 for all values; P < 0.05, P < 0.01). Error bars indicate mean ± s.e.m. If not otherwise stated, P values (unpaired comparisons) by two-tailed Student’s t-test. Scale bars, 100 μm.
Extended Data Figure 5 Post-transcriptional regulation of hypothalamic pro-protein convertases, normal Cnr1 expression in Ucp2−/− mice and presence of CB1R in POMC neurons.
a, b, ACEA did not affect transcripts of pro-protein convertases 1 (Pcsk1) and 2 (Pcsk2) (in fold change; Pcsk1: vehicle, n = 11 mice, 1.00 ± 0.07; ACEA, n = 10 mice, 1.17 ± 0.09; Pcsk2: vehicle, n = 11 mice, 1.00 ± 0.13; ACEA, n = 11 mice, 1.14 ± 0.19; P > 0.05; two independent experiments). c, Representative western blot membranes for PC-1 (∼80 kilodaltons (kDa)) and PC-2 (∼72 kDa) immunolabelling. d, Equal Cnr1 expression in wild-type and Ucp2−/− mice (in fold change: all groups n = 6 mice; wild type, 1.00 ± 0.1; Ucp2−/−, 0.98 ± 0.12; P > 0.05). e, We have previously shown that antibodies raised against CB1R also recognized the mitochondrial protein, stomatin-like protein 2 (ref. 21). In line with this, mitochondrial labelling of CB1R was found substantially diminished but not completely eliminated in CB1R-KO (Cnr1−/−) mice23,24,25. We observed that in contrast to wild-type animals (Cnr1+/+ mice), which showed ∼80% (77 out of 97, 79.5 ± 3.9%) of POMC neurons (red fluorescence) to contain labelling with the CB1R antisera (green fluorescence), in CB1R knockout (KO; Cnr1−/−) mice, less than 30% (37 out of 128, 29.2 ± 3.3%) of POMC neurons retained immunolabelling. Thus, we concluded that a large population of POMC neurons contains CB1R (P < 0.001). All values (biological replicates: a–c, d; biological replicates including technical replicates: e) denote mean ± s.e.m. If not otherwise stated, P values (unpaired comparisons) by two-tailed Student’s t-test. Scale bar, 25 μm.
Extended Data Figure 6 Bimodal CB1R-dependent regulation of mitochondrial respiration and UCP2-dependent control of POMC.
a, b, Bimodal CB1R-controlled mitochondrial respiration in hippocampus. a, Hyperphagic (1 mg kg−1 body weight ACEA, intraperitoneal) CB1R activation increased ex vivo mitochondrial respiration (in nmol O2 min−1 mg−1 protein; state 3: vehicle, n = 6 mice, 170.7 ± 12; ACEA, n = 8, 252.7 ± 17.2; state 4: vehicle, 92.7 ± 5.4; ACEA, 139.7 ± 6; P < 0.01, P < 0.001). b, Neutral dose of ACEA on feeding (5 mg kg−1body weight, intraperitoneal) reduced mitochondrial respiration (state 3: vehicle, n = 7 mice, 178.2 ± 12.2; ACEA, n = 5, 118.9 ± 9.4; state 4: vehicle, 100 ± 5.1; ACEA, 64.3 ± 6.3; two independent experiments). c, Representative western blot membranes for POMC (pre-POMC, ∼31 kDa; POMC, ∼27 kDa). d, The 24-h food intake did not differ between wild-type (n = 28 mice, 100 ± 3.2%) and Ucp2−/− (n = 29, 98.9 ± 4.7%; P > 0.05) mice after ACEA (1 mg kg−1 body weight, intraperitoneal) treatment (six independent experiments using litters from different parents). All values (biological replicates) denote ± s.e.m. If not otherwise stated, P values (unpaired comparisons) by two-tailed Student’s t-test.
Supplementary information
Supplementary Table
This file contains Supplementary Table 1. (PDF 103 kb)
Source data
Rights and permissions
About this article
Cite this article
Koch, M., Varela, L., Kim, J. et al. Hypothalamic POMC neurons promote cannabinoid-induced feeding. Nature 519, 45–50 (2015). https://doi.org/10.1038/nature14260
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature14260
This article is cited by
-
The endocannabinoid N-arachidonoyl dopamine is critical for hyperalgesia induced by chronic sleep disruption
Nature Communications (2023)
-
Cannabis Sativa targets mediobasal hypothalamic neurons to stimulate appetite
Scientific Reports (2023)
-
The melanocortin action is biased toward protection from weight loss in mice
Nature Communications (2023)
-
Increased intrinsic and synaptic excitability of hypothalamic POMC neurons underlies chronic stress-induced behavioral deficits
Molecular Psychiatry (2023)
-
SK3 in POMC neurons plays a sexually dimorphic role in energy and glucose homeostasis
Cell & Bioscience (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.