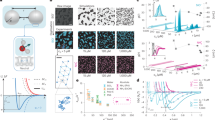
Abstract
Hydrophobic particles in water and hydrophilic particles in oil aggregate, but can form colloidal dispersions if their surfaces are chemically camouflaged with surfactants, organic tethers, adsorbed polymers or other particles that impart affinity for the solvent and increase interparticle repulsion1,2. A different strategy for modulating the interaction between a solid and a liquid uses surface corrugation, which gives rise to unique wetting behaviour3,4,5. Here we show that this topographical effect can also be used to disperse particles in a wide range of solvents without recourse to chemicals to camouflage the particles’ surfaces: we produce micrometre-sized particles that are coated with stiff, nanoscale spikes and exhibit long-term colloidal stability in both hydrophilic and hydrophobic media. We find that these ‘hedgehog’ particles do not interpenetrate each other with their spikes, which markedly decreases the contact area between the particles and, therefore, the attractive forces between them. The trapping of air in aqueous dispersions, solvent autoionization at highly developed interfaces, and long-range electrostatic repulsion in organic media also contribute to the colloidal stability of our particles. The unusual dispersion behaviour of our hedgehog particles, overturning the notion that like dissolves like, might help to mitigate adverse environmental effects of the use of surfactants and volatile organic solvents, and deepens our understanding of interparticle interactions and nanoscale colloidal chemistry.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Israelachvili, J. N. Intermolecular and Surface Forces 253–413 (Academic, 2011)
Sperling, R. A. & Parak, W. J. Surface modification, functionalization and bioconjugation of colloidal inorganic nanoparticles. Phil. Trans. R. Soc. A 368, 1333–1383 (2010)
Feng, L. et al. Super-hydrophobic surfaces: from natural to artificial. Adv. Mater. 14, 1857–1860 (2002)
Lafuma, A. & Quéré, D. Superhydrophobic states. Nature Mater. 2, 457–460 (2003)
Parker, A. R. & Lawrence, C. R. Water capture by a desert beetle. Nature 414, 33–34 (2001)
Yang, P. D. et al. Controlled growth of ZnO nanowires and their optical properties. Adv. Funct. Mater. 12, 323–331 (2002)
Kim, S. H., Abbaspourrad, A. & Weitz, D. A. Amphiphilic crescent-moon-shaped microparticles formed by selective adsorption of colloids. J. Am. Chem. Soc. 133, 5516–5524 (2011)
Walther, A. & Muller, A. H. E. Janus particles: synthesis, self-assembly, physical properties, and applications. Chem. Rev. 113, 5194–5261 (2013)
Yan, J., Bloom, M., Bae, S. C., Luijten, E. & Granick, S. Linking synchronization to self-assembly using magnetic Janus colloids. Nature 491, 578–581 (2012)
Papadopoulos, P., Mammen, L., Deng, X., Vollmer, D. & Butt, H. J. How superhydrophobicity breaks down. Proc. Natl Acad. Sci. USA 110, 3254–3258 (2013)
Verho, T. et al. Mechanically durable superhydrophobic surfaces. Adv. Mater. 23, 673–678 (2011)
Wang, S. & Jiang, L. Definition of superhydrophobic states. Adv. Mater. 19, 3423–3424 (2007)
Quéré, D. Wetting and roughness. Annu. Rev. Mater. Res. 38, 71–99 (2008)
Whyman, G. & Bormashenko, E. How to make the Cassie wetting state stable? Langmuir 27, 8171–8176 (2011)
Tyrrell, J. W. G. & Attard, P. Images of nanobubbles on hydrophobic surfaces and their interactions. Phys. Rev. Lett. 87, 176104 (2001)
Mao, M., Zhang, J. H., Yoon, R. H. & Ducker, W. A. Is there a thin film of air at the interface between water and smooth hydrophobic solids? Langmuir 20, 1843–1849 (2004)
Martines, E. et al. Air-trapping on biocompatible nanopatterns. Langmuir 22, 11230–11233 (2006)
Poetes, R., Holtzmann, K., Franze, K. & Steiner, U. Metastable underwater superhydrophobicity. Phys. Rev. Lett. 105, 166104 (2010)
Tang, Z. Y., Kotov, N. A. & Giersig, M. Spontaneous organization of single CdTe nanoparticles into luminescent nanowires. Science 297, 237–240 (2002)
Tang, Z. Y., Zhang, Z. L., Wang, Y., Glotzer, S. C. & Kotov, N. A. Self-assembly of CdTe nanocrystals into free-floating sheets. Science 314, 274–278 (2006)
Dong, A. G., Chen, J., Vora, P. M., Kikkawa, J. M. & Murray, C. B. Binary nanocrystal superlattice membranes self-assembled at the liquid–air interface. Nature 466, 474–477 (2010)
Takahashi, M. Zeta potential of microbubbles in aqueous solutions: electrical properties of the gas-water interface. J. Phys. Chem. B 109, 21858–21864 (2005)
Graciaa, A., Morel, G., Saulner, P., Lachaise, J. & Schechter, R. S. The zeta-potential of gas-bubbles. J. Colloid Interface Sci. 172, 131–136 (1995)
Beattie, J. K., Djerdjev, A. N. & Warr, G. G. The surface of neat water is basic. Faraday Discuss. 141, 31–39 (2009)
Beattie, J. K. & Djerdjev, A. M. The pristine oil/water interface: surfactant-free hydroxide-charged emulsions. Angew. Chem. Int. Ed. 43, 3568–3571 (2004)
Huang, R. X., Carney, R. P., Stellacci, F. & Lau, B. L. T. Colloidal stability of self-assembled mono layer-coated gold nanoparticles: the effects of surface compositional and structural heterogeneity. Langmuir 29, 11560–11566 (2013)
Saltzman, W. M. & Torchilin, V. Drug Delivery Systems (McGraw-Hill, 2008)
Zhou, H. et al. Self-assembly mechanism of spiky magnetoplasmonic supraparticles. Adv. Funct. Mater. 24, 1439–1448 (2014)
Hollingsworth, A., Leunissen, M., Irvine, W., Chaikin, P. & van Blaaderen, A. Charged colloids in low polar solvents. Bull. Am. Phys. Soc. 53, abstr. L9.00012. (2008)
Acknowledgements
This material is based on work partially supported by the Center for Solar and Thermal Energy Conversion, an Energy Frontier Research Center funded by the US Department of Energy, Office of Science, Office of Basic Energy Sciences under award number DE-SC0000957. We acknowledge support from the US NSF under grant ECS-0601345, CBET 0933384, CBET 0932823 and CBET 1036672. The work is also partly supported by the US Department of Defense under grant awards nos W911NF-10-1-0518, MURI W911NF-12-1-0407 and MURI W911NF-12-1-0407. We want express our appreciation to N. Walter for his leadership in organizing the SMART Center at the University of Michigan; J. Young Kim, Q. Che and X. Lu for help with the synthesis of CdTe nanoparticles; D. Bukharina for help with synthesis of hedgehog particles and SEM imaging of samples for dispersions in hydrophobic solvents; and D. Sorenson for assistance with TEM imaging of nanoparticle assemblies on OTMS-HPs. We also want to express our appreciation of H. Yu and G. Khan, who calculated van der Waals interactions of ZnO nanorods using the coupled dipole method. The guidance of C. Silvera Batista in this process is also much appreciated.
Author information
Authors and Affiliations
Contributions
N.K. had the idea for the project. N.K. and J.H.B. designed the experiments. J.H.B. designed and made the hedgehog particles, did the experiments and did the calculations of the pairwise interaction potential between the hedgehog particles. J.H.B. and B.Y. did the initial design of the hedgehog particles. Y.W. did biological experiments and helped to make the hedgehog particles. S.O.T. made the Fourier transform infrared spectroscopy measurements and helped to make the hedgehog particles. J.D.H. did confocal microscopy using an Olympus IX81 inverted with an ISS Alba 5 to detect concentric mesoscale shells on hedgehog particles indicating differences in refractive indices.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Text and Data, Supplementary Figures 1-28, Supplementary Tables 1-2 and Supplementary References – see Contents for details. (PDF 4895 kb)
A film of hydrophobic Hedgehog Particles with “Lotus Effect”
Filtrate from aqueous dispersion of hydrophobic OTMS-modified Hedgehog Particles (OTMS-HPs) exhibits “lotus effect” with a contact angle above 160o. This confirms the hydrophobic modification of the HPs which in turn displays superhydrophobicity. (MP4 2115 kb)
Procedure to create superhydrophobic film
This video contains a typical procedure for creating a filtrate on a polycarbonate membrane from aqueous suspension of OTMS- HPs that exhibits “lotus effect” as in Supplementary Video 1. (MP4 15942 kb)
VOC-less rendering of surfaces superhydrophobic
Filtrate from aqueous dispersion of surfactant-less hydrophobic OTMS- HPs on Kevlar fabric demonstrates facile and VOC-less rendering of surfaces superhydrophobic. (MP4 3188 kb)
Hydrophilic HPs with rapid droplet spreading
Filtrate from aqueous dispersion of hydrophilic HPs without OTMS treatment shows hydrophilic behavior of the film with a contact angle of 1-2o. (MP4 4251 kb)
Existence of air-pockets in dispersions of hydrophobic HPs in water
Addition of ethanol into aqueous dispersion of OTMS-HPs releases entrapped air due to lowering of the surface tension due to reduction in the surface tension of the meniscus. (MP4 4201 kb)
No air entrapment for dispersions of hydrophilic HPs in non-polar media
Addition of ethanol into dispersion of hydrophilic HPs in organic solvents does not produce air bubbles. This demonstrates differences in the dispersion mechanism of hydrophilic HPs in non-polar media compared to OTMS-HPs in aqueous media. (MP4 5687 kb)
Rights and permissions
About this article
Cite this article
Bahng, J., Yeom, B., Wang, Y. et al. Anomalous dispersions of ‘hedgehog’ particles. Nature 517, 596–599 (2015). https://doi.org/10.1038/nature14092
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature14092
This article is cited by
-
Photocatalytic cyclohexane oxidation and epoxidation using hedgehog particles
Nature Communications (2023)
-
Heterostructure particles enable omnidispersible in water and oil towards organic dye recycle
Nature Communications (2023)
-
Hot spots engineering by dielectric support for enhanced photocatalytic redox reactions
Nano Research (2023)
-
Construction of Au/Cu hierarchically organized particles with dual-functional enzyme-like activity
Science China Materials (2023)
-
Frustrated self-assembly of non-Euclidean crystals of nanoparticles
Nature Communications (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.