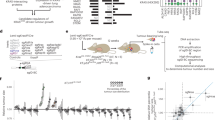
Abstract
Cancer is a multistep process that involves mutations and other alterations in oncogenes and tumour suppressor genes1. Genome sequencing studies have identified a large collection of genetic alterations that occur in human cancers2,3,4. However, the determination of which mutations are causally related to tumorigenesis remains a major challenge. Here we describe a novel CRISPR/Cas9-based approach for rapid functional investigation of candidate genes in well-established autochthonous mouse models of cancer. Using a KrasG12D-driven lung cancer model5, we performed functional characterization of a panel of tumour suppressor genes with known loss-of-function alterations in human lung cancer. Cre-dependent somatic activation of oncogenic KrasG12D combined with CRISPR/Cas9-mediated genome editing of tumour suppressor genes resulted in lung adenocarcinomas with distinct histopathological and molecular features. This rapid somatic genome engineering approach enables functional characterization of putative cancer genes in the lung and other tissues using autochthonous mouse models. We anticipate that this approach can be used to systematically dissect the complex catalogue of mutations identified in cancer genome sequencing studies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
References
Hanahan, D. & Weinberg, R. A. The hallmarks of cancer. Cell 100, 57–70 (2000)
Imielinski, M. et al. Mapping the hallmarks of lung adenocarcinoma with massively parallel sequencing. Cell 150, 1107–1120 (2012)
Govindan, R. et al. Genomic landscape of non-small cell lung cancer in smokers and never-smokers. Cell 150, 1121–1134 (2012)
The Cancer Genome Atlas Research Network Comprehensive molecular profiling of lung adenocarcinoma. Nature 511, 543–550 (2014)
Jackson, E. L. et al. Analysis of lung tumor initiation and progression using conditional expression of oncogenic K-ras. Genes Dev. 15, 3243–3248 (2001)
McFadden, D. G. et al. Genetic and clonal dissection of murine small cell lung carcinoma progression by genome sequencing. Cell 156, 1298–1311 (2014)
Frese, K. K. & Tuveson, D. A. Maximizing mouse cancer models. Nature Rev. Cancer 7, 645–658 (2007)
Xue, W. et al. CRISPR-mediated direct mutation of cancer genes in the mouse liver. Nature 514, 380–384 (2014)
Farago, A. F., Snyder, E. L. & Jacks, T. SnapShot: Lung cancer models. Cell 149, 246–246.e1 (2012)
Winslow, M. M. et al. Suppression of lung adenocarcinoma progression by Nkx2-1. Nature 473, 101–104 (2011)
DuPage, M. et al. Endogenous T cell responses to antigens expressed in lung adenocarcinomas delay malignant tumor progression. Cancer Cell 19, 72–85 (2011)
Madisen, L. et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nature Neurosci. 13, 133–140 (2010)
Rock, J. R. & Hogan, B. L. Epithelial progenitor cells in lung development, maintenance, repair, and disease. Annu. Rev. Cell Dev. Biol. 27, 493–512 (2011)
Song, M. S., Salmena, L. & Pandolfi, P. P. The functions and regulation of the PTEN tumour suppressor. Nature Rev. Mol. Cell Biol. 13, 283–296 (2012)
Curry, N. L. et al. Pten-null tumors cohabiting the same lung display differential AKT activation and sensitivity to dietary restriction. Cancer Discov 3, 908–921 (2013)
Snyder, E. L. et al. Nkx2-1 represses a latent gastric differentiation program in lung adenocarcinoma. Mol. Cell 50, 185–199 (2013)
Schwank, G. et al. Functional repair of CFTR by CRISPR/Cas9 in intestinal stem cell organoids of cystic fibrosis patients. Cell Stem Cell 13, 653–658 (2013)
Cheung, A. F. et al. Complete deletion of Apc results in severe polyposis in mice. Oncogene 29, 1857–1864 (2010)
Moon, R. T., Kohn, A. D., De Ferrari, G. V. & Kaykas, A. WNT and β-catenin signalling: diseases and therapies. Nature Rev. Genet. 5, 691–701 (2004)
Pacheco-Pinedo, E. C. et al. Wnt/β-catenin signaling accelerates mouse lung tumorigenesis by imposing an embryonic distal progenitor phenotype on lung epithelium. J. Clin. Invest. 121, 1935–1945 (2011)
Kormish, J. D., Sinner, D. & Zorn, A. M. Interactions between SOX factors and Wnt/β-catenin signaling in development and disease. Dev. Dyn. 239, 56–68 (2010)
Hogan, B. L. et al. Repair and regeneration of the respiratory system: complexity, plasticity, and mechanisms of lung stem cell function. Cell Stem Cell 15, 123–138 (2014)
Juan, J., Muraguchi, T., Iezza, G., Sears, R. C. & McMahon, M. Diminished WNT → β-catenin → c-MYC signaling is a barrier for malignant progression of BRAFV600E-induced lung tumors. Genes Dev. 28, 561–575 (2014)
Hsu, P. D. et al. DNA targeting specificity of RNA-guided Cas9 nucleases. Nature Biotechnol. 31, 827–832 (2013)
Fu, Y. et al. High-frequency off-target mutagenesis induced by CRISPR-Cas nucleases in human cells. Nature Biotechnol. 31, 822–826 (2013)
Wu, X. et al. Genome-wide binding of the CRISPR endonuclease Cas9 in mammalian cells. Nature Biotechnol. 32, 670–676 (2014)
Kuscu, C., Arslan, S., Singh, R., Thorpe, J. & Adli, M. Genome-wide analysis reveals characteristics of off-target sites bound by the Cas9 endonuclease. Nature Biotechnol. 32, 677–683 (2014)
Shalem, O. et al. Genome-scale CRISPR-Cas9 knockout screening in human cells. Science 343, 84–87 (2014)
Heigwer, F., Kerr, G. & Boutros, M. E-CRISP: fast CRISPR target site identification. Nature Methods 11, 122–123 (2014)
Psarras, S. et al. Gene transfer and genetic modification of embryonic stem cells by Cre- and Cre-PR-expressing MESV-based retroviral vectors. J. Gene Med. 6, 32–42 (2004)
Jackson, E. L. et al. The differential effects of mutant p53 alleles on advanced murine lung cancer. Cancer Res. 65, 10280–10288 (2005)
DuPage, M., Dooley, A. L. & Jacks, T. Conditional mouse lung cancer models using adenoviral or lentiviral delivery of Cre recombinase. Nature Protocols 4, 1064–1072 (2009)
Li, H. & Durbin, R. Fast and accurate long-read alignment with Burrows-Wheeler transform. Bioinformatics 26, 589–595 (2010)
McKenna, A. et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 20, 1297–1303 (2010)
Li, H. et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 25, 2078–2079 (2009)
Wang, K., Li, M. & Hakonarson, H. ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 38, e164 (2010)
Breese, M. R. & Liu, Y. NGSUtils: a software suite for analyzing and manipulating next-generation sequencing datasets. Bioinformatics 29, 494–496 (2013)
Quinlan, A. R. & Hall, I. M. BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26, 841–842 (2010)
Thorvaldsdóttir, H., Robinson, J. T. & Mesirov, J. P. Integrative Genomics Viewer (IGV): high-performance genomics data visualization and exploration. Brief. Bioinform. 14, 178–192 (2013)
Acknowledgements
We thank D. McFadden and Y. Soto-Feliciano for critical reading of the manuscript, H. Yin, S. Levine and T. Mason for MiSeq sequencing support, R. Stott, J. Bartlebaugh and C. Shivalila for technical assistance and K. Cormier and C. Condon from the Hope Babette Tang (1983) Histology Facility for technical support. This work was supported by the Howard Hughes Medical Institute, the Ludwig Center for Molecular Oncology at MIT and in part by Cancer Center Support (core) grant P30-CA14051 from the National Cancer Institute. T.P. is supported by the Hope Funds for Cancer Research. T.J. is a Howard Hughes Medical Institute Investigator, the David H. Koch Professor of Biology, and a Daniel K. Ludwig Scholar.
Author information
Authors and Affiliations
Contributions
F.J.S.-R, T.P. and T.J. designed the study; F.J.S.-R, T.P., R.R., M.R.B. and L.S. performed experiments; T.T. generated KrasLSL-G12D/+; Apcfl/fl data; A.B. conducted bioinformatic analyses; N.S.J. generated GG cells; R.T.B. provided pathology assistance; W.X. gave conceptual advice; F.J.S.-R, T.P. and T.J. wrote the manuscript with comments from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Illumina MiSeq sequence datasets have been deposited into the NCBI repository under BioProjectID PRJNA256245.
Extended data figures and tables
Extended Data Figure 1 In vitro validation of pSECC.
a, The Green-Go Cre-reporter cell line used to validate pSECC lentiviruses in vitro. Upon infection with a Cre-containing lentivirus, such as pSECC, cells become GFP+, allowing for purification of pSECC-containing cells by FACS. Red and blue triangles denote pairs of loxP sites, with red loxP sites being able to recombine only with other red loxP sites and blue loxP sites being able to recombine only with other blue loxP sites. b, Validation of sgPten-pSECC. Numbers below the bands denote quantitation of protein level relative to empty vector control. c, Validation of sgNkx2-1-pSECC in a cell line that expresses Nkx2-1. d, e, Validation of sgTom-pSECC by fluorescence activated cell sorting (FACS). Briefly, a cell line obtained from a KrasLSL-G12D/+; p53fl/fl;Rosa26LSL-tdTomato/LSL-tdTomato mouse was infected with either empty-pSECC (d) or sgTom-pSECC (e) and cultured for 10 days post-infection, after which time the cells were collected and analysed by FACS.
Extended Data Figure 2 In vivo validation of pSECC.
a, Representative H&E and tdTomato IHC staining of serial sections from lung tumours of KrasLSL-G12D/+; p53fl/fl;Rosa26LSL-tdTomato/LSL-tdTomato mice infected with Empty-pSECC. b–d, Representative H&E and IHC staining of serial sections from negative (b), mixed (c) and positive (d) lung tumours of KrasLSL-G12D/+; p53fl/fl;Rosa26LSL-tdTomato/LSL-tdTomato mice infected with sgTom-pSECC (n = 6). e, Distribution of lung tumours from all mice infected with sgTom-pSECC (n = 6) that were scored as negative, mixed or positive based on tdTomato IHC.
Extended Data Figure 3 Histological analysis of lung tumours obtained from mice infected with pSECC lentiviruses.
a–e, Representative H&E images of lung tumours obtained from mice infected with Empty-pSECC (a), sgTom-pSECC (b), sgNkx2-1-pSECC (c), sgPten-pSECC (d), and sgApc-pSECC (e). f, g, Quantification of tumour burden (total tumour area/total lung area) in KrasLSL-G12D/+ (f) or KrasLSL-G12D/+; p53fl/fl (g) animals 10 weeks after infection with pSECC lentiviruses expressing: control (empty or sgTom, KrasLSL-G12D/+ (n = 4) and KrasLSL-G12D/+; p53fl/fl (n = 7)), sgNkx2-1 (KrasLSL-G12D/+ (n = 2) and KrasLSL-G12D/+; p53fl/fl (n = 6)), sgApc (KrasLSL-G12D/+ (n = 3) and KrasLSL-G12D/+; p53fl/fl (n = 6)) and sgPten (KrasLSL-G12D/+ (n = 4) and KrasLSL-G12D/+; p53fl/fl (n = 3)). h, Quantification of BrdU incorporation (BrdU+ cells per mm2) to assess proliferation of tumour cells from lung tumours in KrasLSL-G12D/+; p53fl/fl animals 10 weeks after infection with pSECC lentiviruses expressing: control (empty or sgTom, n = 4 tumours), sgNkx2-1 (n = 11 tumours), sgApc (n = 10 tumours) and sgPten (n = 15 tumours). Mice were given a pulse of BrdU for 4 h before being euthanized. n.s., not significant, *P < 0.05, **P < 0.01, ***P < 0.001 obtained from two-sided Student’s t-test. All error bars denote s.e.m.
Extended Data Figure 4 IHC-based analysis of mice infected with sgNkx2-1-pSECC.
a–c, Negative (a), mixed (b) and positive (c) lung tumours of mice infected with sgNkx2-1-pSECC. d, Distribution of Nkx2-1 IHC staining status in all sgNkx2-1-pSECC infected animals (n = 8) represented as percent of negative, mixed and positive tumours. Positive tumour, ∼100% of the tumour cells stained positive for Nkx2-1. Mixed tumour, at least ∼30% of tumour cells stained positive for Nkx2-1. Negative tumour, < 25% of the tumour cells stained positive for Nkx2-1.
Extended Data Figure 5 IHC-based analysis of mice infected with sgPten-pSECC.
a–c, Negative (a), mixed (b) and positive (c) lung tumours of mice infected with sgPten-pSECC (n = 9). Positive tumour, ∼100% of the tumour cells stained positive for Pten. Mixed tumour, at least ∼30% of tumour cells stained positive for Pten. Negative tumour, < 25% of the tumour cells stained positive for Pten. Dashed line in b demarcates the positive/negative tumour area.
Extended Data Figure 6 IHC-based analysis of KrasLSL-G12D/+- and KrasLSL-G12D/+; p53fl/fl-sgApc tumours.
a, Representative H&E and IHC staining of serial sections from KrasLSL-G12D/+; p53fl/fl-sgTom (control, denoted as KP-sgTom here), KrasLSL-G12D/+-sgApc (denoted as K-sgApc here) and KrasLSL-G12D/+; p53fl/fl-sgApc (denoted as KP-sgApc here) lung tumours. CCSP, Clara cell secretory protein; SP-C, surfactant protein C. b, Contingency table demonstrating a statistically significantly higher number of β-catenin/Sox9 double-positive tumours in KrasLSL-G12D/+; p53fl/fl-sgApc mice (29/33 tumours, 88%) vs K-sgApc mice (41/58 tumours, 71%) (one-sided chi-square test, P < 0.05). c, Percentage of all tumours that stained positive for nuclear β-catenin that stained positive or negative for Sox9 in KrasLSL-G12D/+- and KrasLSL-G12D/+; p53fl/fl-sgApc mice. d, Contingency table demonstrating a statistically significantly higher number of tumours with Nkx2-1 low/negative areas (which are also SP-C low/negative) in sgApc-pSECC animals compared to sgTom-pSECC control animals (two-sided Fisher’s exact test, P < 0.0001). e, Representative IHC staining of serial sections from an Nkx2-1 Low/Neg lung tumour obtained from a KrasLSL-G12D/+; Apcfl/fl mouse 18 weeks after infection with Adeno-Cre. Inset shows Sox9 staining. Low/neg = tumour that had areas with clear downregulation or complete loss of Nkx2-1 or SP-C as assessed by IHC staining.
Extended Data Figure 7 Representative examples of indels observed in lungs and tumours from mice infected with pSECC lentiviruses.
a–c, Representative indels observed in the Nkx2-1 (a), Pten (b) and Apc (c) locus from sgNkx2-1T1, sgPtenL1 and sgApcT3 samples, respectively. Left panel, details of sequence alignments around the PAM sequence. Right panel, overview of sequence alignments around the PAM sequence. Deletions and insertions are highlighted in black and purple bars, respectively. Inset in a depicts a magnification of an insertion. d, Distribution of indels (in-frame insertions, frameshift insertions, in-frame deletions and frameshift deletions) observed in samples from mice infected with sgNkx2-1-pSECC, sgPten-pSECC and sgApc-pSECC. Amp, mutations across whole PCR amplicon; PAM, mutations across 7 base pair region upstream of the PAM sequence. e, Table summarizing percentages of indels from total mutant reads (left percentage indicates Amp (mutations across whole PCR amplicon) and right percentage indicates PAM (mutations across 7 base pair region upstream of the PAM sequence). All error bars denote s.e.m.
Extended Data Figure 8 Off-target analysis.
a–i, Analysis of off-target editing for sgNkx2-1 (a–c), sgPten (d–f) and sgApc (g–i). Briefly, potential off-target cutting at the top three predicted off-target sites (obtained from (http://crispr.mit.edu/); see Supplementary Table 2) for each sgRNA was assayed by Illumina MiSeq. Each plot corresponds to the fraction of bases mutated per position in 10 bp flanks on either side of the PAM sequence (highlighted in red). Samples were obtained from entire lobes (L) from mice 10 weeks after infection with pSECC lentiviruses expressing sgNkx2-1, sgPten, sgApc or sgTom (control).
Supplementary information
Supplementary Information.
This file contains the reference sequences for Nkx2.1, Pten, Apc and top three predicted off-targets of sgNkx2.1, sgPten and sgApc. (PDF 132 kb)
Supplementary Table 1
This file contains the sgRNA sequences and Primer sequences (XLSX 44 kb)
Supplementary Table 2
The file contains the top 25 predicted off-targets for sgNkx2.1, sgPten and sgApc. Score is likelihood of off-target binding. (XLSX 52 kb)
Supplementary Table 3
This file contains the MiSeq counts and quantification of indels (in-frame insertions, frameshift insertions, in-frame deletions and frameshift deletions) observed in purity-corrected samples from mice infected with sgApc-pSECC, sgNkx2.1-pSECC and sgPten-pSECC. (XLSX 29 kb)
Supplementary Table 4
This file contains the read mapping statistics. (XLSX 13 kb)
Supplementary Table 5
This file contains the mutation calls for Nkx2.1 and related OTs. (XLSX 843 kb)
Supplementary Table 6
This file contains the mutation calls for Pten and related OTs. (XLSX 716 kb)
Supplementary Table 7
The file contains the mutation calls for Apc and related OTs. (XLSX 781 kb)
Rights and permissions
About this article
Cite this article
Sánchez-Rivera, F., Papagiannakopoulos, T., Romero, R. et al. Rapid modelling of cooperating genetic events in cancer through somatic genome editing. Nature 516, 428–431 (2014). https://doi.org/10.1038/nature13906
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature13906
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.