Abstract
The eukaryotic RNA exosome processes and degrades RNA by directing substrates to the distributive or processive 3′ to 5′ exoribonuclease activities of Rrp6 or Rrp44, respectively. The non-catalytic nine-subunit exosome core (Exo9) features a prominent central channel. Although RNA can pass through the channel to engage Rrp44, it is not clear how RNA is directed to Rrp6 or whether Rrp6 uses the central channel. Here we report a 3.3 Å crystal structure of a ten-subunit RNA exosome complex from Saccharomyces cerevisiae composed of the Exo9 core and Rrp6 bound to single-stranded poly(A) RNA. The Rrp6 catalytic domain rests on top of the Exo9 S1/KH ring above the central channel, the RNA 3′ end is anchored in the Rrp6 active site, and the remaining RNA traverses the S1/KH ring in an opposite orientation to that observed in a structure of a Rrp44-containing exosome complex. Solution studies with human and yeast RNA exosome complexes suggest that the RNA path to Rrp6 is conserved and dependent on the integrity of the S1/KH ring. Although path selection to Rrp6 or Rrp44 is stochastic in vitro, the fate of a particular RNA may be determined in vivo by the manner in which cofactors present RNA to the RNA exosome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Wasmuth, E. V. & Lima, C. D. in The Enzymes (eds Chanfreau, G.F. & Tamanoi, F. ) Ch. 31, 53–76 (Elsevier, 2012)
Mitchell, P., Petfalski, E., Shevchenko, A., Mann, M. & Tollervey, D. The exosome: A conserved eukaryotic RNA processing complex containing multiple 3′ to 5′ exoribonucleases. Cell 91, 457–466 (1997)
Tomecki, R. et al. The human core exosome interacts with differentially localized processive RNases: hDis3 and hDis3L. EMBO J. 29, 2342–2357 (2010)
Houseley, J. & Tollervey, D. The many pathways of RNA degradation. Cell 136, 763–776 (2009)
Briggs, M. W., Burkard, K. T. & Butler, J. S. Rrp6p, the yeast homologue of the human PM-Scl 100-kDa autoantigen, is essential for efficient 5.8S rRNA 3′ end formation. J. Biol. Chem. 273, 13255–13263 (1998)
Yang, W., Lee, J. Y. & Nowotny, M. Making and breaking nucleic acids: two-Mg+2 ion catalysis and substrate specificity. Mol. Cell 22, 5–13 (2006)
Beese, L. S. & Steitz, T. A. Structural basis for the 3′-5′ exonuclease activity of Escherichia coli DNA polymerase I: a two metal ion mechanism. EMBO J. 10, 25–33 (1991)
Stead, J. A., Costello, J. L., Livingstone, M. J. & Mitchell, P. The PMC2NT domain of the catalytic exosome subunit Rrp6p provides the interface for binding with its cofactor Rrp47p, a nucleic acid-binding protein. Nucleic Acids Res. 35, 5556–5567 (2007)
Callahan, K. P. & Butler, J. S. Evidence for core exosome independent function of the nuclear exoribonuclease Rrp6p. Nucleic Acids Res. 36, 6645–6655 (2008)
Midtgaard, S. F. et al. Structure of the nuclear exosome component Rrp6p reveals an interplay between the active site and the HRDC domain. Proc. Natl Acad. Sci. USA 103, 11898–11903 (2006)
Januszyk, K., Liu, Q. & Lima, C. D. Activities of human RRP6 and structure of the human RRP6 catalytic domain. RNA 17, 1566–1577 (2011)
Makino, D. L., Baumgärtner, M. & Conti, E. Crystal structure of an RNA-bound 11-subunit eukaryotic exosome complex. Nature 495, 70–75 (2013)
Wasmuth, E. V. & Lima, C. D. Exo- and endoribonucleolytic activities of yeast cytoplasmic and nuclear RNA exosomes are dependent on the noncatalytic core and central channel. Mol. Cell 48, 133–144 (2012)
Assenholt, J. et al. Exonucleolysis is required for nuclear mRNA quality control in yeast THO mutants. RNA 14, 2305–2313 (2008)
Liu, Q., Greimann, J. C. & Lima, C. D. Reconstitution, activities, and structure of the eukaryotic RNA exosome. Cell 127, 1223–1237 (2006)
Bonneau, F., Basquin, J., Ebert, J., Lorentzen, E. & Conti, E. The yeast exosome functions as a macromolecular cage to channel RNA substrates for degradation. Cell 139, 547–559 (2009)
Drążkowsak, K. et al. The RNA exosome complex central channel controls both exonuclease and endonuclease Dis3 activities in vivo and in vitro. Nucleic Acids Res. 41, 3845–3858 (2013)
Liu, J. J. et al. Visualization of distinct substrate-recruitment pathways in the yeast exosome by EM. Nature Struct. Mol. Biol. 21, 95–102 (2014)
Milligan, L. et al. A yeast exosome cofactor, Mpp6, functions in RNA surveillance and in the degradation of noncoding RNA transcripts. Mol. Cell. Biol. 28, 5446–5457 (2008)
LaCava, J. et al. RNA degradation by the exosome is promoted by a nuclear polyadenylation complex. Cell 121, 713–724 (2005)
Makino, D. L., Halbach, F. & Conti, E. The RNA exosome and proteasome: common principles of degradation control. Nature Rev. Mol. Cell Biol. 14, 654–660 (2013)
Kish-Trier, E. & Hill, C. P. Structural biology of the proteasome. Annu. Rev. Biophys. 42, 29–49 (2013)
Sen, M. et al. The ClpXP protease unfolds substrates using a constant rate of pulling but different gears. Cell 155, 636–646 (2013)
Effantin, G., Maurizi, M. R. & Steven, A. C. Binding of the ClpA unfoldase opens the axial gate of ClpP peptidase. J. Biol. Chem. 285, 14834–14840 (2010)
Cha, S. S. et al. Crystal structure of Lon protease: molecular architecture of gated entry to a sequestered degradation chamber. EMBO J. 29, 3520–3530 (2010)
Greimann, J. C. & Lima, C. D. Reconstitution of RNA exosomes from human and Saccharomyces cerevisiae cloning, expression, purification, and activity assays. Methods Enzymol. 448, 185–210 (2008)
The PyMOL Molecular Graphics System, Version 1.5.0.4 Schrödinger, LLC
Smart, O. S., Neduvelil, J. G., Wang, X., Wallace, B. A. & Sansom, M. S. HOLE: a program for the analysis of the pore dimensions of ion channel structural models. J. Mol. Graph. 14, 354–360 (1996)
Mossessova, E. & Lima, C. D. Ulp1-SUMO crystal structure and genetic analysis reveal conserved interactions and a regulatory element essential for cell growth in yeast. Mol. Cell 5, 865–876 (2000)
Otwinowski, Z. & Minor, W. in Methods in Enzymology (eds Carter, C. W. Jr & Sweet, R. M. ) Vol. 276, 307–326 (Academic Press, 1997)
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D 66, 213–221 (2010)
McCoy, A. J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007)
Jones, T. A., Zou, J. Y., Cowan, S. W. & Kjeldgaard, M. Improved methods for building protein models in electron density maps and the location of errors in these models. Acta Crystallogr. A 47, 110–119 (1991)
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D 66, 486–501 (2010)
Ashkenazy, H., Martz, E., Pupko, T. & Ben-Tal, N. ConSurf 2010: Calculating evolutionary conservation in sequence and structure of proteins and nucleic acids. Nucleic Acids Res. 38, W529–W533 (2010)
Chen, V. B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D 66, 12–21 (2010)
Acknowledgements
We thank NE-CAT beamlines (Advanced Photon Source) supported by P41GM103403 (NIH NIGMS). APS is supported by the US Department of Energy, Office of Basic Energy Sciences, under Contract No. DE-AC02-06CH11357. Beamline X29 (National Synchrotron Light Source) supported by the US Department of Energy, the Office of Basic Energy Sciences and P41RR012408 (NIH NCRR) and P41GM103473 (NIH NIGMS). Research reported in this publication was supported by the National Institute of General Medical Sciences of the National Institutes of Health under award numbers F31GM097910 (E.V.W.) and R01GM079196 (C.D.L.). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. C.D.L. is an investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
E.V.W. and C.D.L. designed and E.V.W. performed experiments for the S. cerevisiae exosomes. E.V.W. and C.D.L. determined the structure. K.J. and C.D.L. designed and K.J. performed the experiments for the Homo sapiens exosome. E.V.W. and C.D.L. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 View highlighting electron densities covering RNA in the Exo10Rrp6–poly(A) structure.
a, Simulated annealing 2Fo − Fc omit map contoured at 1.0σ. b, Simulated annealing Fo − Fc omit map contoured at 2.0σ. c, Final 2Fo − Fc omit map contoured at 1.0 σ. RNA and phosphate oxygen, nitrogen, carbon and phosphate atoms are coloured red, blue, orange and dark orange, respectively. Electron density maps shown as wire mesh coloured blue for 2Fo − Fc maps and green for the Fo − Fc map. Rrp6, Rrp40, Rrp4 and Csl4 are labelled and shown in cartoon representation in teal, magenta, green and light blue, respectively. d, 20 to 24 nucleotides of poly(A) RNA are present in crystals of Exo10Rrp6. Crystals of Exo10Rrp6 bound to poly(A)24 RNA were first washed by two rounds of transfer to 1 μl well solution, then dissolved in water and TBE-urea sample buffer, and analysed by 15% TBE-urea PAGE. A drop without crystals was run as a control. RNA was stained by Sybr Gold.
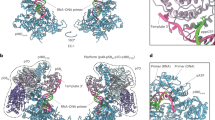
Extended Data Figure 2 Rrp6 features within Exo10Rrp6.
a, The primary sequence of the catalytic module of Rrp6 (EXO and HRDC) is highly conserved, whereas sequence conservation within the CTD varies. Rrp6 in Exo10Rrp6 is represented as a surface, and coloured according to sequence conservation as calculated by ConSurf35, coloured from red (highly conserved) to blue (variable). The Exo9 core is depicted as a transparent cartoon, with the S1/KH ring in orange, and the PH-like ring in grey. b, Superposed structures of the Rrp6 CTD from Exo10Rrp6 (residues 525–628; teal) and Exo10Rrp44+Rrp6Cterm (residues 532–557, 565–619; magenta; PDB 4IFD) reveal overall similarity for residues within the respective models. c, The conserved linker between the EXO and HRDC domain of Rrp6 adopts an α-helix when associated with the exosome core. Superposition of the Rrp6 catalytic domains from the structures of Exo10Rrp6 and Rrp6–AMP (PDB: 2HBL) shows residues Pro 424 to Asn 433 rearrange from a loop in the AMP-bound structure to a helix that is adjacent to a conserved, albeit disordered, loop in the S1 domain of Rrp4. These residues were previously thought to comprise a protein interaction module10. Rrp6 is represented as cartoons, with Rrp6 from the Exo10Rrp6 structure in teal, and the Rrp6–AMP structure in grey. Rrp4 is in light green and depicted as a cartoon with a transparent surface, with the S1 loop representing residues Gly 145 to Gln 158 shown as a dashed line.
Extended Data Figure 3 The Rrp6 CTD is required for interaction with human and yeast RNA exosome cores.
a, Schematic representation of Rrp6 including from N to C terminus the PMC2NT, EXO, HRDC and C-terminal (CTD) domains. EXO and HRDC comprise Rrp6CAT. Below are lines representing individual elements used in gel-filtration analysis in c and d along with amino acid numbering specific for S. cerevisiae (Sc) Rrp6 and Homo sapiens (Hs) RRP6. b, Gel-filtration profile for reconstituted human Exo10RRP6 (left) with SDS–PAGE analysis of the peak fractions (right). Subunits labelled in capital letters corresponding to yeast nomenclature for clarity. Apparent molecular weights denoted in kilodaltons. c, Interaction studies of human RRP6 with the HsExo9. The human RRP6 catalytic domain (residues 180–606) does not interact well with human Exo9; however, addition of RRP6 180–804 suggests that CTD residues 607–804 are important for stable interaction with Exo9. Reconstitution experiments were performed as described above on mixtures of HsExo9, HsExo9 with twofold excess of RRP6180–606 (the catalytic region), HsExo9 with twofold excess of RRP6180–804 (the catalytic region with a portion of the carboxy terminus), and RRP6180–606. Gel filtration analysis on RRP6180–804 in isolation was not possible as this construct is unstable in the absence of the SMT3 tag. Mixtures (0.15 ml at ∼2 mg ml−1) were dialysed overnight at 4 °C against reconstitution buffer (50 mM NaCl, 20 mM Tris-HCl pH 8.0, and 10 mM DTT). Mixtures were applied to a Superose 6 10/300 GL column (G.E. Health Sciences) equilibrated with reconstitution buffer with the resulting ultraviolet traces (upper panels) and analysed fractions (lower panel). d, Interaction studies of budding yeast Rrp6 with ScExo10Rrp44. Rrp6 (residues 128–733) and Rrp6CTD, but not Rrp6CAT, interact with Exo10Rrp44. Rrp6 (residues 128–733), Rrp6CAT (residues 128–518), the Rrp6CTD (residues 518–733), or both were incubated with Exo10Rrp44 for one hour on ice before analysis by size exclusion chromatography (Superose 6) in reconstitution buffer. Rrp6 and Rrp6CAT were added in 1.5-fold molar excess to Exo10Rrp44, while Rrp6CTD was added in threefold molar excess. For panel c and d, fractions were analysed by SDS–PAGE and protein detected with Sypro Ruby.
Extended Data Figure 4 RNA interactions within the Rrp6 active site.
a, b, View of RNA nucleotides 24 to 20 from the 3′ end in the active site of S. cerevisiae Rrp6 from the structure of Exo10Rrp6 (a) and the same RNA modelled into the active site of H. sapiens RRP6 (PDB: 3SAF) based on superposing the respective EXO domains (b). Residues involved in RNA contacts in the structure of yeast Exo10Rrp6 are shown as teal sticks in a; the corresponding residues in human are shown in b. In both structures, a catalytic aspartate is mutated to asparagine and labelled in red (N238 in yeast; N313 in human). RNA is shown as orange sticks, and magnesium ions are represented as green spheres. c, Structure of the yeast Rrp6 catalytic domain in complex with zinc (Zn; blue sphere) and manganese (Mn; yellow sphere) in complex with AMP (PDB 2HBL). Active site residues are depicted as in a, b. The conserved Tyr 361 side chain was mutated to alanine in this structure (labelled Y361A in red). d, Model of the Rrp6 active site constructed by superposing the 3′ nucleotide in the Exo10Rrp6 poly(A) structure to the AMP ligand from PDB 2HBL. Amino acid side chains are labelled and putative interactions between metal ligands, active site residues and RNA are depicted by dashed lines.
Extended Data Figure 5 Central channel mutations and their impact on Rrp6 degradation of AU-rich RNA.
a, 1 nM of reconstituted wild-type and mutant yeast Exo10Rrp6 and Exo11Rrp44/Rrp6 were incubated with 10 nM 49-nt 5′ fluorescein AU-rich RNA at 30 °C. Reaction intermediates were analysed after indicated time points by denaturing PAGE and imaged with a fluoroimager. Rrp6 activity decreases in the presence of the Rrp40 K107E/K108E/R110D and Csl4 R145A/R150A/R202D mutations in both Exo10Rrp6 and Exo11Rrp44/Rrp6 but is mostly unaffected by the channel occlusion in the Rrp45Large exosome. Rrp44 exoribonuclease activity is attenuated by mutations in both the S1/KH and PH-like ring in Exo11Rrp44/Rrp6. b, Exoribonuclease activities of wild-type and mutant forms of human Exo10RRP6 were performed with 5 nM 49-nt 5′ fluorescein AU-rich RNA and 50 nM protein at 37 °C. Reaction intermediates were analysed after indicated time points as described in a. Mutation studies reveal that the human RRP6 exoribonucleolytic activity decreases in the presence of a channel occlusion near the S1/KH ring (RRP41Large), while a channel occlusion deeper in the central channel (RRP45Large) of HsExo10RRP6 has little effect on activity.
Extended Data Figure 6 Ultraviolet crosslinking with 36-nt 4-thioU RNAs for yeast exosome complexes.
a, Raw data used to generate Fig. 4a in main text for poly(A) and AU-rich RNA. See Methods for crosslinking details. Presence or absence of crosslinks to given subunits is summarized in the table and denoted by colour-coded dots, with red corresponding to 4-thioU 6 nt from the 3′ end; purple for 4-thioU 21 nt from the 3′ end; and blue for 4-thioU 29 nt from the 3′ end. b, RNA–protein adducts observed in ultraviolet crosslinking to Exo10Rrp6 correspond to the S1/KH cap proteins. Exo10Rrp6 bearing N-terminal hexahistine tags on the S1/KH cap proteins Rrp4, Rrp40 and Csl4 (ref. 13) or tag-free variants were crosslinked to the three 4-thioU RNAs described above, and resolved by SDS–PAGE and imaged with a fluoroimager. In comparing the His6-tagged and tagless complexes, mobility shifts are apparent in crosslinks corresponding to the S1/KH cap proteins. c, 4-thioU RNAs are bona fide decay substrates for exosome complexes and catalytic subunits, Rrp6 and Rrp44. Conditions using substrate excess (10 nM RNA, 1 nM enzyme), 4-thioU poly(A) RNAs (left) and AU-rich RNAs (right) are comparably degraded to corresponding 36-nt RNAs lacking 4-thioU. Reactions were performed at 30 °C and stopped after indicated time points and resolved by denaturing PAGE and imaged with a fluoroimager. 4-thioU RNAs are colour-coded as in a and b.
Extended Data Figure 7 Ultraviolet crosslinking to AU-rich RNA to components of human Exo10RRP6.
a, Ultraviolet crosslinking of RNA to HsExo10RRP6 with open and occluded channels. Human RRP6 (Smt3–RRP6180–804), HsExo9 and wild type as well as three different pore occlusion mutants of HsExo10RRP6 were incubated and ultraviolet crosslinked to a 24-nt fluorescein-labelled AU-rich RNA substrate. Samples were analysed for the presence of RNA-protein crosslinks (left panel) or integrity of protein via Sypro staining (right panel). Mutant forms of HsExo10RRP6 with insertion loops in RRP41-L and RRP45-L are labelled with red asterisks. b, Identification of ultraviolet-induced crosslinks. Identification of RNA-protein crosslinks between RNA to cap components of the human Exo9 core (RRP4, RRP40, and CSL4) was facilitated by reconstituting His6-tagged versions of these subunits into HsExo9 complexes, performing crosslinking assays with a 36-nt fluorescein-labelled AU-rich RNA, and detecting slower mobility for the His6-tagged crosslinked adducts. Similarly, identification of RRP45-RNA adducts was based on changes in adduct mobility when using isoforms with wild-type carboxy termini (RRP451–456) and the long insertions (RRP45-L1–302). RRP6–RNA adducts were determined by crosslinking of a Smt3–RRP6180–804 fusion and comparison of 9- and 10-subunit exosomes (in a).
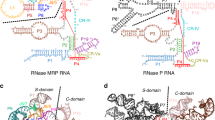
Extended Data Figure 8 RNA paths to Rrp6 and Rrp44.
Structure of an Exo11Rrp44/Rrp6 model derived by superposing the PH-like ring subunits in Exo10Rrp6 and Exo10Rrp44+6Cterm (PDB 4IFD). a, RNA paths to the Rrp6 active site. RNA (yellow) shown as derived from the Exo10Rrp6 poly(A) complex. Three views are provided from the top, side and backside of the complex with subunits coloured as in Fig. 1 in the main text (left), surfaces coloured according to conservation as calculated by ConSurf35 (middle) from red (highly conserved) to blue (variable), and surfaces coloured according to electrostatic potential as calculated by Pymol27 (right). Subunits that are visible are labelled in the left panel as is the RNA. Three putative paths that traverse the S1/KH ring into the Rrp6 active site are denoted by yellow arrows and labels (P1, P2, P3) in the top and side views and a fourth path (P4) under the Rrp6 catalytic domain is denoted in the backside view. Based on conservation, electrostatics and crosslinking we deem P1 or P2 as the most likely path of ingress for the incoming RNA substrate. The top and side views use cutaway surfaces to illustrate the putative paths and surface properties. b, RNA paths to the Rrp44 active site. Subunits and surfaces depicted as in a with the exception that the RNA is now derived from the structure of Exo10Rrp44+6Cterm (PDB 4IFD) with the RNA edited to remove the stem loop after nucleotide 36. Top, side and bottom views are depicted with the RNA path indicated by a yellow arrow with 5′ and 3′ ends labelled. The position of the central channel is also labelled and indicated by a yellow arrow in the top and bottom views. The top and side views show that RNA could pass by Rrp6 to penetrate the S1/KH and PH-like ring central channel.
Rights and permissions
About this article
Cite this article
Wasmuth, E., Januszyk, K. & Lima, C. Structure of an Rrp6–RNA exosome complex bound to poly(A) RNA. Nature 511, 435–439 (2014). https://doi.org/10.1038/nature13406
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature13406
This article is cited by
-
Nucleolar URB1 ensures 3′ ETS rRNA removal to prevent exosome surveillance
Nature (2023)
-
The nucleolus is the site for inflammatory RNA decay during infection
Nature Communications (2022)
-
Identification and application of piwi-interacting RNAs from seminal plasma exosomes in Cynoglossus semilaevis
BMC Genomics (2020)
-
EXOSC9 depletion attenuates P-body formation, stress resistance, and tumorigenicity of cancer cells
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.